 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| With reduced Sn/hydrotalcite catalyst at 250℃; under 25502.6 Torr; Catalytic behavior; Reagent/catalyst; Temperature; Pressure; | 99% |
| With SA5 at 199.84℃; Catalytic behavior; Reagent/catalyst; Temperature; Inert atmosphere; | 2.6% |
| With sulfuric acid at 130 - 140℃; Darstellung im grossen; |

| Conditions | Yield |
|---|---|
| With alumina at 449.84℃; Catalytic behavior; Reagent/catalyst; Temperature; Inert atmosphere; Overall yield = 100 %; | A 0.1% B 98.9% |
| C2I2O2Rh(1-)*C8H20N(1+); tetraethylammonium iodide; hydrogen iodide In water at 110℃; Product distribution / selectivity; Inert atmosphere; Autoclave; | A 10% B 50% |
| 1-methyl-3-(propyl-3-sulfonyl)imidazolium trifluoromethanesulfonate; CF3O3S(1-)*CHF3O3S*C7H13N2O3S(1+) at 240 - 260℃; for 4h; Product distribution / selectivity; | A n/a B 12% |
-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
65039-09-0
1-ethyl-3-methyl-1H-imidazol-3-ium chloride

-
A
-
377739-43-0
1-ethyl-3-methyl-imidazolium tris(pentafluoroethyl)trifluorophosphate

-
B
-
60-29-7
diethyl ether

-
C
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; Product distribution / selectivity; | A 98.9% B n/a C n/a |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
30388-20-6
N,N,N',N',N'',N''-hexamethylguanidinium chloride

-
B
-
60-29-7
diethyl ether

-
C
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A 98.9% B n/a C n/a |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
155371-19-0
1-ethyl-3-methylimidazolium hexafluorophosphate

-
A
-
377739-43-0
1-ethyl-3-methyl-imidazolium tris(pentafluoroethyl)trifluorophosphate

-
B
-
60-29-7
diethyl ether

-
C
-
353-36-6
1-fluoroethane

-
D
-
7647-19-0, 874483-74-6
phosphorus pentafluoride

| Conditions | Yield |
|---|---|
| at 100℃; for 10h; Product distribution / selectivity; | A 98.7% B n/a C n/a D n/a |

-
-
945614-34-6
triethyloxonium bis(trifluoromethylsulfonyl)imide

-
-
59016-54-5
1-cyano-4-N,N-dimethylaminopyridinium bromide

-
A
-
74-96-4
ethyl bromide

-
B
-
60-29-7
diethyl ether

-
C
-
945614-38-0
1-cyano-4-dimethylaminopyridinium bis(trifluoromethylsulfonyl)imide

| Conditions | Yield |
|---|---|
| at 60℃; for 5h; | A n/a B n/a C 98.2% |

-
-
945614-34-6
triethyloxonium bis(trifluoromethylsulfonyl)imide

-
-
65039-09-0
1-ethyl-3-methyl-1H-imidazol-3-ium chloride

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

-
C
-
174899-82-2
1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A n/a B n/a C 97.9% |

-
-
766-77-8
Dimethylphenylsilane

-
-
123-63-7
paracetaldehyde

-
A
-
60-29-7
diethyl ether

-
B
-
56-33-7
1,1,3,3-tetramethyl-1,3-diphenyldisiloxane

| Conditions | Yield |
|---|---|
| With (pentamethylcyclopentadienyl)Ge(II)+B(ArF)4- In dichloromethane-d2 at 50℃; Catalytic behavior; Reagent/catalyst; | A n/a B 97% |


| Conditions | Yield |
|---|---|
| With triethylsilane; [CpW(CO)2(1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene)]B(C6F5)4 at 23℃; for 26h; Conversion of starting material; | A 5.9% B 96.3% |

-
-
3790-23-6
tetraethyldiphosphine disulfide

-
A
-
115018-02-5
((CH3)5C5)2Yb(S2P(C2H5)2)

