Yixing Zeyuan Chemical Technology Co Ltd
We can supply: 1..Place of Origin China with Super quality by reasonable price. 2. sample orders are welcome 3.Customer Satisfaction is our priority and guarantee for the future. 4.Fast delivery by safe express way to all the country. 5.Has a
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:7782-61-8
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Purple crystal Purple crystal Assay [Fe(NO3)3·9H2O]
Cas:7782-61-8
Min.Order:1 Kilogram
FOB Price: $3.0 / 5.0
Type:Manufacturers
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:7782-61-8
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:7782-61-8
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages High purity, all above 98.5%, no impurities after dissolution We will test each batch to ensure quality OEM and private brand services designed for free Various cap colors available We can also provide MT1 peptide powd
Cas:7782-61-8
Min.Order:1 Kilogram
FOB Price: $2.0 / 3.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Chemical Name: Ferric nitrate nonahydrate CAS No.: 7782-61-8 Molecular Fomula:Fe(NO3)3•9H2O Chemical Structure: Molecular weight: M=404.2 Appearance: Purple or white HS CODE: 28342990 Appearance: Purple or white powder Storage:Store
Hubei Langyou International Trading Co., Ltd
High purity Ferric nitrate nonahydrate with high quality and best price cas:7782-61-8 1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competit
Cas:7782-61-8
Min.Order:10 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:7782-61-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Ocean Chemical Co., Ltd.
Ocean inorganic department is a professional supplier and exporter engaging in inorganic chemical materials and metal organic compounds. Over the past years, our company is committed to improving the product quality and developing new products, in
Cas:7782-61-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Ferric nitrate nonahydrate CAS:7782-61-8 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organi
Cas:7782-61-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:7782-61-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
1)quick response within 12 hours; 2)quality guarantee: all products are strictly tested by our qc, confirmed by qa and approved by third party lab in china, usa, canada, germany, uk, italy, france etc. 3) oem/odm available; 4) reasonable &
Cas:7782-61-8
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:7782-61-8
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:7782-61-8
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryHangzhou Fonlynn Health Technology Co., Ltd.
Packing: According to customer requirements Delivery time: In stock or depands Port of shipment: Ningbo/Shanghai/Qingdao OEM/ODM:Welcome Sample:We can offer our existing samples at once Transportation:Express/Sea/Air Port:Ningbo/Shanghai/Qingd
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:7782-61-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7782-61-8
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Ferric Nitrate CAS No.: 7782-61-8 Molecule Formula:FeH18N3O18 Molecule Weight:404.00 Purity: 98.0% Package: 25kg/bag Description:Light purple crystalline powder Manufacture Standards:Enterprise Standard TESTING ITEM
Cas:7782-61-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
Ferric nitrate nonahydrate Basic information Product Name: Ferric nitrate nonahydrate Synonyms: IRON(+3)NITRATE ENNEAHYDRATE;IRON (III) NITRATE;IRON(III) NITRATE-9-HYDRATE;IRON(III) NITRATE ENNEAHYDRATE;IRON (III) NITRATE, HYDROUS;IRON(III) NI
Cas:7782-61-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:7782-61-8
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:7782-61-8
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryHANGZHOU TIANYE CHEMICALS CO., LTD.
We product this chemical more than 10 years . We are very experience to export it to many countries, Our superior & stable quality , competitive price gain warm reception from our customers. Product Name:Ferric nitrate nonahydrate CAS No:7782
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:7782-61-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:7782-61-8
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquiryBluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:7782-61-8
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
1, High quality with competitive price:2, Fast and safe delivery3.Excellent pre-sales and after-sales service4. Well-trained and professional technologist and sales with rich experience in the field for 5-10 yearsAppearance:see detailed specification
Henan Tianfu Chemical Co., Ltd.
Our advantage:Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientif
Cas:7782-61-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:7782-61-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route

| Conditions | Yield |
|---|---|
| In nitric acid byproducts: H2SO4, NO, CO; stirring for 0.5 h at 25°C; condensing silane in a trap cooled with solid CO2, treating aq. layer with NaOH, evapn. to dryness, IR; | A n/a B 99% |


| Conditions | Yield |
|---|---|
| In nitric acid byproducts: H2SO4, NO, CO; stirring for 0.5 h at 25°C; condensing silane in a trap cooled with solid CO2, treating aq. layer with NaOH, evapn. to dryness, IR; | A 96% B n/a |


| Conditions | Yield |
|---|---|
| In water dissolving of Fe2O3 in excess of HNO3 for 30 - 45 days; not isolated, measuring of conc. of Fe(3+) by photometrically; |

