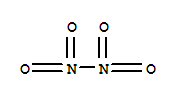Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7727-37-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Henan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:7727-37-9
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hunan Longxianng Runhui Trading Co.,Ltd
Nitrogen N2 Package:depends Application:To fill light bulbs, Liquid nitrogen Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport) or by sea LCL/FCL for large amount.
HENAN SUNLAKE ENTERPRISE CORPORATION
Our Services 1.Certificate Of Analysis (COA) 2.Material Safety Data Sheet (MSDS) 3.Route of synthesis (ROS) 4.Method of Aanlysis (MOA) 5.Nuclear Magnetic Resonance (NMR) 6.Packing pictures and loading video before loading 7.Free S
Changzhou Extraordinary Pharmatech co.,LTD
Changzhou Extraordinary Pharmatech co., LTD. As a leading chemical manufacturer and supplier in China.DAS authentication is passed.We can provide the popular precursor chemicals, we have our own strong R & D team, have our own laboratories and fa
HuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of pl
Cas:7727-37-9
Min.Order:1 Kilogram
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryChengdu Taiyu Industrial Gases Co., Ltd.
Nitrogen N2 CAS No.: 7727-37-9 EINECS No.: 231-783-9 UN No.: UN1066 Purity: 99.999%-99.9999% Dot Class: 2.2 Appearance: Colorless Grade Standard: Food Grade, Industrial Grade, Electronic Grade. In various analytical inst
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Chungking Joyinchem Co., Ltd.
Joyinchem have been committed to chemical supply for several years and have built good cooperation records with multinational chemical corporations and importers from all over the world. Our services include:-Spot goods-Contract manufacturing-Custom
Hebei Minshang Biotechnology Co., Ltd
Nitrogen N2 Application:To fill light bulbs, Liquid nitrogen
Springchem New Material Technology Co.,Limited
≥99%, stockAppearance:A colorless odorless gas. Package:1kg/bottle;50kg/100kg/drum Application:Used in food processing, in purging air conditioning and refrigeration systems, and in pressurizing aircraft tires. Transportation:by air, by sea
Skyrun Industrial Co.,Ltd
Good Price, Sufficient capacity Application:Catalytic agent; Petrochemical additive; Used in organic synthesis
Hebei Ruishun Trade Co.,Ltd
Supply top quality products with a reasonable price Application:api
Chemical Co.Ltd
Nitrogen Application:for chemcial
NovaChemistry
high purity Application:Drug intermediates Materials intermediates and active molecules
Sigma-Aldrich Chemie GmbH
Application:Nitrogen is a colourless, odourless, and asphyxiating gas that is slightly lighter than air. Nitrogen is used as a carrier gas in gas chromatography, an inert gas in chemical and pharmaceutical in...
Synthetic route
-
-
10102-43-9
nitrogen(II) oxide

-
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| With Pd supported ZrO2-CeO2 catalyst at 175 - 400℃; for 1h; Reagent/catalyst; Temperature; Inert atmosphere; | 100% |
| With H2; O2; catalyst: 0.1 wtpercent Pt/La0.7Sr0.2Ce0.1FeO3 In neat (no solvent, gas phase) Kinetics; NO:H2:O2 = 1:0.25:5% gas mixt. with 5% H2O added in feed stream; at 140°C for 20 h; H2O as reagent; detd. by mass spectrometry, gas chromy.; | 93% |
| With (Y0.90Pr0.10)2O(3+x) at 900℃; Temperature; Inert atmosphere; | 79% |
-
-
7664-41-7
ammonia

-
-
80937-33-3
oxygen

-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) Fe-ZSM-5 catalyst prepared by ion exchange and heat-treated at 400, 425or 450 °C, 100 % NH3 conversion, 100 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 100% B 0% C 0% |
| With catalyst:Fe-mordenite In neat (no solvent) Fe-mordenite catalyst prepared by ion exchange and heat-treated at 425 °C, 92 % NH3 conversion, 99 % N2 selectivity, 1000 ppm NH3 in 2 %O2-contg. He; | A 92% B n/a C 0% |
| With catalyst:Fe-ZSM-5 In neat (no solvent) Fe-ZSM-5 catalyst prepared by ion exchange and heat-treated at 375 °C, 90 % NH3 conversion, 99 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 90% B n/a C 0% |


