-
Name
2-Fluoro-4-iodoaniline
- EINECS
- CAS No. 29632-74-4
- Article Data8
- CAS DataBase
- Density 2.008 g/cm3
- Solubility insoluble in water
- Melting Point 55-57 °C(lit.)
- Formula C6H5FIN
- Boiling Point 248.6 °C at 760 mmHg
- Molecular Weight 237.015
- Flash Point 104.2 °C
- Transport Information
- Appearance off-white to brown powder
- Safety 26-36-37/39-26/37/39
- Risk Codes 36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  T,
T,  Xi
Xi
- Synonyms 2-fluoro-4-iodin anline;
- PSA 26.02000
- LogP 2.59370
Synthetic route
-
-
348-54-9
2-Fluoroaniline

-
-
29632-74-4
2-fluro-4-iodoaniline

| Conditions | Yield |
|---|---|
| With iodine; sodium hydrogencarbonate In water at 60 - 80℃; for 2h; | 77% |
| With iodine; sodium hydrogencarbonate In water at 60℃; for 3h; | 76% |
| Stage #1: 2-Fluoroaniline With n-butyllithium In diethyl ether; hexane at -78℃; for 1h; Stage #2: With poly[4-(dibutyliodostannyl)butyl]styrene In diethyl ether; hexane at 20℃; for 18h; Stage #3: With Iodine monochloride In diethyl ether; hexane at -78 - 20℃; | 67% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
1066-54-2
trimethylsilylacetylene

-
-
518342-58-0
2-fluoro-4-((trimethylsilyl)ethynyl)aniline

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; for 21h; Sonogashira Cross-Coupling; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; Inert atmosphere; | 100% |
| With triethylamine; bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide at 20℃; for 15h; Sonogashira Coupling; | 88% |
-
-
867130-26-5
4-chloro-1-methyl-6-oxo-1,6-dihydro-pyridazine-3-carboxylic acid

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
934664-24-1
4-(2-fluoro-4-iodophenylamino)-1-methyl-6-oxo-1,6-dihydropyridazine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-chloro-1-methyl-6-oxo-1,6-dihydro-pyridazine-3-carboxylic acid; 2-fluro-4-iodoaniline With lithium hexamethyldisilazane In tetrahydrofuran at 20℃; for 4h; Stage #2: With hydrogenchloride In tetrahydrofuran; water Product distribution / selectivity; | 100% |
| Stage #1: 4-chloro-1-methyl-6-oxo-1,6-dihydro-pyridazine-3-carboxylic acid; 2-fluro-4-iodoaniline With lithium hexamethyldisilazane In tetrahydrofuran at 20℃; for 4.08333h; Stage #2: With hydrogenchloride In tetrahydrofuran; water Product distribution / selectivity; | 100% |
| Stage #1: 4-chloro-1-methyl-6-oxo-1,6-dihydro-pyridazine-3-carboxylic acid; 2-fluro-4-iodoaniline With lithium hexamethyldisilazane In tetrahydrofuran at 20℃; for 4.08333h; Stage #2: With hydrogenchloride In tetrahydrofuran; water Product distribution / selectivity; | 100% |
-
-
463-71-8
thiophosgene

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
945530-32-5
2-fluoro-4-iodo-1-isothiocyanatobenzene

| Conditions | Yield |
|---|---|
| In chloroform; water at 20℃; for 16h; | 100% |
| In chloroform; water at 20℃; for 16h; | 100% |
| In dichloromethane; water at 20℃; | 96.8% |

| Conditions | Yield |
|---|---|
| With acetic anhydride In tetrahydrofuran; toluene at 20℃; | 100% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
140-90-9
sodium O-ethyl dithiocarbonate

-
-
54420-94-9
6-iodobenzo[d]thiazole-2-thiol

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 95℃; for 5h; | 100% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
61079-72-9
2,3,4,-trifluorobenzoic acid

-
-
391211-97-5
3,4-difluoro-2-(2-fluoro-4-iodo-phenylamino)-benzoic acid

| Conditions | Yield |
|---|---|
| With lithium amide In tetrahydrofuran | 99% |
| With lithium amide In N,N-dimethyl-formamide Inert atmosphere; | 85% |
| With lithium amide In tetrahydrofuran at 45 - 55℃; for 1.25 - 2.5h; Product distribution / selectivity; | 80% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
945559-14-8
1-azido-2-fluoro-4-iodobenzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-fluro-4-iodoaniline With trifluoroacetic acid; sodium nitrite at 0℃; for 0.5h; Stage #2: With sodium azide In water at 0℃; for 1h; | 99% |
| Stage #1: 2-fluro-4-iodoaniline With hydrogenchloride; sodium nitrite In water at -5℃; for 0.5h; Stage #2: With sodium azide In water at -5℃; for 1h; | 97% |

| Conditions | Yield |
|---|---|
| With N,N`-dimethylethylenediamine; copper (I) trifluoromethane sulfonate benzene In dimethyl sulfoxide at 120℃; for 16h; | 99% |
| copper(I) trifluoromethanesulfonate benzene complex; N,N`-dimethylethylenediamine In dimethyl sulfoxide at 120℃; for 16h; | 99% |
| With N,N`-dimethylethylenediamine; copper(I) trifluoromethanesulfonate benzene In dimethyl sulfoxide at 120℃; for 16h; | 99% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
4023-34-1
cyclopropanecarboxylic acid chloride