-
B
-
60-29-7
diethyl ether

-
C
-
3040-63-9
1,1,2,2-tetraethyldiphosphane

| Conditions | Yield |
|---|---|
| In toluene stirring, 2 h, under N2; concn., cooling to -10°C; elem. anal.; | A 96% B n/a C n/a D n/a |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

-
C
-
916807-26-6
1-decyl-3-methylimidazolium tris(pentafluoroethyl)trifluorophosphate

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A n/a B n/a C 96% |

-
-
776-76-1
methyldiphenylsilane

-
-
123-63-7
paracetaldehyde

-
A
-
60-29-7
diethyl ether

-
B
-
807-28-3
1,3-Dimethyl-1,1,3,3-tetraphenyldisiloxan

| Conditions | Yield |
|---|---|
| With (pentamethylcyclopentadienyl)Ge(II)+B(ArF)4- In dichloromethane-d2 at 50℃; Catalytic behavior; | A n/a B 96% |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
76-83-5
trityl chloride

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| at 80℃; for 10h; | A n/a B n/a C 93.6% |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
59016-54-5
1-cyano-4-N,N-dimethylaminopyridinium bromide

-
A
-
74-96-4
ethyl bromide

-
B
-
60-29-7
diethyl ether

-
C
-
945614-37-9
1-cyano-4-dimethylaminopyridinium tris(pentafluoroethyl)trifluorophosphate

| Conditions | Yield |
|---|---|
| at 60℃; for 5h; | A n/a B n/a C 93.2% |


-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

-
C
-
945614-40-4
1-hexyl-1-methylpyrrolidinium tris(pentafluoroethyl)trifluorophosphate

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A n/a B n/a C 93% |


| Conditions | Yield |
|---|---|
| With NaX faujasite at 180 - 240℃; for 6h; | A 93% B 5% |
-
-
74-85-1
ethene

-
-
64-19-7
acetic acid

-
A
-
60-29-7
diethyl ether

-
B
-
64-17-5
ethanol

-
C
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| Stage #1: acetic acid With water at 92.4℃; Stage #2: ethene; cesium nitrate; tungstophosphoric acid; water; mixture of, dried, tabletted at 92.4 - 194.4℃; under 6750.68 Torr; Product distribution / selectivity; Gas phase; | A 3.2% B 3.6% C 92.7% |
| With water; cesium nitrate; tungstophosphoric acid; water; mixture of, dried, tabletted at 92.4 - 165℃; under 6750.68 Torr; Product distribution / selectivity; Gas phase; | A 3% B 3.4% C 91.5% |
| With water; lithium nitrate; silica; tungstophosphoric acid; water; mixture of, heated at 150 C at 102.2 - 165℃; under 6750.68 Torr; Product distribution / selectivity; Gas phase; | A 2.2% B 5% C 90.1% |
| With water; lithium nitrate; silica; tungstosilicic acid; water; mixture of, heated at 150 C at 102.2 - 165℃; under 6750.68 Torr; Conversion of starting material; Gas phase; | A 4.7% B 7.6% C 87.7% |

-
-
74-85-1
ethene

-
-
79-10-7
acrylic acid

-
A
-
60-29-7
diethyl ether

-
B
-
64-17-5
ethanol

-
C
-
140-88-5
ethyl acrylate

| Conditions | Yield |
|---|---|
| With water; cesium nitrate; tungstophosphoric acid; water; mixture of, dried, tabletted at 85.6 - 165℃; under 2250.23 Torr; Product distribution / selectivity; Gas phase; | A 3.5% B 4.3% C 91.8% |

-
-
64-17-5
ethanol

-
-
15148-19-3
1,3-bis(p-nitrophenyl)-2-thia-1,3-diazaallene

-
A
-
60-29-7
diethyl ether

-
B
-
623-81-4
diethyl sulphite

-
C
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With copper dichloride for 24h; Product distribution; Ambient temperature; other reagent; | A 93.6 % Chromat. B 70% C 91% |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
143314-16-3
1-ethyl-3-methylimidazolium tetrafluoroborate

-
A
-
377739-43-0
1-ethyl-3-methyl-imidazolium tris(pentafluoroethyl)trifluorophosphate