| Conditions | Yield |
|---|---|
| at room temperature; | A n/a B 100% |
| at room temperature; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| silver (I) ion In water catalytic oxydation in presence of AgCl at pH=7.2;; | 100% |
| silver (I) ion In water catalytic oxydation in presence of AgI at pH=12.7;; | 91% |
| silver (I) ion In water catalytic oxydation in presence of (Ag(S2O3)2)(3-) at pH=12.7;; | 89% |

| Conditions | Yield |
|---|---|
| 5 d; | A 100% B n/a |
| 5 d; | A 100% B n/a |

| Conditions | Yield |
|---|---|
| With HCl In tetrahydrofuran acidolysis (excess of HCl); | A 100% B 96% |
| In sulfuric acid aq. H2SO4; | A 77% B 90% |

| Conditions | Yield |
|---|---|
| In neat (no solvent, gas phase) by heating to 130-150°C at low pressure; at temp. > 450°CS2, N2, S2N2 are produced; at temp. < 130°C S4N2, S4N4, S2N2 areproduced; various product ratio for various conditions; further products; detected by He I photoelectron spectroscopy and quadrupole mass specrometry; | A 100% B <1 |
-
-
21373-88-6, 16920-54-0
(PPh3)3CoH(N2)

-
-
1579-72-2
2,2,2-trifluoroethyl benzoate

-
A
-
99668-73-2
(trifluoroethoxo)tris(triphenylphosphine)cobalt(I)

-
B
-
120-51-4
benzoic acid benzyl ester

-
C
-
7727-37-9
nitrogen

-
D
-
1333-74-0
hydrogen

-
E
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| In toluene PhCOOCH2CF3 added to toluene soln. of CoH(N2)(PPh3)3, evacuated, stirred at 20°C for 2 days; | A n/a B 28% C 100% D 17% E 32% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) loading and unloading carried out in inert gas; heated at 920 K and pressure of 9 bar; elem. anal.; | A 100% B n/a |

| Conditions | Yield |
|---|---|
| In neat (no solvent) High Pressure; loading and unloading carried out in inert gas; heated at 920 K and pressure of 400 bar; elem. anal.; | A 100% B n/a |

| Conditions | Yield |
|---|---|
| In neat (no solvent) loading and unloading carried out in inert gas; heated at 618 K and pressure of 1E-5 bar; elem. anal.; | A 100% B n/a |


-
B
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In tetrahydrofuran -35°C or 25°C; followed by IR and NMR spectroscopy; | A 100% B n/a |


| Conditions | Yield |
|---|---|
| With oxygen In gaseous matrix 450°C; (NH3) = 1000 ppm; (O2) = 2%; He = balance; total flow rate = 500 ml/min; space velocity = 2.3 10**5 1/h; | 99% |
| With oxygen In gaseous matrix 450°C; (NH3) = 1000 ppm; (O2) = 2%; He = balance; total flow rate = 500 ml/min; space velocity = 2.3 10**5 1/h; | 97% |
| With oxygen In gaseous matrix byproducts: N2O, NO; 400°C; (NH3) = 1000 ppm; (O2) = 2%; He = balance; total flow rate = 500 ml/min; space velocity = 2.3 10**5 1/h; | 93% |
-
-
7664-41-7
ammonia

-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) Fe-mordenite catalyst prepared by ion exchange and heat-treated at 450 °C, 99 % NH3 conversion, 100 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 99% B 0% C 0% |
| With oxygen; platinum high excess O2,500 °C; | A <1 B n/a C n/a |
| With oxygen; platinum at 300 °C; | A n/a B <9 C n/a |


| Conditions | Yield |
|---|---|
| Sr0.22La0.78CoO2.843 In neat (no solvent) byproducts: H2O; 800°C; | A n/a B 98.6% |
| Sr0.42La0.68CoO2.80 In neat (no solvent) byproducts: H2O; 800°C; | A n/a B 98.1% |
| Sr0.46La0.54CoO2.79 In neat (no solvent) byproducts: H2O; 800°C; | A n/a B 98% |


| Conditions | Yield |
|---|---|
| With ClC6H4N2(1+) In tetrahydrofuran byproducts: ClC6H5, CH4, C2H6; -78°C; | A 98% B n/a |
| With 2,4,6-trichlorobenzenediazonium PF6 In acetonitrile byproducts: 2,4,6-trichlorobenzene, CH4, C2H6; -78°C; | A 96% B n/a |
| With CH3OC6H4N2(1+) In tetrahydrofuran byproducts: CH3OC6H5, C2H6; -78°C; | A 96% B n/a |

