-
Name
Ethylene glycol monoethyl ether acetate
- EINECS 203-839-2
- CAS No. 111-15-9
- Article Data15
- CAS DataBase
- Density 0.961 g/cm3
- Solubility 230 g/L (20 °C) in water
- Melting Point -61 °C
- Formula C6H12O3
- Boiling Point 156.1 °C at 760 mmHg
- Molecular Weight 132.159
- Flash Point 50.6 °C
- Transport Information UN 1172 3/PG 3
- Appearance colourless liquid
- Safety 53-45
- Risk Codes 60-61-20/21/22-10
-
Molecular Structure
-
Hazard Symbols
 T
T
- Synonyms b-Ethoxyethyl acetate;Ethanol,2-ethoxy-, acetate (6CI,7CI,8CI,9CI);1-Acetoxy-2-ethoxyethane;2-Ethoxyethanolacetate;Ethanol, 2-ethoxy-,1-acetate;Cellosolve acetate;Ethyl cellosolve acetate;Ethylene glycol acetate monoethyl ether;Ethylene glycol ethyl ether acetate;Ethylene glycol monoethyl ether acetate;Ethylene glycol monoethyl ethermonoacetate;Glycol monoethyl ether acetate;Poly-Solv EE acetate;
- PSA 35.53000
- LogP 0.58600
Synthetic route

| Conditions | Yield |
|---|---|
| With dilithium tetra(tert-butyl)zincate In toluene at 0℃; for 1h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With indium (III) iodide for 17h; Heating; | 82% |

| Conditions | Yield |
|---|---|
| With indium; iodine for 14h; Heating; | 78% |

| Conditions | Yield |
|---|---|
| With indium (III) iodide for 12h; Heating; | 78% |
-
-
110-80-5
2-ethoxy-ethanol

-
-
546-67-8
lead(IV) tetraacetate

-
A
-
497-26-7
2-methyl-1,3-dioxolane

-
B
-
111-15-9
2-ethoxyethyl acetate

-
C
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| In chlorobenzene at 140℃; for 0.5h; | A 27% B 63% C 10% |

-
-
110-80-5
2-ethoxy-ethanol

-
A
-
497-26-7
2-methyl-1,3-dioxolane

-
B
-
111-15-9
2-ethoxyethyl acetate

-
C
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With lead(IV) acetate In chlorobenzene at 140℃; for 0.5h; glass tube; | A 27% B 63% C 10% |


| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
506-96-7
Acetyl bromide

-
-
61911-33-9
benzyl cellosolve

-
A
-
74-96-4
ethyl bromide

-
B
-
111-15-9
2-ethoxyethyl acetate

-
C
-
111-55-7
ethylene glycol diacetate

-
D
-
100-39-0
benzyl bromide

| Conditions | Yield |
|---|---|
| at 92℃; |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid man entfernt das entstehende Wasser durch azeotrope Destillation mit Benzol; |
-
-
13988-69-7
2-ethoxy-2-methyl-[1,3]dioxolane

-
-
111-15-9
2-ethoxyethyl acetate

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate |
-
-
129751-10-6
(Z)-1-acetoxy-2-ethoxyethylene

-
-
111-15-9
2-ethoxyethyl acetate

| Conditions | Yield |
|---|---|
| With hydrogen; nickel In acetone |
-
-
129751-09-3
(E)-2-Ethoxyvinylacetate

-
-
111-15-9
2-ethoxyethyl acetate

| Conditions | Yield |
|---|---|
| With hydrogen; nickel In acetone |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Al2O3 / 320 - 330 °C 2: H2 / Raney-Ni / acetone View Scheme | |
| Multi-step reaction with 2 steps 1: Al2O3 / 320 - 330 °C 2: H2 / Raney-Ni / acetone View Scheme | |
| Multi-step reaction with 2 steps 1: Al2O3 / 320 - 330 °C 2: H2 / Raney-Ni / acetone View Scheme |

| Conditions | Yield |
|---|---|
| With tetrabutoxytitanium at 120℃; for 4h; |

| Conditions | Yield |
|---|---|
| With C15H17ClIrNOP; potassium tert-butylate In toluene at 60℃; for 12h; Schlenk technique; Inert atmosphere; Glovebox; | 82% |
-
-
628-81-9
butyl ethyl ether

