-
Name
2,2-Dimethyl-1,3-propanediamine
- EINECS 230-819-0
- CAS No. 7328-91-8
- Article Data19
- CAS DataBase
- Density 0.862 g/cm3
- Solubility
- Melting Point 29-31 °C(lit.)
- Formula C5H14N2
- Boiling Point 150.4 °C at 760 mmHg
- Molecular Weight 102.18
- Flash Point 47.2 °C
- Transport Information UN 2921 8/PG 2
- Appearance clear to yellow liquid or semi-solid
- Safety 16-26-27-36/37/39-45
- Risk Codes 10-34
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms (3-Amino-2,2-dimethylpropyl)amine;1,2-Diamino-2,2-dimethylpropane;1,3-Diamino-2,2-dimethylpropane;2,2-Dimethyltrimethylenediamine;NSC 17719;
- PSA 52.04000
- LogP 1.33060
Synthetic route
-
-
41882-44-4
2,2-dimethylmalonamide

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex In tetrahydrofuran for 2h; Heating; | 75% |
-
-
67744-70-1
3-amino-2,2-dimethyl-propionitrile

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With methanol; nickel Hydrogenation; |

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With potassium hydroxide; water |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water; iron at 100℃; durch Reduktion; | |
| With methanol; nickel under 30891.3 Torr; Hydrogenation; | |
| With methanol; nickel at 50 - 75℃; under 51485.6 Torr; Hydrogenation; | |
| With hydrogen; nickel |
-
-
762-98-1
2,2-dimethyl-1,3-dinitropropane

-
A
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
B
-
41882-44-4
2,2-dimethylmalonamide

| Conditions | Yield |
|---|---|
| With ethanol; nickel at 60℃; under 51485.6 Torr; Hydrogenation; | |
| With ethanol; nickel at 60℃; under 51485.6 Torr; Hydrogenation; |
-
-
31044-89-0
1,3-diazido-2,2-dimethyl-propane

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran for 18h; Heating; |
-
-
126-30-7
2,2-Dimethyl-1,3-propanediol

-
A
-
26734-09-8
3-amino-2,2-dimethylpropan-1-ol

-
B
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; Co-Fe catalyst at 210℃; under 37503 - 112509 Torr; Product distribution; |
-
-
7647-01-0
hydrogenchloride

-
-
859065-87-5
2-(5,5-dimethyl-hexahydro-pyrimidin-2-yl)-2-methyl-propionic acid amide

-
-
60-29-7
diethyl ether

-
-
64-17-5
ethanol

-
A
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
64-17-5
ethanol

-
-
762-98-1
2,2-dimethyl-1,3-dinitropropane

-
A
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
B
-
41882-44-4
2,2-dimethylmalonamide

| Conditions | Yield |
|---|---|
| at 60℃; under 51485.6 Torr; Hydrogenation; | |
| at 60℃; under 51485.6 Torr; Hydrogenation; |

-
-
7647-01-0
hydrogenchloride

-
-
114583-17-4
α-(nitromethyl)isobutyronitrile

-
A
-
859065-87-5
2-(5,5-dimethyl-hexahydro-pyrimidin-2-yl)-2-methyl-propionic acid amide

-
B
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
C
-
67744-70-1
3-amino-2,2-dimethyl-propionitrile

-
D
-
324763-51-1
aminopivalinamide

| Conditions | Yield |
|---|---|
| at 60 - 70℃; | |
| at 60 - 70℃; |

-
-
67-56-1
methanol

-
-
114583-17-4
α-(nitromethyl)isobutyronitrile

-
A
-
859065-87-5
2-(5,5-dimethyl-hexahydro-pyrimidin-2-yl)-2-methyl-propionic acid amide

-
B
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
C
-
67744-70-1
3-amino-2,2-dimethyl-propionitrile

