 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route
-
-
7446-09-5
sulfur dioxide

-
-
7732-18-5
water

-
-
7553-56-2
iodine

-
A
-
7664-93-9
sulfuric acid

-
B
-
10034-85-2
hydrogen iodide

| Conditions | Yield |
|---|---|
| 0 - 25 °C; part of a Mg-S-I water splitting cycle; | A 100% B 100% |


| Conditions | Yield |
|---|---|
| at 170-180°C; in very dilute soln. complete decompn. in 2 h, incomplete decompn. in concd. solns.; | A n/a B 100% |
| byproducts: H2S4O6; | |
| sodium thiosulfate In water 100°C; |

-
-
463-58-1
carbon oxide sulfide

-
-
7722-84-1
dihydrogen peroxide

-
A
-
124-38-9
carbon dioxide

-
B
-
7664-93-9
sulfuric acid

-
C
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With potassium sulfate; potassium hydrogensulfate; potassium peroxomonosulfate In water Kinetics; oxidation of OCS studied in round-bottom Pyrex bulbs, acid-water mixtures introduced into bulbs and degassed, bulb reactors filled with with a gas mixture slightly above 1 atm total pressure with a typical mixing ratio of OCS:Ar:He=40:60:700 Torr; gas chromy. and mass spectroscopy applied for determination of product content; | A 100% B n/a C n/a |
| With sulfuric acid In water Kinetics; oxidation of OCS studied in round-bottom Pyrex bulbs, acid-water mixtures introduced into bulbs and degassed, bulb reactors filled with with a gas mixture slightly above 1 atm total pressure with a typical mixing ratio of OCS:Ar:He=40:60:700 Torr; gas chromy. and mass spectroscopy applied for determination of product content; | A 100% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With sulfuric acid; ammonia absorption of dild. SO2 in aq. soln. of NH3, eliberation with H2SO4, O2 from water electrolysis, 3-layer contact bed, heat exchanger, 3 H2SO4 absorbers, coke filter, tail gas recirculation, 528-682°C; | 99.5% |
| With NH3; H2SO4; catalyst: V compd. absorption of dild. SO2 in aq. soln. of NH3, eliberation with H2SO4, O2 from water electrolysis, 3-layer contact bed, heat exchanger, 3 H2SO4 absorbers, coke filter, tail gas recirculation, 528-682°C; | 99.5% |
| ferric hydroxide In not given 125-130°C, Fe(OH)3 on pumice or asbestos; |

| Conditions | Yield |
|---|---|
| With air moist air introductions into four-stage contact furnace, 2-stage drum absorber, washing of tail gas in Venturi app.; 93-95 % acid concn.; | 99% |
| With air moist air introductions into four-stage contact furnace, 2-stage drum absorber, washing of tail gas in Venturi app.; 93-95 % acid concn.; | 99% |
| With catalyst: V compd. moist S roaster gas, absorption in concd. acid; |
-
-
7647-01-0
hydrogenchloride

-
-
7758-99-8
copper(II) sulfate

-
A
-
7664-93-9
sulfuric acid

-
B
-
7782-50-5
chlorine

-
C
-
12775-96-1, 15158-11-9, 15721-63-8, 16941-75-6, 17493-86-6, 19498-52-3, 20499-83-6, 20499-84-7, 20499-85-8, 20499-86-9, 20573-10-8, 20573-11-9, 21595-51-7, 21595-52-8, 22206-52-6, 26445-28-3, 28959-95-7, 37362-93-9, 39417-05-5, 54603-16-6, 54603-23-5, 54603-32-6, 54603-40-6, 54603-48-4, 54603-81-5, 54603-89-3, 56316-56-4, 95985-91-4, 122297-32-9, 7440-50-8
copper

| Conditions | Yield |
|---|---|
| In water Electrolysis; | A n/a B 90% C 99% |
| In water |


| Conditions | Yield |
|---|---|
| In hydrogenchloride room temp.; 10-15 min.; | A n/a B 99% |
| In hydrogenchloride room temp.; 10-15 min.; | A n/a B 99% |


| Conditions | Yield |
|---|---|
| With sulphurous acid byproducts: H2O; | A n/a B 99% |
| With H2SO3 byproducts: H2O; | A n/a B 99% |

| Conditions | Yield |
|---|---|
| In not given Electrolysis; Pt anode, graphite cathode, area of the electrodes 30 cm^2, 1 A, 20 min, 0.208 mg/l SO2 soln.; | A 98.16% B 70.87% |
| In not given Electrolysis; Pt anode, graphite cathode, area of the electrodes 30 cm^2, 1 A, 20 min, 0.420 mg/l SO2 soln.; | A 98.52% B 74.28% |
| In not given Electrolysis; Pt anode, graphite cathode, area of the electrodes 30 cm^2, 1 A, 20 min, 1.123 mg/l SO2 soln.; | A 98.86% B 74.2% |