-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10102-44-0
Nitrogen dioxide

-
D
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| at 220-260°C, nearly 98% N2O, 2% N2; troces of NO and NO2 (0.001%); | A 2% B n/a C n/a D 98% |


-
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| With copper(II) oxide In neat (no solvent) heating in a stream of CO2, ratio KCN:CuO=2:10, gegin of react. at 115°C, formation of 98% N2 at 420°C;; | 98% |
| With CuO In neat (no solvent) heating in a stream of CO2, ratio KCN:CuO=2:10, gegin of react. at 115°C, formation of 98% N2 at 420°C;; | 98% |
| heating; exclusion of air; anhydrous KCN; |
-
-
21373-88-6, 16920-54-0
(PPh3)3CoH(N2)

-
-
406-95-1
2,2,2-trifluoroethyl acetate

-
A
-
34557-54-5
methane

-
B
-
99668-73-2
(trifluoroethoxo)tris(triphenylphosphine)cobalt(I)

-
C
-
7727-37-9
nitrogen

-
D
-
1333-74-0
hydrogen

-
E
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| In toluene CH3COOCH2CF3 added to toluene soln. of CoH(N2)(PPh3)3, evacuated, stirred at room temp. for 2 days; hexane added, solid recrystd. from toluene-hexane; elem. anal.; | A 1% B 40% C 98% D 23% E 30% |

-
-
1333-74-0
hydrogen

-
-
10102-43-9
nitrogen(II) oxide

-
-
10102-44-0
Nitrogen dioxide

-
A
-
7727-37-9
nitrogen

-
B
-
7664-41-7
ammonia

| Conditions | Yield |
|---|---|
| With catalyst: industrial nickel methanation catalyst In neat (no solvent) reduction of mixt. of NO/NO2 in gas mixt. of N2/H2 on zeolite catalyst (pretreated in H2 at 550°C, 200°C reaction temp.); gas chromy. (dimethylsulfolane coated diatomite); | A 96% B 0% |
| With catalyst: industrial nickel methanation catalyst In neat (no solvent) reduction of mixt. of NO/NO2 in gas mixt. of N2/H2 on zeolite catalyst (pretreated in H2 at 300°C, 200°C reaction temp.); gas chromy. (dimethylsulfolane coated diatomite); | A 92.5% B 7.5% |
| With catalyst: industrial nickel methanation catalyst In neat (no solvent) reduction of mixt. of NO/NO2 in gas mixt. of N2/H2 on zeolite catalyst (pretreated in H2 at 300°C, 150°C reaction temp.); gas chromy. (dimethylsulfolane coated diatomite); | A 91% B 0% |

-
-
84806-15-5
1,3-di-tert-butyl-2,2-dimethyl-1,3,2,4-diazasilagermaetidine

-
-
622-37-7
Phenyl azide

-
A
-
138174-58-0
1,3,7,9-tetra-tert-butyl-2,2,8,8-tetramethyl-5,10-diphenyl-1,3,5,7,9,10-hexaaza-2,8-disila-4,6-digermadispiro{3.1.3.1}decane

-
B
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under Ar, dropwise addn. of THF soln. of diaminogermylene to THF soln. of phenyl azide at -25.degreeC, 3h; pptn., recrystn. (THF), elem. anal.; | A 64% B 96% |
| In tetrahydrofuran under Ar, dropwise addn. of THF soln. of diaminogermylene to THF soln. of phenyl azide at 25.degreeC, 3h; pptn., recrystn. (THF), elem. anal.; | A 64% B 83% |