-
-
111-15-9
2-ethoxyethyl acetate

-
-
117491-35-7
2-(1-(1H-benzo[d]imidazol-1-yl)ethoxy)ethyl acetate

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iron(III) chloride hexahydrate In decane; ethyl acetate at 80℃; for 3h; Inert atmosphere; Molecular sieve; | 73% |
-
-
111-15-9
2-ethoxyethyl acetate

| Conditions | Yield |
|---|---|
| With tin(II) trifluoromethanesulfonate at 60℃; for 14h; Criegee Rearrangement; Sealed tube; Inert atmosphere; | 70% |
-
-
120-72-9
indole

-
-
111-15-9
2-ethoxyethyl acetate

-
A
-
5030-91-1
vibrindole A

-
B
-
88321-08-8
2,2-di-(1H-indol-3-yl)ethyl acetate

| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide; iron(II) chloride at 80℃; for 1h; Inert atmosphere; | A 30% B 15% |

-
-
111-15-9
2-ethoxyethyl acetate

-
A
-
29776-97-4
1,2-ethanediol acetate formate

-
B
-
111-55-7
ethylene glycol diacetate

-
C
-
109-94-4
formic acid ethyl ester

| Conditions | Yield |
|---|---|
| With methyl nitrite; nitrogen(II) oxide In gas at 25℃; Product distribution; Rate constant; Irradiation; gas-phase reaction of 2-ethoxyethyl acetate with generated hydroxy radicals; proposed reaction mechanism; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; water at 27.5 - 48.5℃; Kinetics; |
2-Ethoxyethyl acetate Chemical Properties
Molecular Structure of 2-Ethoxyethyl acetate (CAS NO.111-15-9):
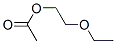
IUPAC Name: 2-Ethoxyethyl acetate
Molecular Weight: 132.15768 g/mol
Molecular Formula: C6H12O3
Density: 0.961 g/cm3
Melting Point: -61 °C
Boiling Point: 156.1 °C at 760 mmHg
Flash Point: 50.6 °C
Index of Refraction: 1.4
Molar Refractivity: 33.36 cm3
Molar Volume: 137.4 cm3
Surface Tension: 27.6 dyne/cm
Enthalpy of Vaporization: 39.28 kJ/mol
Vapour Pressure: 2.94 mmHg at 25 °C
Water Solubility: 230 g/L (20 °C)
XLogP3-AA: 0.3
H-Bond Acceptor: 3
Rotatable Bond Count: 5
Exact Mass: 132.078644
MonoIsotopic Mass: 132.078644
Topological Polar Surface Area: 35.5
Heavy Atom Count: 9
Canonical SMILES: CCOCCOC(=O)C
InChI: InChI=1S/C6H12O3/c1-3-8-4-5-9-6(2)7/h3-5H2,1-2H3
InChIKey: SVONRAPFKPVNKG-UHFFFAOYSA-N
EINECS: 203-839-2
Product Categories: Ethylene-Ethyl Acrylate Copolymer
2-Ethoxyethyl acetate Uses
2-Ethoxyethyl acetate (CAS NO.111-15-9) is used as a solvent dissolve resin, leather, ink.
2-Ethoxyethyl acetate Toxicity Data With Reference
| 1. | skn-rbt 490 mg open MLD | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 1/13 ,1967. | ||
| 2. | eye-rbt 40 mg MOD | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 1/13 ,1967. | ||
| 3. | orl-rat LD50:2900 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 51 (1979),117. | ||
| 4. | ihl-rat LC50:12,100 mg/m3/8H | AIHAAP American Industrial Hygiene Association Journal. 20 (1959),364. | ||
| 5. | ipr-mus LD50:1420 mg/kg | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) | ||
| 6. | orl-rbt LD50:1950 mg/kg | EPASR* United States Environmental Protection Agency, Office of Pesticides and Toxic Substances. (U.S. Environmental Protection Agency, 401M St., S.W., Washington, DC 20460) History Unknown 8EHQ-0682-0450 . | ||
| 7. | skn-rbt LD50:10,500 mg/kg | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 1/13 ,1967. |
2-Ethoxyethyl acetate Consensus Reports
Reported in EPA TSCA Inventory. Glycol ether compounds are on the Community Right-To-Know List.
2-Ethoxyethyl acetate Safety Profile
Hazard Codes:  T