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| 3-stage contact with heat exchangers, for pyrite roaster gas; | 98% |
| With air burning at 820°C in rotating furnace, elec. filter, washing, air addn., wet elec. filter, drying, heat exchanger, two-stage inner cooling, cooler, absorption, cooling; | |
| With nitric acid formation of H2SO4 by reaction of HNO3 with pyrite; faster reaction at 100 °C than at higher temperature;; | |
| With air; catalyst: V2O5 - K2O - SiO2 burning at 820°C in rotating furnace, elec. filter, washing, air addn., wet elec. filter, drying, heat exchanger, two-stage inner cooling, cooler, absorption, cooling; |

| Conditions | Yield |
|---|---|
| With (CN)2 In water byproducts: HCN; with (CN)2 excess for 4-5 days at room temp.; | 94% |
| contact oven with intermediate heat exchangers, temp. regulation by fresh air addn., H2O to inlet gas, SO2 recycling; | 80% |
| condensation after contact furnace at 175. degree.C; 66.3 % acid concn.; | 46.7% |

| Conditions | Yield |
|---|---|
| In water Electrolysis; formation of PbSO4 on Pb-anode; electrolysis of PbSO4; | A 91% B 91% |
| With H2O Electrolysis; | |
| In water Electrolysis; |
-
-
7782-99-2
sulphurous acid

-
-
7790-93-4
chloric acid

-
A
-
7664-93-9
sulfuric acid

-
B
-
10049-04-4, 25052-55-5
chlorine dioxide

-
C
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With air; sulfur dioxide In sulfuric acid byproducts: HClO, HCl, Cl2; Holst-process;; | A n/a B 90% C n/a |


| Conditions | Yield |
|---|---|
| uranyl chloride In neat (no solvent) equilibrium on thermal decompn.;; distillation;; | A 90% B n/a |
| mercury In neat (no solvent) equilibrium on thermal decompn.;; distillation;; | A 90% B n/a |
| bismuth(III) chloride In neat (no solvent) equilibrium on thermal decompn.;; distillation;; | A 90% B n/a |

-
-
4199-89-7
5-Chloro-1,10-phenanthroline

-
-
4370-59-6
bromanilic acid

-
-
230953-51-2
(VO)2(Br2C6O4)(C12H7ClN2)2(2+)*SO4(2-)=[(VO)2(Br2C6O4)(C12H7ClN2)2](SO4)

-
B
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In methanol dropwise addn. of soln. of VOSO4 to soln of bromanilic acid, addn. of soln. of N-ligand (stirring, room temp.), refluxing (8 h; crystn.); filtration, washing (MeOH, Et2O), drying (vac., over P2O5), recrystn. (DMF/EtOH); elem. anal.; | A 88% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: chlorosulfonic acid With Acetanilid at 26 - 45℃; for 9.2h; Stage #2: With thionyl chloride at 55℃; for 6.4h; Temperature; Further stages; | A 87% B n/a |

-
-
484-11-7
2.9-dimethyl-1,10-phenanthroline

-
-
4370-59-6
bromanilic acid

-
-
230953-49-8
(VO)2(Br2C6O4)((CH3)2C12H6N2)2(2+)*SO4(2-)=[(VO)2(Br2C6O4)((CH3)2C12H6N2)2](SO4)

-
B
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In methanol dropwise addn. of soln. of VOSO4 to soln of bromanilic acid, addn. of soln. of N-ligand (stirring, room temp.), refluxing (8 h; crystn.); filtration, washing (MeOH, Et2O), drying (vac., over P2O5), recrystn. (DMF/EtOH); elem. anal.; | A 85% B n/a |

-
-
66-71-7
1,10-Phenanthroline

-
-
4370-59-6
bromanilic acid

-
-
230953-47-6
(VO)2(Br2C6O4)(C12H8N2)2(2+)*SO4(2-)=[(VO)2(Br2C6O4)(C12H8N2)2](SO4)