| Conditions | Yield |
|---|---|
| In nitric acid Fe is dissolved in dild. HNO3 with evolution of gas, formation of Fe(NO3)3;; | |
| In nitric acid byproducts: basic iron(III)nitrate; Fe is dissolved in HNO3 (d = 1.115), formation of Fe(NO3)3 and basic iron(III)nitrate;; | |
| In nitric acid byproducts: basic iron(III)nitrate; aq. HNO3; Fe is dissolved in HNO3 (d = 1.115), formation of Fe(NO3)3 and basic iron(III)nitrate;; | |
| In nitric acid aq. HNO3; Fe is dissolved in dild. HNO3 with evolution of gas, formation of Fe(NO3)3;; | |
| In ethanol; water for 24h; |

| Conditions | Yield |
|---|---|
| In nitric acid storing of Fe for a longer period of time in fuming HNO3;; | |
| In nitric acid storing of Fe for a longer period of time in fuming HNO3;; |


| Conditions | Yield |
|---|---|
| In nitric acid Fe is dissolved in HNO3 (d = 1.073), formation of a mixture of Fe(NO3)2 Fe(NO3)3 and NH4NO3;; | |
| In nitric acid aq. HNO3; Fe is dissolved in HNO3 (d = 1.073), formation of a mixture of Fe(NO3)2 Fe(NO3)3 and NH4NO3;; |

-
-
7697-37-2
nitric acid

-
-
7439-89-6
iron

-
A
-
7664-41-7
ammonia

-
B
-
10102-44-0
Nitrogen dioxide

-
C
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In nitric acid Fe is dissolved in HNO3 (34%, d =1.21), at the beginning Fe is dissolved quickly with evolution of gas, then slowly without evolution of gas and formation of Fe(NO3)3 and traces of NO2 and NH3;; | |
| In nitric acid aq. HNO3; Fe is dissolved in HNO3 (34%, d =1.21), at the beginning Fe is dissolved quickly with evolution of gas, then slowly without evolution of gas and formation of Fe(NO3)3 and traces of NO2 and NH3;; |


| Conditions | Yield |
|---|---|
| In water addn. of 10 % soln. of NH4OH to soln. of Fe-salt (pptn.), filtration, washing (hot water) to remove NH4(1+), and SO4(2-), redissolving of Fe(OH)3 in 0.5 M HNO3; |


| Conditions | Yield |
|---|---|
| In tetrachloromethane byproducts: CO, H2, nitrogen oxides; | |
| In diethyl ether byproducts: CO, H2, nitrogen oxides; |


| Conditions | Yield |
|---|---|
| With nitric acid In nitric acid dissoln. of Fe in HNO3 (d > 1.115); formation of Fe(NO3)3 and basic iron nitrates;; | |
| With nitric acid In nitric acid byproducts: HNO2; dissoln. of Fe in a 20-fold excess of HNO3 at 8°C; first formation of Fe(NO3)2, which is oxidized by formed HNO2 to Fe(NO3)3;; | |
| With nitric acid In nitric acid dissoln. of Fe in HNO3 (d = 1.115); formation of Fe(NO3)3;; |
-
-
13463-40-6
iron pentacarbonyl

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In nitric acid fast react.;; |

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating (p(N2O4)=2.0 MPa, 105-135°C); DTA; |

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| With nitric acid byproducts: KNO3, H2O, O2; nascent oxygen formed; | |
| With HNO3 byproducts: KNO3, H2O, O2; nascent oxygen formed; |

| Conditions | Yield |
|---|---|
| In water Kinetics; byproducts: H2SO4, NO, NO2; heating of aq. soln. of HNO3 with rotating disk from natural pyrite crystals at 303 - 333 K; |

| Conditions | Yield |
|---|---|
| With nitric acid In nitric acid dissoln. of Fe in HNO3 (d > 1.05) at lower temp.; formation of Fe(NO3)2 and Fe(NO3)3;; | |
| With HNO3 In nitric acid aq. HNO3; dissoln. of Fe in HNO3 (d > 1.05) at lower temp.; formation of Fe(NO3)2 and Fe(NO3)3;; |

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| With nitric acid In water heating an aq. soln. of Fe(NO3)2 with HNO3; formation of Fe(NO3)3;; | |
| With HNO3 In water heating an aq. soln. of Fe(NO3)2 with HNO3; formation of Fe(NO3)3;; |

| Conditions | Yield |
|---|---|
| With nitric acid In water discoloration of the aq. soln. of FeF2 with HNO3; formation of FeF3 and Fe(NO3)3;; pptn. as a solid mixt. on evapn.;; | |
| With HNO3 In water discoloration of the aq. soln. of FeF2 with HNO3; formation of FeF3 and Fe(NO3)3;; pptn. as a solid mixt. on evapn.;; |

