-
Name
2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose
- EINECS 619-281-1
- CAS No. 97614-43-2
- Article Data12
- CAS DataBase
- Density 1.35 g/cm3
- Solubility
- Melting Point 74-77°C
- Formula C26H21FO7
- Boiling Point 584.1 °C at 760 mmHg
- Molecular Weight 464.447
- Flash Point 295.7 °C
- Transport Information
- Appearance
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms a-D-Arabinofuranose,2-deoxy-2-fluoro-, tribenzoate (9CI);2'-Fluoro-2'-deoxy-1,3,5-tri-O-benzoyl-a-D-arabinofuranose;1,3,5-Tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinose;
- PSA 88.13000
- LogP 3.98900
Synthetic route
-
-
97614-41-0
2-O-<(trifluoromethyl)sulfonyl>-1,3,5-tri-O-benzoyl-α-D-ribofuranose

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| With triethylamine tris(hydrogen fluoride) In ethyl acetate at 80℃; for 24h; Substitution; | 85% |
| With triethylamine tris(hydrogen fluoride); triethylamine In acetonitrile at 50℃; for 18h; | |
| With hydrogen fluoride; tetrabutylammonium bicarbonate In acetonitrile at 80℃; for 0.5h; |
-
-
22224-41-5
1,3,5-tri-O-benzoyl-α-D-ribofuranoside

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| With (bis-(2-methoxyethyl)amino)sulfur trufluoride In dichloromethane at 50℃; | 83% |
| With diethylamino-sulfur trifluoride In dichloromethane at 40℃; | 75% |
| Multi-step reaction with 2 steps 1: pyridine / acetonitrile / 0.5 h / 20 °C 2: Et3N(HF)3; Et3N / acetonitrile / 18 h / 50 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 100 percent / CH2Cl2; pyridine / -10 °C 2: 85 percent / Et3N*3HF / ethyl acetate / 24 h / 80 °C View Scheme | |
| With diethylamino-sulfur trifluoride |
-
-
97614-42-1
2-O-(imidazolylsulfonyl)-1,3,5-tri-O-benzoyl-α-D-ribofuranose

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| With triethylamine tris(hydrogen fluoride) In ethyl acetate at 60 - 70℃; for 4.5h; | 66.4% |
| Multi-step reaction with 2 steps 1: 77 percent / Et3N*HF / ethyl acetate 2: (But)4NF / ethyl acetate / 0.33 h / 70 °C View Scheme | |
| Multi-step reaction with 2 steps 1: Et3N*3HF / ethyl acetate / 20 - 60 °C 2: Et3N*3HF / ethyl acetate / 70 °C View Scheme |
-
-
97614-42-1
2-O-(imidazolylsulfonyl)-1,3,5-tri-O-benzoyl-α-D-ribofuranose

-
A
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
B
-
97614-50-1
2-O-(fluorosulfonyl)-1,3,5-tri-O-benzoyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| With potassium hydrogen bifluoride; hydrogen fluoride In various solvent(s) at 160℃; for 1h; | A 62.8% B n/a |
-
-
97614-50-1
2-O-(fluorosulfonyl)-1,3,5-tri-O-benzoyl-α-D-ribofuranose

-
A
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; triethylamine In ethyl acetate at 71 - 72℃; for 4h; Product distribution; Further Variations:; Reagents; Solvents; reaction time; Substitution; | A 60.5% B n/a |
-
-
97614-41-0
2-O-<(trifluoromethyl)sulfonyl>-1,3,5-tri-O-benzoyl-α-D-ribofuranose

-
A
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In ethyl acetate at 71 - 72℃; for 0.333333h; Substitution; | A 34% B n/a |
-
-
107-21-1
ethylene glycol

-
-
97614-42-1
2-O-(imidazolylsulfonyl)-1,3,5-tri-O-benzoyl-α-D-ribofuranose

-
A
-
38491-60-0
β-phenyl hydroxypropionate

-
B
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
C
-
97614-49-8
C28H26O9

| Conditions | Yield |
|---|---|
| With potassium hydrogen bifluoride; hydrogen fluoride In various solvent(s) at 160℃; for 1h; Product distribution; other reaction conditions; |