-
D
-
324763-51-1
aminopivalinamide

| Conditions | Yield |
|---|---|
| Hydrogenation; | |
| Hydrogenation; |

-
-
67-56-1
methanol

-
-
114583-17-4
α-(nitromethyl)isobutyronitrile

-
A
-
859065-87-5
2-(5,5-dimethyl-hexahydro-pyrimidin-2-yl)-2-methyl-propionic acid amide

-
B
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
C
-
67744-70-1
3-amino-2,2-dimethyl-propionitrile

-
D
-
324763-51-1
aminopivalinamide

| Conditions | Yield |
|---|---|
| Hydrogenation; | |
| Hydrogenation; |

-
-
114583-17-4
α-(nitromethyl)isobutyronitrile

-
A
-
859065-87-5
2-(5,5-dimethyl-hexahydro-pyrimidin-2-yl)-2-methyl-propionic acid amide

-
B
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
C
-
67744-70-1
3-amino-2,2-dimethyl-propionitrile

-
D
-
324763-51-1
aminopivalinamide

| Conditions | Yield |
|---|---|
| at 100℃; Hydrogenation; | |
| at 100℃; Hydrogenation; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 82 percent / 28percent NH3 2: 75 percent / BH3*SMe2 / tetrahydrofuran / 2 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 71 percent / SOCl2 / 20 h / Heating 2: 82 percent / 28percent NH3 3: 75 percent / BH3*SMe2 / tetrahydrofuran / 2 h / Heating View Scheme |
-
-
114583-17-4
α-(nitromethyl)isobutyronitrile

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: iron; aqueous HCl / 60 - 70 °C 2: Raney nickel; methanol / Hydrogenation View Scheme |
-
-
31044-87-8
neopentyl glycol bis(benzenesulfonate)

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaN3 / bis-(2-hydroxy-ethyl) ether / 16 h / 135 °C 2: LiAlH4 / tetrahydrofuran / 18 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: pyridine / 24 h / 10 °C 2: NaN3 / bis-(2-hydroxy-ethyl) ether / 16 h / 135 °C 3: LiAlH4 / tetrahydrofuran / 18 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| With silver nitrate In sodium hydroxide; water |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
40244-90-4
Chlorodiisopropylphosphane

-
-
596106-62-6
N,N'-1,3-bis(P,P'-diisopropylthiophosphinic)-2,2-dimethylpropylene diamine

| Conditions | Yield |
|---|---|
| Stage #1: 2,2-Dimethyl-1,3-diaminopropane; Chlorodiisopropylphosphane With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 23℃; Stage #2: With sulfur at 80℃; for 0.5h; | 100% |
| Stage #1: 2,2-Dimethyl-1,3-diaminopropane; Chlorodiisopropylphosphane With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 25℃; Stage #2: With sulfur In dichloromethane at 20℃; for 2h; | 72% |
| Stage #1: 2,2-Dimethyl-1,3-diaminopropane; Chlorodiisopropylphosphane With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; Stage #2: With sulfur In dichloromethane at 20℃; for 2h; | 68% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 4h; Reflux; | 100% |
| With formic acid In methanol for 24h; Reflux; Inert atmosphere; | 54% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
148-53-8
3-methoxy-2-hydroxybenzaldehyde

-
-
188482-90-8
N,N′-bis(3-methoxysalicylidene)-2,2-dimethyl-1,3-propanediamine

| Conditions | Yield |
|---|---|
| In methanol at 22℃; for 4h; | 100% |
| In ethanol for 1h; Reflux; | 92% |
| In ethanol for 1h; Reflux; | 92% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
383-63-1
ethyl trifluoroacetate,

-
-
1321622-57-4
N-(3-amino-2,2-dimethylpropyl)-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 20℃; for 15h; Inert atmosphere; | 100% |
| In tetrahydrofuran at 20℃; for 14.5h; Cooling with ice; |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
563-63-3
silver(I) acetate