T
Risk Statements: 60-61-20/21/22-10
R60:May impair fertility.
R61:May cause harm to the unborn child.
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
R10:Flammable.
Safety Statements: 53-45
S53:Avoid exposure - obtain special instructions before use.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
RIDADR: UN 1172 3/PG 3
WGK Germany: 1
RTECS: KK8225000
HazardClass: 3
PackingGroup: III
Moderately toxic by ingestion and intraperitoneal routes. A skin and eye irritant. An experimental teratogen. Other experimental reproductive effects. Flammable liquid when exposed to heat or flame; can react with oxidizing materials. Moderate explosion hazard in the form of vapor when heated. Mild explosions have occurred at the end of distillations. To fight fire, use alcohol foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also GLYCOL ETHERS.
2-Ethoxyethyl acetate Standards and Recommendations
OSHA PEL: TWA 100 ppm (skin)
ACGIH TLV: TWA 5 ppm (skin); BEI: 100 mg/g creatinine of 2-ethoxyacetic acid in urine end of shift at end of workweek
DFG MAK: 5 ppm (27 mg/m3)
DOT Classification: 3; Label: Flammable Liquid
2-Ethoxyethyl acetate Analytical Methods
For occupational chemical analysis use OSHA: #53 or NIOSH: Esters I, 1450.
2-Ethoxyethyl acetate Specification
2-Ethoxyethyl acetate (CAS NO.111-15-9) is also named as 1-Acetoxy-2-ethoxyethane ; 2-Aethoxy-aethylacetat ; 2-Aethoxy-aethylacetat [German] ; 2-Ethoxy-ethylacetaat ; 2-Ethoxy-ethylacetaat [Dutch] ; 2-Ethoxyethanol, ester with acetic acid ; 2-Ethoxyethylacetate ; 2-Ethoxyethyle, acetate de ; 2-Ethoxyethyle, acetate de [French] ; 2-Ethoxyethylester kyseliny octove ; 2-Ethoxyethylester kyseliny octove [Czech] ; 2-Etossietil-acetato ; 2-Etossietil-acetato [Italian] ; 4-02-00-00214 (Beilstein Handbook Reference) ; AI3-01955 (USDA) ; Acetate d'ethylglycol ; Acetate d'ethylglycol [French] ; Acetate de cellosolve ; Acetate de cellosolve [French] ; Acetate de l'ether monoethylique de l'ethylene-glycol ; Acetate de l'ether monoethylique de l'ethylene-glycol [French] ; Acetato di cellosolve ; Acetato di cellosolve [Italian] ; Acetic acid, 2-ethoxyethyl ester ; Ethanol, 2-ethoxy-, acetate ; Ethoxyethyl acetate ; Ethyleneglycol monoethyl ether acetate ; HSDB 539 ; NSC 8658 ; Octan etoksyetylu ; Octan etoksyetylu [Polish] ; Oxitol acetate ; Oxytol acetate ; Poly-Solv EE acetate . 2-Ethoxyethyl acetate (CAS NO.111-15-9) is colourless liquid with a pleasant odor. Vapors are heavier than air. It is flammable and slightly soluble in water.Mixing Ethylene glycol monoethyl ether acetate in equal molar portions with any of the following substances in a closed container caused the temperature and pressure to increase: chlorosulfonic acid, oleum, and vinyl acetate. Vapors irritate nose and eyes in high concentrations. Liquid irritates skin in prolonged or repeated contact. It will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated.
Related Products
- 2-Ethoxyethyl 2-bromoacetate
- 2-Ethoxyethyl acetate
- 2-Ethoxyethyl acrylate
- 2-Ethoxyethyl Benzoate
- 2-Ethoxyethyl chloroformate
- 2-Ethoxyethyl cyanoacetate
- 2-Ethoxyethyl ether
- 2-Ethoxyethyl methacrylate
- 2-Ethoxyethylamine
- 11115-92-7
- 111-16-0
- 11116-16-8
- 11116-32-8
- 1111637-68-3
- 1111637-69-4
- 1111637-72-9
- 1111637-93-4
- 1111637-94-5
- 1111638-01-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View