-
-
1061683-60-0
N-(2-fluoro-4-iodophenyl)cyclopropanecarboxamide

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 0 - 20℃; | 99% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
530-62-1
1,1'-carbonyldiimidazole

-
-
871700-48-0
1-(2-fluoro-4-iodophenyl)urea

| Conditions | Yield |
|---|---|
| Stage #1: 2-fluro-4-iodoaniline; 1,1'-carbonyldiimidazole With triethylamine In chloroform at 0 - 20℃; for 4.25h; Stage #2: With ammonia In chloroform; water at 0 - 20℃; for 1.5h; | 98.8% |
| With triethylamine In chloroform at 0 - 30℃; for 4h; Inert atmosphere; | |
| With triethylamine In chloroform at 0 - 20℃; for 4.25h; |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
110-13-4
2,5-hexanedione

-
-
217314-30-2
1-(2-fluoro-4-iodophenyl)-2,5-dimethyl-1H-pyrrole

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid In toluene for 1h; Heating / reflux; | 98% |
| With toluene-4-sulfonic acid In toluene at 110℃; for 1h; | 49% |
| With p-toluenesulfonic acid monohydrate In toluene at 110℃; for 1h; Dean-Stark; | 67.5 g |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
124-63-0
methanesulfonyl chloride

-
-
143937-74-0
2-fluoro-4-iodo-1-methanesulfonylaminobenzene

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 2h; | 98% |
| In pyridine at 20℃; for 2h; | 98% |
| With pyridine at 20℃; for 2h; | 95% |
-
-
29632-74-4
2-fluro-4-iodoaniline

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite; boron trifluoride diethyl etherate In tetrahydrofuran; diethyl ether at -10 - 0℃; | 98% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 0 - 60℃; for 6h; | 97.6% |

| Conditions | Yield |
|---|---|
| Stage #1: formic acid; 2-fluro-4-iodoaniline With acetic anhydride In tetrahydrofuran; toluene at 20℃; Stage #2: With hydrogenchloride In tetrahydrofuran; water; ethyl acetate; toluene | 97% |
-
-
52449-98-6
tetrahydrofuran-2-carbonyl chloride

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
641612-64-8
tetrahydro-furan-2-carboxylic acid (2-fluoro-4-iodophenyl)amide

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; for 18h; | 96% |
| With dmap In dichloromethane |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
536-74-3
phenylacetylene

-
-
1241745-70-9
2-fluoro-4-phenylethynyl-phenylamine

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride In acetonitrile at 20℃; for 18h; Inert atmosphere; | 95% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
75264-92-5
tetramethylammonium trifluoromethylselenate(0)

-
-
15946-31-3
1-amino-2-fluoro-4-[(trifluoromethyl)seleno]benzene

| Conditions | Yield |
|---|---|
| With palladium(l) tri-tert-butylphosphine iodide dimer In toluene at 40℃; for 24h; Glovebox; Inert atmosphere; | 95% |
-
-
109-11-5
morpholine-3-one

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
742073-22-9
4-(4-amino-3-fluoro-phenyl)-morpholin-3-one

| Conditions | Yield |
|---|---|
| With potassium phosphate; copper(l) iodide; N,N`-dimethylethylenediamine In toluene at 160℃; under 4500.45 - 5250.53 Torr; for 2h; Ullmann-Goldberg Substitution; Microwave irradiation; | 94% |
| With potassium phosphate; copper(l) iodide; N,N`-dimethylethylenediamine In 1,4-dioxane Reflux; Inert atmosphere; | 74% |
| With potassium phosphate; copper(l) iodide; N,N`-dimethylethylenediamine In 1,4-dioxane Reflux; Inert atmosphere; | 74% |
| With potassium carbonate; N,N`-dimethylethylenediamine; copper(l) iodide In toluene at 140℃; for 2h; microwave; | 33% |
| With copper(l) iodide; N,N`-dimethylethylenediamine In 1,4-dioxane at 120℃; for 16h; |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
393-53-3
3-fluoroisonicotinic acid

-
-
885588-03-4
3-((2-fluoro-4-iodophenyl)amino)isonicotinic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-fluro-4-iodoaniline With lithium hexamethyldisilazane In tetrahydrofuran at -55℃; Inert atmosphere; Stage #2: 3-fluoroisonicotinic acid In tetrahydrofuran at 20℃; for 96h; Stage #3: With hydrogenchloride In water pH=2; | 94% |
| Stage #1: 2-fluro-4-iodoaniline; 3-fluoroisonicotinic acid With lithium hexamethyldisilazane In tetrahydrofuran at -78 - 20℃; Stage #2: With ammonium chloride In tetrahydrofuran for 1h; Product distribution / selectivity; | 33% |
| Stage #1: 2-fluro-4-iodoaniline With lithium hexamethyldisilazane In tetrahydrofuran at -67 - -59℃; for 2.5h; Inert atmosphere; Stage #2: 3-fluoroisonicotinic acid In tetrahydrofuran for 96h; |
-
-
848852-32-4
2,3,4-trifluoro-5-vinylbenzoic acid