-
B
-
60-29-7
diethyl ether

-
C
-
353-36-6
1-fluoroethane

-
D
-
7637-07-2
boron trifluoride

| Conditions | Yield |
|---|---|
| at 100℃; for 10h; Product distribution / selectivity; | A 90.3% B n/a C n/a D n/a |

-
-
60-29-7
diethyl ether

-
-
74415-68-2
hexafluoro-3-oxatricyclo<3.2.0.02,4>hept-6-ene

-
A
-
353-36-6
1-fluoroethane

| Conditions | Yield |
|---|---|
| for 2160h; Ambient temperature; | A 100% B 75% |
| for 2160h; Yields of byproduct given; | A n/a B 21% |


| Conditions | Yield |
|---|---|
| With hexafluoro-3-oxatricyclo<3.2.0.02,4>hept-6-ene for 2160h; Ambient temperature; | A 100% B 75% |
-
-
60-29-7
diethyl ether

-
-
68602-57-3
trifluoroacetyl triflate

-
A
-
383-63-1
ethyl trifluoroacetate,

-
B
-
425-75-2
trifluoromethanesulfonic acid ethyl ester

| Conditions | Yield |
|---|---|
| at 0℃; | A 100% B 100% |

-
-
60-29-7
diethyl ether

| Conditions | Yield |
|---|---|
| With lithium at 20℃; for 20h; Reduction; | 100% |
-
-
60-29-7
diethyl ether


| Conditions | Yield |
|---|---|
| With lithium at 20℃; for 20h; Reduction; | 100% |
-
-
60-29-7
diethyl ether

-
-
444-30-4
2-(trifluoromethyl)phenol

-
-
35335-16-1
1,4-bis(bromomethyl)-2,5-dibromobenzene

-
-
474330-25-1
1,4-dibromo-2,5-bis(2-trifluoromethylphenoxymethyl)benzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide; potassium carbonate In dichloromethane; acetone | 100% |

-
-
193902-67-9
4-[[[4-[4-(ethyloxycarbonyl)-1-piperazinyl]phenyl]amino]carbonyl]-1-t-butyloxycarbonyl-piperidine

-
-
60-29-7
diethyl ether

-
-
193902-68-0
4-[[[4-[4-(ethyloxycarbonyl)-1-piperazinyl]phenyl]amino]carbonyl]piperidine

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; hydrogenchloride | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 3,4-dihydro-2H-pyran | 100% |
-
-
79-37-8
oxalyl dichloride

-
-
60-29-7
diethyl ether

-
-
20017-67-8
3,3-diphenylpropan-1-ol

-
-
4279-81-6
3,3-diphenylpropanal

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide; triethylamine In dichloromethane | 100% |

-
-
63606-55-3
2-(2-ethyl-benzofuran-3-yl)-propionic acid

-
-
60-29-7
diethyl ether

-
-
63606-56-4
2-(2-ethyl-benzofuran-3-yl)-propionamide

| Conditions | Yield |
|---|---|
| In thionyl chloride | 100% |

| Conditions | Yield |
|---|---|
| With n-butyllithium In hexane | 100% |
| With n-butyllithium In hexane |
-
-
60-29-7
diethyl ether

-
-
52341-13-6, 12264-20-9
trans-dichloro(ethylene)(2,4,6-trimethylpyridine)platinum

-
-
91068-18-7
trans-dichloro(diethyl ether)(2,4,6-trimethylpyridine)platinum(II)

| Conditions | Yield |
|---|---|
| In diethyl ether byproducts: ethylene; Irradiation (UV/VIS); | 100% |
| In diethyl ether Irradiation (UV/VIS); the Pt-complex dissolved in Et2O was introduced into a muffshaped Schlenk tube surrounding a 125-W medium-pressure mercury lamp, Philips HPK 125, irradn. for 15 min at room temp., λ<310 nm was eliminated by Pyrex filter; the solvent was removed under reduced pressure at -30°C, the solid was recrystd. at -30°C in pentane-CH2Cl2; | 95% |

| Conditions | Yield |
|---|---|
| In diethyl ether (high vac. line); condensing gallium complex in an ampoule with Et2O, warming to room temp. over a period of 30 min; fractionation, collection in a trap at -30°C; | 100% |
-
-
60-29-7
diethyl ether