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

| Conditions | Yield |
|---|---|
| Stage #1: 2,2-Dimethyl-1,3-diaminopropane; C40H76N2O2(2+)*2NO3(1-) In dichloromethane at 20℃; Stage #2: cobalt(II) acetate In dichloromethane at 20℃; for 12h; Stage #3: silver(I) acetate In dichloromethane for 12h; | 100% |


| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 4h; Reflux; | 100% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With calcium hydride In acetonitrile at 0℃; Reflux; | 99.23% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
14562-10-8
2-hydroxy-3-phenylbenzaldehyde

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 99% |
| In methanol at 20℃; for 0.333333h; | 75% |
| In methanol | |
| In methanol for 2h; Reflux; | |
| In methanol at 20℃; for 0.5h; |

| Conditions | Yield |
|---|---|
| Stage #1: pent-4-enoic acid With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0℃; for 0.5h; Stage #2: 2,2-Dimethyl-1,3-diaminopropane In dichloromethane at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| In methanol for 3h; | 99% |
-
-
140840-53-5
3-(tert-butyldimethylsilyl)-2-hydroxybenzaldehyde

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
1211569-73-1
tert-butyl [4-(4,5-dihydro-1H-imidazol-2-yl)benzyl]methylcarbamate

-
-
1211569-84-4
tert-butyl [4-(5,5-dimethyl-1,4,5,6-tetrahydropyrimidin-2-yl)benzyl]methylcarbamate

| Conditions | Yield |
|---|---|
| at 155℃; for 1h; | 98% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
37942-07-7
3,5-di-tert-butyl-2-hydroxybenzaldehyde

-
-
175291-16-4
(2,2-dimethylpropandiyl)bis(nitrilomethylidyne)bis(2,4-di-tert-butyl)phenol

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |
| In ethanol at 50 - 70℃; for 2.25h; | 90% |
| 88% |

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
5970-45-6
zinc(II) acetate dihydrate

-
-
148063-59-6
3,6-diformylcatechol

| Conditions | Yield |
|---|---|
| Stage #1: lanthanum(III) nitrate hexahydrate; zinc(II) acetate dihydrate; 3,6-diformylcatechol In methanol; chloroform; water at 20℃; for 0.5h; Stage #2: 2,2-Dimethyl-1,3-diaminopropane In chloroform at 20℃; for 18h; | 98% |

-
-
573675-27-1
3-amino-5-bromopyridine-2-carbonitrile

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| In N,N-dimethyl acetamide at 160℃; Sealed tube; | 98% |

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
148063-59-6
3,6-diformylcatechol

| Conditions | Yield |
|---|---|
| Stage #1: zinc(II) nitrate hexahydrate; cerium(III) acetate monohydrate; 3,6-diformylcatechol In methanol; chloroform; water at 20℃; for 17h; Stage #2: 2,2-Dimethyl-1,3-diaminopropane In methanol at 20℃; for 1h; | 98% |

-
-
83-72-7
2-Hydroxy-1,4-naphthoquinone

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
403-32-7
(4-fluorophenyl)glyoxal

-
-
13623-94-4
1,1-di(methylsulfanyl)-2-nitroethylene

| Conditions | Yield |
|---|---|
| Stage #1: 2,2-Dimethyl-1,3-diaminopropane; 1,1-di(methylsulfanyl)-2-nitroethylene In ethanol; water for 4h; Reflux; Stage #2: 2-hydroxynaphtho-1,4-quinone; (4-fluorophenyl)glyoxal In ethanol; water at 60℃; for 0.0833333h; regioselective reaction; | 97% |

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| In ethanol for 5h; Reflux; | 97% |

| Conditions | Yield |
|---|---|
| at 90℃; | 96% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
596106-64-8
N,N'-bis(P,P-diphenylphosphinoyl)-2,2-dimethyl-1,3-propanediamine

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; | 96% |
| With triethylamine at 0℃; for 6h; |
-
-
865075-67-8
[bis(1H-benzotriazol-1-yl)methylidene](4-chlorophenyl)amine