-
B
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| With 4-chlorobenzenediazonium hexafluorophosphate byproducts: CH4, ClC6H5; | A 91% B 96% |

| Conditions | Yield |
|---|---|
| With CH3OC6H4N2(1+) In tetrahydrofuran byproducts: CH3OC6H5, C2H6, CH4; -78°C; | A 95% B n/a |
| With 2,4,6-trichlorobenzenediazonium PF6 In tetrahydrofuran byproducts: 2,4,6-trichlorobenzene, CH4, C2H6; -78°C; | A 94% B n/a |
| With 2,4,6-trichlorobenzenediazonium PF6 In acetonitrile byproducts: 2,4,6-trichlorobenzene, CH4, C2H6; -78°C; | A 94% B n/a |

| Conditions | Yield |
|---|---|
| A 95% B n/a |


| Conditions | Yield |
|---|---|
| With oxygen In gaseous matrix byproducts: NO; 0.2 g of catalyst, 1000 ppm NH3, 2% O2, He as balance, GHSV=2.0E+5 h**-1, at 450°C; mass spect.; | A 94% B 0% |
| With oxygen In gaseous matrix byproducts: NO; 0.2 g of catalyst, 1000 ppm NH3, 2% O2, He as balance, GHSV=2.0E+5 h**-1, at 400°C; mass spect.; | A 93% B 0% |
| With oxygen In gaseous matrix byproducts: NO; 0.2 g of catalyst, 1000 ppm NH3, 2% O2, He as balance, GHSV=2.0E+5 h**-1, at 400°C; mass spect.; | A 92% B 0% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) NH3:Cl2 = 39; at ambient temp.;; | A 94% B n/a C n/a |
| In neat (no solvent) NH3:Cl2 = 9; at ambient temp.;; | A 5% B n/a C n/a |
| In neat (no solvent) Cl2:NH3 = 1:10-50; reaction of Cl2 and NH3 dild. with N2 below 180°C under pressure;; | |
| In neat (no solvent) Cl2:NH3:N2 = 1:20:8; at 100°C;; |


-
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) thermolysis (150°C, 3 h); | 94% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Mg (purity: 99.5%) was heated in N2 (free of O2) at 800-850 °C for 4-5 h;; | 100% |
| In neat (no solvent) Mg was heated in a stream of N2 at 900 °C;; | 95% |

| Conditions | Yield |
|---|---|
| 260-560°C, then 0.5h at 600°C; | 100% |
| In neat (no solvent) exothermic formation out of the elements at temperatures under the melting point of Ba; after heating cooling to keeping the necessary temperature;; | |
| above 190°C; 96.15% Ba3N2; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) loading and unloading carried out in inert gas; Ba metal loaded in tungsten crucible, which palced in a steel tube before loaded in quartz glasstube; evacuated (p<1E-6 bar); heated in stream of nitrogen at 1120 K fo r 24 h; evacuated again (p<1E-6 bar) for 24 h at 900 K; elem. anal.; | 100% |
| In further solvent(s) absence of air and moisture; passing N2 through soln. of Ba in liquid Naat 573 K (pptn.); filtration; reaction followed by resistivity measurements; | |
| In further solvent(s) under inert atmosphere; Ba submerged in molten Na in stainless steel crucible; sealed; evacuated; filled with N2; heated at 973 K for 48 h; cooled under vac.; heated at 723 K for 24 h; cooled to room temp.; opened in Ar filled glovebox; detd. by X-ray powder diffraction; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Electrochem. Process; cothodic pulverization within 10 hours of Bi under pure N2 (0.1 - 0.3 Torr), glow-discharge at 1000-1500 V; anode Fe; description of the aparatus given;; 359.8 mg BiN; deposition of 12.1 weight % on cathode, of 45.4 weight % on anode, 25.2 weight % on glass parts of the aparatus, 17.3 weight % on Fe- (or Al-) protecting ring of the aparatus;; | 100% |
| In further solvent(s) Electric Arc; between Bi-cathode and Cu-anode in a liquid mixture of argon and nitrogen;; | |
| In further solvent(s) Electric Arc; between Bi-cathode and Cu-anode in a liquid mixture of argon and nitrogen;; | |
| In neat (no solvent) cathodic pulverization of Bi during glow discharge under N2 (0.1 - 0.3 torr);; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) loading and unloading carried out in inert gas; Sr metal loaded in tungsten crucible, which palced in a steel tube before loaded in quartz glasstube; evacuated (p<1E-6 bar); heated in stream of nitrogen at 1120 K fo r 24 h; evacuated again (p<1E-6 bar); for 24 h at 900 K; elem. anal.; | 100% |
| In gas other Radiation; strontium deposited on MgO substrate at temp. 673 K under irradn. of nitrogen radicals (RF power 350 W, N2 feed 3.0 cm**+3(STP)min**-1); annealed in vac. at 673 K or continuous nitrogen radical irradn. for 30 min; cooled; | |
| In neat (no solvent) Fresh Sr filings are loaded into a Al2O3 jacket which is itself placed in a quartz tube. The assembly is evacuated to E-5 Torr. N2 is introduced into the vacuum line to a total pressure of 100 Torr. The temp. is slowly increased to 750°C.; |

| Conditions | Yield |
|---|---|
| With catalyst: Pt-oxide at 1000°C; | 100% |
| platinum at 1000°C; | 100% |
| platinum at 800°C; | 62.8% |
| With catalyst: Pt-oxide at 800°C; | 62.8% |


