 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| In water Electrolysis; anodic oxidation of 3.6 n chloride soln.;; |

-
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| In water Electrolysis; | |
| Electrolysis; acidic electrolyte; | |
| Electrolysis; alk. electrolyte, max. current efficiency 60-65 %; |

| Conditions | Yield |
|---|---|
| In water equilibrium reaction; examination at different temperatures;; | |
| In water equilibrium reaction; examination at different temperatures;; | |
| In water equilibrium reaction; examination at different temperatures;; | |
| In water | |
| In water concd. CaCl2 soln. as by-product; stepwise concg.;; |

-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
-
584-08-7
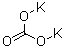
potassium carbonate

-
A
-
3811-04-9
potassium chlorate

-
B
-
14314-27-3
potassium chlorite

| Conditions | Yield |
|---|---|
| In water byproducts: H2O; | |
| In water byproducts: H2O; |


| Conditions | Yield |
|---|---|
| Electrolysis; electrolysis of NaCl, addn. of K2CO3 to obtained NaClO3; | |
| Electrolysis; electrolysis of NaCl, addn. of K2CO3 to obtained NaClO3; |

| Conditions | Yield |
|---|---|
| With oxygen Addn. of O2 to introduced Cl2 does not increase yield.; |


| Conditions | Yield |
|---|---|
| chlorination and treating obtd. soln. with KCl; | |
| chlorination and treating obtd. soln. with KCl; |
-
-
13444-99-0, 501680-61-1
perchloric acid monohydrate

-
A
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| With 0.1n-KOH In neat (no solvent) byproducts: chlorine oxides, Cl2, O2; neutralisation of just melting HClO4*H2O with 0.1n KOH; yield of KClO3 much lower than expected from hydrolysis of Cl2O6; The products do not contain HCl;; |

| Conditions | Yield |
|---|---|
| With potassium permanganate; water In water byproducts: MnO2; | |
| With KMnO4; H2O In water byproducts: MnO2; |
-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
A
-
3811-04-9
potassium chlorate

-
B
-
14314-27-3
potassium chlorite

| Conditions | Yield |
|---|---|
| With potassium hydroxide In not given byproducts: KClO3, H2O; | |
| With potassium hydroxide In water addn. of a KOH soln. to a ClO2 soln. containing CO2 and protected from light;; multiple evaporation in the vac. at 45 - 50 °C until KClO3 pptd.; pptn. of K2CO3 with alc.; evaporation in the cold and in the vac.; adding alc. (95 %) to the residue; filtration; evaporation of the filtrate in the cold and in the vac.;; | |
| With potassium hydroxide In water addn. of a KOH soln. to a ClO2 soln. containing CO2 and protected from light;; multiple evaporation in the vac. at 45 - 50 °C until KClO3 pptd.; pptn. of K2CO3 with alc.; fractionated crystn. in the vac.;; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In not given byproducts: H2O; ClO2 contents max. 2% Cl2; crystn. at -8-5°C, grad of purification: 80%; | |
| With potassium hydroxide In not given byproducts: H2O; ClO2 contents max. 2% Cl2; crystn. at -8-5°C, grad of purification: 80%; |

-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
A
-
3811-04-9
potassium chlorate

-
B
-
14314-27-3
potassium chlorite

| Conditions | Yield |
|---|---|
| In water byproducts: H2O; dild. soln.;; | |
| In water byproducts: H2O; | |
| In water |

-
-
7722-64-7
potassium permanganate

-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
-
3811-04-9
potassium chlorate
-
-
14998-27-7
chlorite

-
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| With potassium hydroxide; manganese(IV) oxide In not given | |
| With KOH; manganese(IV) oxide In not given |

| Conditions | Yield |
|---|---|
| Electrolysis; | |
| Electrolysis; |

| Conditions | Yield |
|---|---|
| Electrolysis; perchlorate formation decreases at higher temps.; | |
| Electrolysis; perchlorate formation decreases at higher temps.; |

| Conditions | Yield |
|---|---|
| Electrolysis; effect of cathode substance; | |
| Electrolysis; effect of cathode substance; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride byproducts: Cl2; Electrolysis; |

| Conditions | Yield |
|---|---|
| Electrolysis; electrolysis of NaCl, addn. of KCl to obtained NaClO3; | |
| Electrolysis; electrolysis of NaCl, addn. of KCl to obtained NaClO3; |

| Conditions | Yield |
|---|---|
| at 95-98 °C; | |
| at 95-98 °C; |


| Conditions | Yield |
|---|---|
| Electrolysis; electrolysis of CaCl2 and addn. of KCl to obtained Ca(ClO3)2; | |
| Electrolysis; electrolysis of CaCl2 and addn. of KCl to obtained Ca(ClO3)2; |



| Conditions | Yield |
|---|---|
| addn. of Ca(OH)2 to soln. remained after crystn. of bulk of KClO3; Calciumoxychloride can be pptd. by cooling soln., causing negligible chlorate loss.; | |
| addn. of Ca(OH)2 to soln. remained after crystn. of bulk of KClO3; Calciumoxychloride can be pptd. by cooling soln., causing negligible chlorate loss.; |