-
B
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In methanol dropwise addn. of soln. of VOSO4 to soln of bromanilic acid, addn. of soln. of N-ligand (stirring, room temp.), refluxing (8 h; crystn.); filtration, washing (MeOH, Et2O), drying (vac., over P2O5), recrystn. (DMF/EtOH); elem. anal.; | A 81% B n/a |

-
-
5329-14-6
aminosulfonic acid

-
A
-
7664-93-9
sulfuric acid

-
B
-
7727-37-9
nitrogen

-
C
-
7647-14-5
sodium chloride

| Conditions | Yield |
|---|---|
| With chlorine In water introduction of chlorine into alkaline soln.; intermediates (NaSO3NCl2/NaSO3NHCl) decompose after short time in this soln.;; | A n/a B 79.65% C n/a |
| With Cl2 In water introduction of chlorine into alkaline soln.; intermediates (NaSO3NCl2/NaSO3NHCl) decompose after short time in this soln.;; | A n/a B 79.65% C n/a |

-
-
366-18-7
[2,2]bipyridinyl

-
-
4370-59-6
bromanilic acid

-
-
230953-45-4
(VO)2(Br2C6O4)(NC5H4C5H4N)2(2+)*SO4(2-)=[(VO)2(Br2C6O4)(NC5H4C5H4N)2](SO4)

-
B
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In methanol dropwise addn. of soln. of VOSO4 to soln of bromanilic acid, addn. of soln. of N-ligand (stirring, room temp.), refluxing (8 h; crystn.); filtration, washing (MeOH, Et2O), drying (vac., over P2O5), recrystn. (DMF/EtOH); elem. anal.; | A 72% B n/a |

-
-
13954-94-4
trisulfimide

-
A
-
7803-63-6
ammonium bisulfate

-
B
-
7664-93-9
sulfuric acid

-
C
-
7803-58-9
SULFAMIDE

| Conditions | Yield |
|---|---|
| With water heat of evapn., 2 h, 2 M HCl; | A n/a B 67% C n/a |
| With H2O heat of evapn., 2 h, 2 M HCl; | A n/a B 67% C n/a |

-
-
13954-94-4
trisulfimide

-
A
-
7664-93-9
sulfuric acid

-
B
-
5329-14-6
aminosulfonic acid

-
C
-
7803-58-9
SULFAMIDE

| Conditions | Yield |
|---|---|
| With water in the cold, 48 h, 2 M HCl; | A 33% B n/a C n/a |
| With H2O in the cold, 48 h, 2 M HCl; | A 33% B n/a C n/a |
| In water decompn. in aq. soln.; 0 and 22°C discussed; presece of acid discussed;; |


| Conditions | Yield |
|---|---|
| In water 0.2362 mM/g decompn. 46.8°C, 245 h.; | A 19.96% B n/a |
| In water 0.2401 mM/g decompn. 25.3°C, 945.25 h.; | A 2.96% B n/a |
| In water Kinetics; |

| Conditions | Yield |
|---|---|
| condensation of humidified S burner gas; | |
| absorption of moist gas to H2SO4; | |
| In water in sealed tube;at 150°C;under 20 atm O2;; | 0.06% |
-
-
7440-69-9
bismuth

-
-
7664-93-9
sulfuric acid

-
-
7440-44-0
pyrographite

-
-
68283-19-2
2-butyl-5-hydroxymethyl-1H-imidazole

-
-
7440-06-4
platinum

-
-
68282-49-5
2-butyl-1H-imidazole-5-carboxaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water | 100% |
| With sodium hydroxide In water | 98.2% |
| With sodium hydroxide In methanol; water | 94.5% |

-
-
170142-44-6
7-Chloro-4-hydroxy-2-(4-carboxyphenyl)-1,2,5,10-tetrahydropyridazino [4,5-b]quinoline-1,10-dione

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In methanol | 100% |

| Conditions | Yield |
|---|---|
| In hydrogenchloride; sulfuric acid dissolving CdS in HCl, evapn. to dryness, dissoln. in dild. H2SO4, evapn. with a drop of concd. HNO3; | 100% |
| In hydrogenchloride; sulfuric acid aq. H2SO4; dissolving CdS in HCl, evapn. to dryness, dissoln. in dild. H2SO4, evapn. with a drop of concd. HNO3; | 100% |

| Conditions | Yield |
|---|---|
| 70 °C; 1-2. step of Mg-S-I water splitting cycle; | A 100% B 100% |