-
-
293764-40-6
[(C5H4N)C(CH3)(CH2N(C6H2(CH3)3))2]Zr(CH3)2

-
-
917-54-4
methyllithium

-
-
486413-21-2
[C5H4NC(CH3)(CH2NC6H2(CH3)3)2]Zr(methyl)3[Li*diethyl ether]

| Conditions | Yield |
|---|---|
| In diethyl ether N2; addn. of methyllithium as 4.4 M ether soln. to ether suspn. of Zr complex at -30° C, stirring at room temp. for 10 min; filtration through Celite, drying the filtrate in vac.; elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| In diethyl ether; benzene-d6 byproducts: ethane; N2-atmosphere; room temp. (20 min); | A 100% B n/a |


| Conditions | Yield |
|---|---|
| In benzene-d6 byproducts: ethane; N2-atmosphere; room temp. (10 min); evapn. (vac.); | 100% |
-
-
60-29-7
diethyl ether

| Conditions | Yield |
|---|---|
| In dichloromethane (inert conditions); removal of volatiles (vac.); | 100% |

| Conditions | Yield |
|---|---|
| In diethyl ether (N2), GaCl3 in Et2O added dropwise to soln. of LiAlH4 in Et2O at 0°C, stirred at 0°C for 2 h; stored overnight at -20°C, filtered cold, evapd. at -78°C; | 100% |

-
-
60-29-7
diethyl ether

-
A
-
1244773-04-3
(1S,3aR,5S,5'S,6R,6a'R)-2,2'-dimethyl-5'-(2-methyl-1,3-dioxolan-4-yl)dihydro-3a'H-3-oxaspiro[bicyclo[3.2.0.]heptane-6,6'-furo[2,3-d][1,3]dioxole]

-
B
-
108-94-1
cyclohexanone

| Conditions | Yield |
|---|---|
| With copper(II) bis(trifluoromethanesulfonate); benzene Inert atmosphere; Irradiation; | A 65% B 100% |

-
-
60-29-7
diethyl ether

-
-
1235436-62-0
bis(trimethylsilyl)-trifluoromethylsulfonium tetrakis(pentafluorophenyl)borate

| Conditions | Yield |
|---|---|
| react. bis(trimethylsilyl)-trifluoromethylsulfonium tetrakis(pentafluorophenyl)borate with Et2O; | 100% |

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |


| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |


| Conditions | Yield |
|---|---|
| at 20℃; for 1h; Inert atmosphere; | 100% |

-
-
60-29-7
diethyl ether

-
-
1445605-48-0
Ce(decafluorodiphenylamide)3(diethyl ether)2

| Conditions | Yield |
|---|---|
| for 0.5h; Inert atmosphere; | 100% |

-
-
60-29-7
diethyl ether

-
-
1365891-80-0, 1365891-81-1
(R,Rb)-[1,1'-binaphthalene]-2,2'-diyl(2'-methoxy-[1,1'-binaphthalen]-2-yl)phosphonite

-
-
1436385-41-9
[RhCl((R,R)-C41H27O3P)(η4-cod)]*(C2H5)2O

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.5h; Inert atmosphere; Schlenk technique; | 100% |


| Conditions | Yield |
|---|---|
| In toluene Inert atmosphere; Glovebox; | 100% |


| Conditions | Yield |
|---|---|
| With alumina at 270℃; under 760.051 Torr; Inert atmosphere; Gas phase; Green chemistry; | 100% |

| Conditions | Yield |
|---|---|
| With magnesium Heating; | 100% |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View


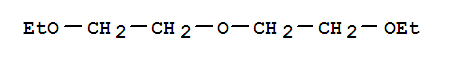












 F+,
F+,  Xn,
Xn,  T
T