-
-
97614-50-1
2-O-(fluorosulfonyl)-1,3,5-tri-O-benzoyl-α-D-ribofuranose

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| With triethylamine tris(hydrogen fluoride) In ethyl acetate at 70℃; Yield given; | |
| With tetrabutyl ammonium fluoride In ethyl acetate at 70℃; for 0.333333h; Substitution; |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
97614-44-3
2-deoxy-2-fluoro-3,5-di-O-benzoyl-α-D-arabinofuranosyl bromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid In dichloromethane at -5 - 20℃; for 18.33h; Inert atmosphere; | 100% |
| With hydrogen bromide; acetic acid In dichloromethane for 20h; Ambient temperature; | 99% |
| With hydrogen bromide; acetic acid In dichloromethane at 20℃; | 99% |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
98855-71-1
((2R,3R,4S)-3-(benzoyloxy)-5-bromo-4-fluorotetrahydrofuran-2-yl)methyl benzoate

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid | 99% |
| With hydrogen bromide; acetic acid In dichloromethane at 20℃; | 99% |
| With acetic acid |
-
-
75-09-2
dichloromethane

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
98855-71-1
((2R,3R,4S)-3-(benzoyloxy)-5-bromo-4-fluorotetrahydrofuran-2-yl)methyl benzoate

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid | 90% |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
28523-86-6
sevoflurane

| Conditions | Yield |
|---|---|
| With potassium fluoride In various solvent(s) at 95℃; for 1h; Substitution; | 71% |
-
-
111-87-5
octanol

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
B
-
1131273-34-1
octyl 2-deoxy-2-fluoro-3,5-di-O-benzoyl-α-D-arabinofuranoside

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose With tin(IV) chloride In acetonitrile at 20℃; for 0.25h; Molecular sieve; Inert atmosphere; Stage #2: octanol In acetonitrile at 20℃; for 1.5h; Inert atmosphere; Stage #3: With triethylamine In dichloromethane | A n/a B 54% |

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
75788-35-1
9-Trimethylsilyl-2,6-dichlorpurin

-
C
-
329187-80-6
((2R,3R,4S,5R)-3-(benzoyloxy)-5-(2,6-dichloro-9H-purin-9-yl)-4-fluorotetrahydrofuran-2-yl)methyl benzoate

-
D
-
329187-81-7
C24H17Cl2FN4O5

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In acetonitrile for 0.333333h; Heating; | A 5% B n/a C 35% D 33% |

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
60855-35-8
6-benzoylamino-9-trimethylsilylpurine

-
A
-
20187-82-0, 20227-41-2, 64183-27-3, 123334-75-8
9-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)adenine

-
B
-
20187-82-0
9-(2-deoxy-2-fluoro-α-D-arabinofuranosyl)adenine

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose; 6-benzoylamino-9-trimethylsilylpurine With trimethylsilyl trifluoromethanesulfonate In acetonitrile for 4h; Heating; Stage #2: With ammonia In methanol at 20℃; for 48h; | A 14% B 14% C 25% |

-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
7288-28-0
2,4-bis(trimethylsiloxy)-5-methylpyrimidine