-
-
392334-61-1, 371241-08-6, 392333-87-8, 1226500-22-6
[Fe(μ-S2(CH2)3)(CN)(CO)4(PMe3)](1-)

-
-
7664-93-9
sulfuric acid

-
-
1333-74-0
hydrogen

| Conditions | Yield |
|---|---|
| In water Electrolysis; electrolysis of soln. of Fe2(CO)4(CN)(PMe3)S2(CH2)3 with 50 equiv. H2SO4at -1.2 V for 15 min; GC analysis; | 100% |

| Conditions | Yield |
|---|---|
| With water In sulfuric acid digesting; concn. of H2SO4 in aq. H2SO4 >93%; pptn. by adjusting H2SO4 concn. to 84.15%-93% with water; | 100% |
| With H2O In sulfuric acid aq. H2SO4; digesting; concn. of H2SO4 in aq. H2SO4 >93%; pptn. by adjusting H2SO4 concn. to 84.15%-93% with water; | 100% |
| With water In sulfuric acid uptake of water from the air; |

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In sulfuric acid aq. H2SO4; Nb-contg. compd. was treated in aq. H2SO4 for 48 h; | 100% |

-
-
7664-93-9
sulfuric acid

-
-
827320-59-2
1-methyl-3-(4-sulfobutyl)-1H-imidazol-3-ium hydrogensulfate

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 24h; | 100% |
-
-
21876-43-7
4-(pyridinium-1-yl)butane-1-sulfonate

-
-
7664-93-9
sulfuric acid

-
-
827320-61-6
N-(4-sulphonic acid)butylpyridinium hydrogen sulphate

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| at 300℃; for 3h; | 100% |

-
-
1633-05-2
strontium(II) carbonate

-
-
7664-93-9
sulfuric acid

-
-
7446-11-9
sulfur trioxide

-
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| at 180℃; for 24h; | 100% |


| Conditions | Yield |
|---|---|
| In further solvent(s) H2SO4 was added to Bi in HNO3 with stirring and heating; more H2SO4 wasadded and suspn. was heated for 40 h; heated in a muffle furnace at 648K; elem. anal.; | 99.8% |
| In sulfuric acid byproducts: SO2; by dissolving in boiling concd. H2SO4;; | |
| In sulfuric acid byproducts: SO2; very weak reaction with diluted H2SO4.;; | |
| In nitric acid Bi dissolved in HNO3 and treated with H2SO4, heated for several hours at 380 °C;; |
-
-
24766-14-1
2-(bromomethyl)-3-methylbutanoic acid

-
-
7664-93-9
sulfuric acid

-
-
115-11-7
isobutene

-
-
224178-55-6
2-Bromomethyl-3-methyl-butyric acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In dichloromethane | 99% |

-
-
108-75-8
2,4,6-trimethyl-pyridine

-
-
100-60-7
N-methylcyclohexylamine

-
-
7664-93-9
sulfuric acid

-
-
1643-19-2
tetrabutylammomium bromide

| Conditions | Yield |
|---|---|
| In 1,1,2,2-tetrachloroethylene | 99% |

-
-
27697-50-3
N-nitroso-N-cyclohexylhydroxylamine potassium salt

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In water; toluene | 99% |

| Conditions | Yield |
|---|---|
| In methanol byproducts: HCl; concd. H2SO4 is added to a soln. of the cluster in MeOH, HCl is evolvedand soln. is warmed up; soln. is heated to remove HCl and concd., soln. is diluted with MeOH, ppt.is sepd., washed and dried over P2O5, elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| With acetic acid In water soln. PdEnCl2 in water was treated under heating in water bath with saturated aq. thiourea acidified with acetic acid, in 30 min 2 M H2SO4 was added; ppt. was filtered, washed with diluted H2SO4 and water, and dried; | 99% |


-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In sulfuric acid aq. H2SO4; room temp.; | 99% |

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In sulfuric acid aq. H2SO4; room temp.; | 99% |

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In sulfuric acid aq. H2SO4; room temp.; | 99% |

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In sulfuric acid aq. H2SO4; room temp.; | 99% |

-
-
7664-93-9
sulfuric acid

| Conditions | Yield |
|---|---|
| In sulfuric acid aq. H2SO4; room temp.; | 99% |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View












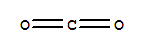



 C
C