-
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| In water byproducts: O2; Irradiation (UV/VIS); U.V. light; | 100% |
| In water byproducts: O2; Irradiation (UV/VIS); U.V. light; | 100% |
| In neat (no solvent) Kinetics; byproducts: O2, NO2, ((C4H9)2NCO)2(CH2)6; sample heating at 5 K/min from up to 620°C; thermosonimetry and TG, high temp. microscopy; |
-
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| In water hot soln. of KClO3 (0.0018 mol) added to hot soln. of Co(NH3)6Cl3 (0.0018 mol); cooled (4 d); crystals filtered off; dried in air; elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In water hot soln. of KClO3 (0.0012 mol) added to hot soln. of Co(NH3)6Br3 (0.0012 mol); cooled (3 d); crystals filtered off; dried in air; elem. anal.; | 99% |
-
-
3811-04-9
potassium chlorate

-
-
14380-61-1
hypochlorite

| Conditions | Yield |
|---|---|
| With H In not given redn. of a boiling KClO3-soln. (1g in 100ml) by a foil of Pd, laden with hydrogen with addn. of 10ml 25% H2SO4;; | 94% |
| With H In not given redn. of a boiling KClO3-soln. (1g in 100ml) by a foil of Pd, laden with hydrogen with addn. of 10ml 25% H2SO4;; | 94% |
| With H redn. by hydrogen (Pd satd. with H);; |
-
-
3811-04-9
potassium chlorate

-
-
19638-17-6, 16894-68-1
tetraphenylantimony(V) chloride

-
-
16894-73-8
tetraphenylantimony chlorate

| Conditions | Yield |
|---|---|
| In water mixt. of Ph4SbCl and KClO3 in H2O was kept for 24 h at 12°C; solvent removed; recrystd. (acetone); elem. anal.; | 89% |

| Conditions | Yield |
|---|---|
| In water anion exchange in aq. soln.; molar ratio (C5H5Ni(P(C4H9-n)3)2)(1+)Cl(1-):KClO3 = 1:3; between O°C and room temp.; product dissolved in benzene; recrystn. from hexane or benzene/hexane in the presence of P(C4H9-n)3; | 87% |
| In water |

| Conditions | Yield |
|---|---|
| In neat (no solvent) refluxing at 170 to 190°C for 3 h;; distillation;; | 80% |
-
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| In water byproducts: KNO3; Co complex dissolved in H2O; KClO3 dissolved in H2O at room temp.; two solns. mixed; crystd. for 0.5 h; crystals collected by drawing off the mother liquor; air dried; elem. anal.; | 80% |
-
-
3811-04-9
potassium chlorate

-
-
13408-72-5, 14040-32-5, 14040-33-6, 20594-10-9, 20594-11-0
trans-dichlorobis(ethylenediamine)cobalt(III) chloride

-
-
26388-78-3
trans-dichlorobis(ethylenediamine)cobalt(III) chlorate

| Conditions | Yield |
|---|---|
| In water mixing aqueous solns. of cobalt compd. and chlorate salt for 0.5 h; drawing off mother liquor, air drying, elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| In water byproducts: KCl; a soln. of Co-complex and KClO3 in water were mixed, allowed to stand for 1 d at room temp.; crystals were collected and dried in air; elem. anal.; | 80% |
-
-
3811-04-9
potassium chlorate

-
-
4426-72-6, 51433-48-8
hydrazine carboxamide

-
A
-
124-38-9
carbon dioxide

-
B
-
7727-37-9
nitrogen

-
C
-
7664-41-7
ammonia

| Conditions | Yield |
|---|---|
| With hydrogenchloride | A n/a B 66% C n/a |

-
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: O2, KCl; heating;; | 66% |
| In neat (no solvent) byproducts: O2, KCl; heating;; | 66% |
| silica gel In neat (no solvent) various m,ixing proportions were investigated; 15 % SiO2 results the greatest yield of KClO4;; | 59% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: O2; 1.5 mol KI/ mol KClO3; start at 350-400°C; violent reaction under formation of O2; mechanism discussed;; product mixture;; | A 65% B n/a |


| Conditions | Yield |
|---|---|
| With potassium iodide molar ratio of KI:KIO3 = 1.5:1; | 65% |
| With KI molar ratio of KI:KIO3 = 1.5:1; | 65% |
| With iodine In water heating an aq. suspension of I2 with KClO3 and some drops concd. HNO3; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) below -20°C;; passing ClO3F over KI; cooling at -183°C; distillation at -183 - 130°C in vacuum;; | 60% |
| In neat (no solvent) below -20°C;; passing ClO3F over KI; cooling at -183°C; distillation at -183 - 130°C in vacuum;; | 60% |
-
-
3811-04-9
potassium chlorate