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| In toluene for 1h; Heating; | 96% |
-
-
107-20-0
2-chloroethanal

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
298-14-6
potassium hydrogencarbonate

-
-
1580482-20-7
(RS)-3,3-dimethylhexahydro[1,3]oxazolo[3,4-a]pyrimidin-6-one

| Conditions | Yield |
|---|---|
| In water at 20℃; for 10h; Green chemistry; | 96% |

-
-
140840-53-5
3-(tert-butyldimethylsilyl)-2-hydroxybenzaldehyde

-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| In ethanol at 20 - 80℃; for 2h; Inert atmosphere; | 96% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 4h; | 96% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
24424-99-5
di-tert-butyl dicarbonate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol | 96% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
67-64-1
acetone

-
-
31892-16-7
N,N'-diisipropyl-2,2-dimethyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol under 51714.8 Torr; | 95% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
57943-60-9
4-hydroxybenzimidic acid methyl ester hydrochloride

| Conditions | Yield |
|---|---|
| In ethanol for 18h; cycloamynation; Pinner synthesis; Heating; | 95% |
2,2-Dimethyl-1,3-propanediamine Specification
The CAS registry number of Neopentyldiamine is 7328-91-8. Its EINECS registry number is 230-819-0. The IUPAC name is 2,2-dimethylpropane-1,3-diamine. In addition, the molecular formula is C5H14N2 and the molecular weight is 102.18. It is also called 1,3-propanediamine,2,2-dimethyl-. What's more, it belongs to the classes of Amine Monomers; Monomers; Primary Amines. And it should be stored in a cool and dry place.
Physical properties about this chemical are: (1)ACD/LogP: -0.73; (2)ACD/LogD (pH 5.5): -4.82; (3)ACD/LogD (pH 7.4): -4.55; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 1; (7)ACD/KOC (pH 7.4): 1; (8)#H bond acceptors: 2; (9)#H bond donors: 4; (10)#Freely Rotating Bonds: 4; (11)Polar Surface Area: 6.48 Å2; (12)Index of Refraction: 1.456; (13)Molar Refractivity: 32.24 cm3; (14)Molar Volume: 118.4 cm3; (15)Polarizability: 12.78 ×10-24cm3; (16)Surface Tension: 33.1 dyne/cm; (17)Density: 0.862 g/cm3; (18)Flash Point: 47.2 °C; (19)Enthalpy of Vaporization: 38.72 kJ/mol; (20)Boiling Point: 150.4 °C at 760 mmHg; (21)Vapour Pressure: 3.86 mmHg at 25°C.
Preparation of Neopentyldiamine: it can be prepared by dimethyl-malonic acid diamide. This reaction will need reagent BH3*SMe2 and solvent tetrahydrofuran. The reaction time is 2 hours by heating. The yield is about 75%.

Uses of Neopentyldiamine: it can react with formaldehyde to get 5,5-dimethylhexahydropyrimidine. This reaction will need solvent H2O. The reaction time is 24 hours at reaction temperature of 20 °C. The yield is about 88%.
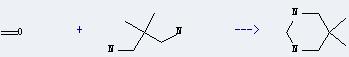
When you are using this chemical, please be cautious about it as the following:
This chemical is flammable and can cause burns. During using it, wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell, seek medical advice immediately (show label where possible). What's more, you should keep away from sources of ignition and no smoking. And you should take off immediately all contaminated clothing.
You can still convert the following datas into molecular structure:
(1)SMILES: NCC(C)(C)CN
(2)InChI: InChI=1/C5H14N2/c1-5(2,3-6)4-7/h3-4,6-7H2,1-2H3
(3)InChIKey: DDHUNHGZUHZNKB-UHFFFAOYAL
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 73289-66-4
- 73290-22-9
- 73290-32-1
- 73291-09-5
- 73292-20-3
- 732-92-3
- 7329-50-2
- 7329-52-4
- 732956-04-6
- 73296-86-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View