-
-
7727-37-9
nitrogen

-
-
163929-87-1
[Mo(N(C((2)H3C)2CH3)((CH3)2C6H3))3N]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene stirring (25°C, N2 atmosphere, 24 h); evapn. (vac.); | 100% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) High Pressure; loading and unloading carried out in inet gas; SrN loaded in a Mo boat, which placed in an autoclave; evacuated; filled up with N2; heated at 920 K for 24 h at 5500 bar; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| With sodium amalgam In tetrahydrofuran stirring (28°C, 1 atm. N2); | 100% |
| With 1-methyl-1H-imidazole In diethyl ether 25°C, ether, 1 atm. of N2, 1 equiv of 1-methylimidazole, 6 h; evapd., extd. with pentane, filtered, evapd., recrystd. from pentane at -35°C; | 91% |
| With dmap In diethyl ether 25°C, ether, 1 atm. of N2, 4-dimethylaminopyridine, 24 h; evapd., extd. with pentane, filtered, evapd., recrystd. from pentane at -35°C; | 85% |

-
-
7727-37-9
nitrogen

-
B
-
163929-87-1
[Mo(N(C((2)H3C)2CH3)((CH3)2C6H3))3N]

| Conditions | Yield |
|---|---|
| With sodium amalgam In tetrahydrofuran addn. of soln. of Mo-complex to stirred suspn. of amalgam (syringe pump,10 h); decantation, filtration (Celite), evapn. (vac.), dissoln. (pentane), filtration (Celite), evapn. (vac.); | A 100% B 1% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) High Pressure; loading and unloading carried out in inet gas; Sr4N3 loaded in a Mo boat, which placed in an autoclave; evacuated; filled up with N2; heated at 920 K for 24 h at 400 bar; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) High Pressure; loading and unloading carried out in inet gas; Sr2N loaded in a Mo boat,which placed in an autoclave; evacuated; filled up with N2; heated at 9 20 K for 24 h at 5500 bar; elem. anal.; | 100% |
| High Pressure; under N2 pressure of 5500 bar; heated at 920 K in Mo-boat for 72 h; initial pressure about 2720 bar; X-ray and neutron diffraction; TG/DTA; elem. anal.; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Li3N fused in pure iron vessel; sealed under 300 kPa of N2; heated at 850-1050°C for 12 h; thermally quenched; detd. by X-ray powder diffraction; | 100% |

-
-
262359-17-1
(RuH(μ-Cl)(triisopropylphosphine)2

-
-
7727-37-9
nitrogen

-
-
262359-18-2
RuHCl(triisopropylphosphine)2(N2)

| Conditions | Yield |
|---|---|
| In benzene-d6 (Ar); soln. of Ru-complex in benzene-d6 added to NMR tube, sample degassed, benzene frozen in ice, tube filled with N2 to ca. 1 atm., tube agitated for 5 min; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) alloy powder transferred into boron nitride crucible, loaded into hot isostatic pressing with graphite heater, N2 pressure raised to 190 MPa at room temp., heated to 2173 K, held for 2 h, temp. reduced to ca. 473 K, pressure to 70 MPa; cooled to room temp.; detd. by XRD; | 100% |

-
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In neat (no solvent) alloy powder transferred into boron nitride crucible, loaded into hot isostatic pressing with graphite heater, N2 pressure raised to 50 MPa at room temp., heated to 2173 K, held for 2 h, temp. reduced to ca. 473 K, pressure to 70 MPa; cooled to room temp.; detd. by XRD; | 100% |

-
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In neat (no solvent) alloy powder transferred into boron nitride crucible, loaded into hot isostatic pressing with graphite heater, N2 pressure raised to 50 MPa at room temp., heated to 2173 K, held for 2 h, temp. reduced to ca. 473 K, pressure to 70 MPa; cooled to room temp.; detd. by XRD; | 100% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View