-
-
10034-93-2
hydrazonium sulfate

-
A
-
916329-38-9
hydrogen azide

-
B
-
7664-41-7
ammonia

| Conditions | Yield |
|---|---|
| In not given yield is higher in presence of solid Ag2SO4; | A 22% B 48% |
| In not given yield is higher in presence of solid Ag2SO4; | A 22% B 48% |

-
-
3811-04-9
potassium chlorate

-
-
7782-41-4
fluorine

-
A
-
7616-94-6
perchloryl fluoride

-
C
-
10049-04-4, 25052-55-5
chlorine dioxide

-
D
-
7790-89-8
chlorine monofluoride

-
E
-
7782-50-5
chlorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: O2; transport reaction: passing F2-N2 mixture over KClO3 at 25 - 60°C;; washing with NaOH; passing ClO3F through Na2S2O3 soln.; drying over NaOH and P2O5;; | A 45% B n/a C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| In not given multiple evapn. with ambient concd. HNO3; nearly no KClO4 is obtained using fuming HNO3;; | 30% |
| In not given multiple evapn. using ambient concd. or water-free HNO3; nearly no KClO4 is formed, if fuming HNO3 is used;; | 30% |
| In not given multiple evapn. with ambient concd. HNO3; nearly no KClO4 is obtained using fuming HNO3;; | 30% |

| Conditions | Yield |
|---|---|
| With alkali | 25% |
| With alkali | 25% |

| Conditions | Yield |
|---|---|
| In not given boiling until yellow color disappears;; | 15% |
| In not given boiling until yellow color disappears;; | 15% |

| Conditions | Yield |
|---|---|
| In not given | 11% |
| In not given | 11% |
| In not given |
-
-
3811-04-9
potassium chlorate

| Conditions | Yield |
|---|---|
| In sulfuric acid addition of a solution of KClO3 in H2O (0.70 M ClO3(1-)) to a solution of the decaborate (0.20 M B10H10(2-)) in aq. H2SO4 (0.5 M H(1+)) and stirring at 25°C, 7 days;; filtration of ((C2H5)3NH)(B20H18);; | 8% |
| In hydrogenchloride addition of a solution of KClO3 in H2O (0.70 M ClO3(1-)) to a solution of the decaborate (0.20 M B10H10(2-)) in aq. HCl (0.5 M H(1+)) and stirring at 25°C, 7 days;; filtration of ((C2H5)3NH)(B20H18);; | 8% |
| In sulfuric acid addition of a solution of KClO3 in H2O (0.50 M ClO3(1-)) to a solution of the decaborate (0.20 M B10H10(2-)) in aq. H2SO4 (0.5 M H(1+)) and stirring at 25°C, 7 days;; filtration of ((C2H5)3NH)(B20H18);; | 5% |

| Conditions | Yield |
|---|---|
| decompn. of Nh4Cl with heating KClO3; | >99 |
| decompn. of Nh4Cl with heating KClO3; | |
| decompn. of Nh4Cl with heating KClO3; | >99 |
| decompn. of Nh4Cl with heating KClO3; |

| Conditions | Yield |
|---|---|
| by ignition of the mixture; |


| Conditions | Yield |
|---|---|
| With methyllithium 550 kg Cr2O3, 225 kg Al powder, 50-55 kg KClO3 and 50-55 kg lime put into Fe covering cylinder; | 80-85 |


| Conditions | Yield |
|---|---|
| In melt byproducts: Cl2; melting of educts;; | |
| In melt byproducts: Cl2; melting of educts;; |

| Conditions | Yield |
|---|---|
| In water heating of Cr2O3 in KClO3 soln.;; |

| Conditions | Yield |
|---|---|
| With water In not given in weakly acidic 0.1 n KCl soln., at 60-70°C; | |
| With H2O In not given in weakly acidic 0.1 n KCl soln., at 60-70°C; |


| Conditions | Yield |
|---|---|
| A n/a B 0% | |
| A n/a B 0% |


| Conditions | Yield |
|---|---|
| byproducts: KCl; gentle glowing; leaching off KCl from the cooled melt (water); hydrated BaO2; |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View













 O,
O, Xn,
Xn, N
N