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In acetonitrile at 80 - 81℃; for 0.75h; |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
909101-48-0
4-amino-7-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)-5-fluoro-7H-pyrrolo[2,3-d]pyrimidine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: HBr; AcOH / CH2Cl2 / 16 h / 20 °C 2.1: KOH; tris[2-(2-methoxyethoxy)ethyl]amine / acetonitrile / 0.25 h / 20 °C 2.2: 61.7 percent / acetonitrile / 0.17 h / 20 °C 3.1: 72 percent / aq. NH3 / dioxane / 16 h / 120 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
909101-50-4
7-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)-5-fluoro-4-methoxy-7H-pyrrolo[2,3-d]pyrimidine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: HBr; AcOH / CH2Cl2 / 16 h / 20 °C 2.1: KOH; tris[2-(2-methoxyethoxy)ethyl]amine / acetonitrile / 0.25 h / 20 °C 2.2: 61.7 percent / acetonitrile / 0.17 h / 20 °C 3.1: 97 percent / NaOMe / 12 h / 20 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: HBr; AcOH / CH2Cl2 / 16 h / 20 °C 2.1: KOH; tris[2-(2-methoxyethoxy)ethyl]amine / acetonitrile / 0.25 h / 20 °C 2.2: 61.7 percent / acetonitrile / 0.17 h / 20 °C 3.1: 97 percent / NaOMe / 12 h / 20 °C 4.1: 37 percent / aq. NaOH / 1 h / Heating View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
909101-46-8
4-chloro-7-(2-deoxy-3,5-di-O-benzoyl-β-D-arabinofuranosyl)-5-fluoro-7H-pyrrolo[2,3-d]pyrimidine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: HBr; AcOH / CH2Cl2 / 16 h / 20 °C 2.1: KOH; tris[2-(2-methoxyethoxy)ethyl]amine / acetonitrile / 0.25 h / 20 °C 2.2: 61.7 percent / acetonitrile / 0.17 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: hydrogen bromide; acetic acid 2.1: Tris(3,6-dioxaheptyl)amine; potassium hydroxide / acetonitrile / 0.25 h / 20 °C 2.2: 0.17 h / 20 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C 3: sodium methoxide / methanol / 0.17 h / 80 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C 3: sodium methoxide / methanol / 0.17 h / 80 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C 3: sodium methoxide / methanol / 0.17 h / 80 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C 3: sodium methoxide / methanol / 0.17 h / 80 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
99097-06-0
1-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)-5-propyluracil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C 3: sodium methoxide / methanol / 0.17 h / 80 °C View Scheme |
-
-
97614-43-2
1,3,5-tri-O-benzoyl-2-deoxy-2-fluoro-α-D-arabinofuranose

-
-
114652-80-1
1-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)-5-(trifluromethyl)uracil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HBr; acetic acid / 1,2-dichloro-ethane / 0.17 h / 80 °C 2: 1,2-dichloro-ethane / 1 h / 110 °C 3: sodium methoxide / methanol / 0.17 h / 80 °C View Scheme |
2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose Chemical Properties
Molecular Structure of 2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose (CAS NO. 97614-43-2):
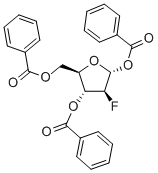
IUPAC Name: [(2S,3S,4S)-5-Benzoyloxy-2-(benzoyloxymethyl)-4-fluorooxolan-3-yl] benzoate
Molecular Formula: C26H21FO7
Molecular Weight: 464.439143 g/mol
XLogP3-AA: 5.2
H-Bond Donor: 0
H-Bond Acceptor: 8
Canonical SMILES: C1=CC=C(C=C1)C(=O)OCC2C(C(C(O2)OC(=O)C3=CC=CC=C3)F)OC(=O)C4=CC=CC=C4
Isomeric SMILES: C1=CC=C(C=C1)C(=O)OC[C@H]2[C@@H]([C@@H](C(O2)OC(=O)C3=CC=CC=C3)F)OC(=O)C4=CC=CC=C4
InChI: InChI=1S/C26H21FO7/c27-21-22(33-24(29)18-12-6-2-7-13-18)20(16-31-23(28)17-10-4-1-5-11-17)32-26(21)34-25(30)19-14-8-3-9-15-19/h1-15,20-22,26H,16H2/t20-,21-,22-,26?/m0/s1
InChIKey: JOAHVPNLVYCSAN-GAUDMLSGSA-N
Index of Refraction: 1.606
Molar Refractivity: 118.52 cm3
Molar Volume: 343.3 cm3
Surface Tension: 56.2 dyne/cm
Density: 1.35 g/cm3
Flash Point: 295.7 °C
Enthalpy of Vaporization: 87.29 kJ/mol
Boiling Point: 584.1 °C at 760 mmHg
Vapour Pressure: 1.24E-13 mmHg at 25 °C
Storage Temp.: 2-8 °C
2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose Safety Profile
Safety Information of 2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose (CAS NO. 97614-43-2):
WGK Germany: 3
2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose Specification
2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose with cas registry number of 97614-43-2 is also called for 1,3,5-Tri-O-benzoyl-2-deoxy-2-fluoro-alpha-D-arabinofuranose ; alpha-D-arabinofuranose, 2-deoxy-2-fluoro-, tribenzoate .
Related Products
- 2-Deoxy-2-fluoro-1,3,5-tri-O-benzoyl-D-ribofuranose
- 97614-44-3
- 97614-47-6
- 97-62-1
- 97628-91-6
- 97-63-2
- 97632-67-2
- 97633-99-3
- 97635-27-3
- 97635-28-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View