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In solid byproducts: H2O; 160°C to 170°C; |
-
-
7782-61-8
ferric nitrate

-
-
28230-32-2
3-hydroxy-3,4-dihydrobenzotriazine-4-one

-
-
261168-39-2
tris[3-hydroxy-1,2,3-benzotriazine-4(3H)-one]iron(III)

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | 100% |

| Conditions | Yield |
|---|---|
| With 1,2-ethanediol In water byproducts: C2H2O4(2-); by thermal decompn. of complex combination isolated in heat by oxidn. inaq. soln. of 1,2-ethanediol to C2H2O4(2-) by Ni(II), Zn(II), Fe(III) ni trates; heated at 500-900°C for 3 h; detd. by XRD; | 100% |

-
-
18362-64-6
2,6-dimethyl-3,5-heptanedione

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25 - 28℃; for 1.5h; | 95.9% |

| Conditions | Yield |
|---|---|
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.66 (ammonia), heating at 192°C for 2 h (microwaves); centrifugation, washing; powder X-ray diffraction; | 95% |
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.66 (ammonia), heating at 164°C for 2 h (microwaves); centrifugation, washing; powder X-ray diffraction; | 88% |
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.66 (ammonia), heating at 164°C for 4 mins. (microwaves); centrifugation, washing; powder X-ray diffraction; | 85% |

| Conditions | Yield |
|---|---|
| In hexane; water at 40℃; | 95% |

| Conditions | Yield |
|---|---|
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.71 (ammonia), heating at 164°C for 4 mins. (microwaves); centrifugation, washing; powder X-ray diffraction; | A 94% B n/a |


-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In water P-compd. was dissolved in the excess of 0.14 M aq. Fe-salt, stirring for10 min, solid KCl was added then soln. was treated with satd. aq. KCl; solid was filtered off, dried in air, elem. anal.; | 94% |


| Conditions | Yield |
|---|---|
| In water at 60 - 550℃; pH=5.0; | 94% |


| Conditions | Yield |
|---|---|
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.73 (ammonia), heating at 164°C for 4 mins. (microwaves); centrifugation, washing; powder X-ray diffraction; | 92% |
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.95 (ammonia), heating at 164°C for 2 h (microwaves); centrifugation, washing; powder X-ray diffraction; | 90% |
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.90 (ammonia), heating at 192°C for 2 h (microwaves); centrifugation, washing; powder X-ray diffraction; | 81% |

| Conditions | Yield |
|---|---|
| With nitric acid; tartaric acid In neat (no solvent) nitrates dissolved in HNO3, tartaric acid added, evapd. by heating understirring, heated at 150 - 160 °C for 1 h, calcined at 400 - 600 °C for 2 h; powder XRD, TG; | 92% |
| With citric acid In neat (no solvent) sols prepared from soln. of nitrates and citric acid in ethylene glycol monomethyl ether (Bi excess), spin coated, dried at 80 °C for 30 min, annealed at 200 and 400 °C for 5 min after each coating, annealed at 630 °C for 300-400 s; powder XRD; | |
| In water Bi- and Fe-nitrates in 1.05:1 molar ratio dissolved in double distd. H2Owith few drops of nitric acid under constant heating and stirring for 1 h, sprayed on LaAlO3 substrate maintained at 300+/-5 °C; |

| Conditions | Yield |
|---|---|
| With oxygen In water at 300℃; for 1h; | 91.6% |


| Conditions | Yield |
|---|---|
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.90 (ammonia), heating at 164°C for 2 h (microwaves); centrifugation, washing; powder X-ray diffraction; | 90% |
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.66 (ammonia), heating at 192°C for 2 h (microwaves); centrifugation, washing; powder X-ray diffraction; | 83% |
| With formaldehyd; triethylamine In water byproducts: (CH2)6N4, H2O, {(C2H5)3NH}NO3; addn. of polyvinyl alcohol to metal nitrate soln., heated with stirring, addn. of mixt. of (NH4)2CO3 or NH4HCO3, triethylamine and formaline to soln., warmed, pptn. (10-12 h); filtered, washed, dried in an oven, heated (1000°C, 8-10 h), XRD, EDX; |

| Conditions | Yield |
|---|---|
| In nitric acid Bi(NO3)3 and Fe(NO3)3 dissolved in 2N nitric acid, oxalic acid added under stirring, heated to boiling temp., evapd., dried at 130°C for 30 min, calcined at 600°C for 2 h; XRD, SEM, TG/DTA; | 90% |


| Conditions | Yield |
|---|---|
| With LiOH In methanol N2; ligand hydrochloride (1 equiv.) and LiOH (2 equiv.) stirred in MeOH for 20 min, evapd., dissolved (MeOH), Fe(NO3)3 (1 equiv.) added dropwise, stirred for 5 min; filtered off, elem. anal.; | 90% |


| Conditions | Yield |
|---|---|
| With sodium dodecylsulfonate In ethanol; water Fe-compd. and o-nitrobenzoic acid dissolved in ethanol-water (5:1) solution respectively, two solution mixed, sodium dodecylsulfonate added, pH ajusted slowly to 2.7 with stirring, aged for 8 h; filtered, washed with ethanol-water (5:1), dried in air at 80°C; | 89% |

| Conditions | Yield |
|---|---|
| With NaHCO3 In methanol; water solid NaHCO3 was added during 20 min to a well-stirred soln. 3-(4-pyridyl)pentane-2,4-dione in CH3OH and H2O, the mixt. was combined with a soln. of Fe-salt in H2O; ppt. was dissolved in dry methanol, slow evapd.; | 88% |
-
-
136950-29-3
N-(5-sulphonyl-8-hydroxyquinoline)-3,5-dimethyl-2-pyrazoline 5-one

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In ethanol refluxing org. ligand and Fe(NO3)3 (molar ratio 2:1) with stirring at 50-60°C, pptn.; filtration, washing (alcohol), drying; elem. anal.; | 87% |

| Conditions | Yield |
|---|---|
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.03 (ammonia), heating at 192°C for 2 h (microwaves); centrifugation, washing; powder X-ray diffraction; | 86% |
| With ammonia In water High Pressure; soln. of nitrates, adjustment of pH to 8.05 (ammonia), heating at 164°C for 2 h or 4 mins. (microwaves); centrifugation, washing; powder X-ray diffraction; | 84% |
| In water High Pressure; rapid expansion of supercritical mixed soln. of Zn(NO3)2 and Fe(NO3)3 (100 MPa, 773 K, dwelling time of 6-7 s, concn. of 0.05 M, pH 1.5), isothermal 1-h anneal at temp. from the range of 823-873 K; SEM, TEM, X-ray diffraction anals.; | 30% |
-
-
136950-28-2
5-<3'-methyl-5'-oxo-4',5'-dihydropyrazol-1-yl-sulfonyl>-8-quinolinol

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In ethanol refluxing org. ligand and Fe(NO3)3 (molar ratio 2:1) with stirring at 50-60°C, pptn.; filtration, washing (alcohol), drying; elem. anal.; | 84% |

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In ethanol; water aq. Te-salt soln. in min. solvent addn. to metal salt soln. (EtOH), mixt. stirring for 2 h; ppt. filtration, washing (water, EtOH), air drying; elem. anal.; | 83.4% |

-
-
7782-61-8
ferric nitrate

-
-
26684-72-0
5-diethylamino-3-pyridin-4-yl-pentan-1-ol

| Conditions | Yield |
|---|---|
| With ammonium chloride In ammonia | 83% |
-
-
7732-18-5
water

-
-
7782-61-8
ferric nitrate

-
-
101732-00-7
N,N'-bis[1,3-benzodioxol-5-ylmethylene]ethane-1,2-diamine

-
-
1231901-00-0
Fe(N,N'-bis[1,3-benzodioxol-5-ylmethylene]ethane-1,2-diamine)(NO3)3(H2O)

| Conditions | Yield |
|---|---|
| In methanol; water Fe-compd. dissolved in CH3OH-H2O, added to refluxing diamine-compd. in CH3OH (molar ratio Fe-diamine=1:1), refluxed for 2 to 3 h; concentrated, washed with Et2O and petroleum ether, filtered, washed with suitable solvents, dried over anhydrous CaCl2, elem. anal.; | 82% |


| Conditions | Yield |
|---|---|
| In water the ligand added to a hot soln. of metal salt; filtered, stand at room temp. for several d; elem. anal.; | 81% |

-
-
136950-27-1
N-(5-sulphonyl-8-hydroxyquinoline)-3-amino-2-pyrazoline 5-one

-
-
7782-61-8
ferric nitrate

| Conditions | Yield |
|---|---|
| In ethanol refluxing org. ligand and Fe(NO3)3 (molar ratio 2:1) with stirring at 50-60°C, pptn.;; filtration, washing (alcohol), drying; elem. anal.;; | 80% |

| Conditions | Yield |
|---|---|
| In ethanol refluxing for 2 h; vol. reduction (pptn.); | 80% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View


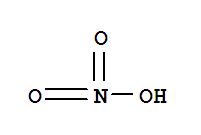






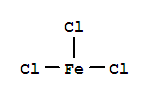
 O,
O, Xi
Xi