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
848852-33-5
3,4-difluoro-2-(2-fluoro-4-iodophenylamino)-5-vinylbenzoic acid

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -78 - 20℃; | 94% |
| With lithium hexamethyldisilazane In tetrahydrofuran at -78 - 20℃; for 5.75h; Inert atmosphere; | 94% |
| With lithium diisopropyl amide In tetrahydrofuran at -78 - 20℃; | 35% |
-
-
5963-77-9
pent-2-ynoic acid

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
1276538-16-9
4-(4-ethyl-1H-1,2,3-triazol-1-yl)-2-fluoroaniline

| Conditions | Yield |
|---|---|
| With sodium azide; copper(ll) sulfate pentahydrate; sodium 2-(1,2-dihydroxyethyl)-4-hydroxy-5-oxo-2,5-dihydro-furan-3-olate; potassium carbonate; L-proline In water; dimethyl sulfoxide at 65℃; | 94% |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
693-13-0
diisopropyl-carbodiimide

-
-
1436412-58-6
2-(2-fluoro-4-iodophenyl)-1,3-diisopropylguanidine

| Conditions | Yield |
|---|---|
| at 130℃; for 15h; Inert atmosphere; | 94% |
| With dichloro bis(acetonitrile) palladium(II) In toluene at 130℃; under 1500.15 Torr; for 16h; Inert atmosphere; | 72% |
-
-
288-13-1
NH-pyrazole

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
1147557-79-6
2-fluoro-4-(1H-pyrazol-1-yl)aniline

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 8-quinolinol; potassium carbonate In dimethylsulfoxide-d6 at 120℃; for 16h; Inert atmosphere; | 94% |
| With copper(l) iodide; caesium carbonate; trans-1,2-cyclohexanediamine In 1,2-dimethoxyethane at 25 - 86℃; for 72.3333h; Inert atmosphere; | 650 g |

| Conditions | Yield |
|---|---|
| With 2,6-di-tert-butyl-4-methyl-phenol; tetrakis(triphenylphosphine) palladium(0) In toluene at 100℃; for 1h; | 93% |
| tetrakis(triphenylphosphine) palladium(0); 2,6-di-tert-butyl-4-methyl-phenol In toluene at 100℃; for 1h; | 93% |
-
-
1227054-00-3
(R)-5-chloro-3-((2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-6-fluoro-8-methylpyrido[2,3-d]pyrimidine-4,7-(3H,8H)-dione

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
1227054-01-4
(R)-3-((2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-6-fluoro-5-(2-fluoro-4-iodophenylamino)-8-methylpyrido-[2,3-d]pyrimidine-4,7(3H,8H)-dione

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -5 - 20℃; Large scale; | 93% |
| With 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene; sodium t-butanolate; palladium diacetate In 1,4-dioxane at 85℃; for 2h; Product distribution / selectivity; De-gassed solvent; |
-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
530-62-1
1,1'-carbonyldiimidazole

-
-
74-89-5
methylamine

-
-
871700-37-7
N-(2-fluoro-4-iodophenyl)-N'-methylurea

| Conditions | Yield |
|---|---|
| Stage #1: 2-fluro-4-iodoaniline; 1,1'-carbonyldiimidazole With triethylamine In N,N-dimethyl-formamide at 0 - 20℃; Inert atmosphere; Stage #2: methylamine In water; N,N-dimethyl-formamide at 0 - 20℃; | 93% |
| Stage #1: 2-fluro-4-iodoaniline; 1,1'-carbonyldiimidazole With triethylamine In N,N-dimethyl-formamide at 0 - 20℃; for 16h; Inert atmosphere; Stage #2: methylamine In water; N,N-dimethyl-formamide at 0 - 20℃; for 1h; | 93% |
| Stage #1: 2-fluro-4-iodoaniline; 1,1'-carbonyldiimidazole With triethylamine In N,N-dimethyl-formamide at 0 - 20℃; Stage #2: methylamine In methanol; N,N-dimethyl-formamide at 0 - 20℃; | 92% |

-
-
29632-74-4
2-fluro-4-iodoaniline

-
-
108-24-7
acetic anhydride

-
-
97760-94-6
N-(2-fluoro-4-iodo-phenyl)-acetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 25℃; for 2.16667h; | 92% |
| In tetrahydrofuran at 0 - 25℃; for 2.16667h; | 92% |
| In tetrahydrofuran at 0 - 25℃; for 2.16667h; | 92% |
-
-
2637-34-5
pyridine-2-thione

-
-
29632-74-4
2-fluro-4-iodoaniline

| Conditions | Yield |
|---|---|
| With 8-quinolinol; copper(l) iodide In dimethyl sulfoxide at 130℃; | 92% |
-
-
1003-56-1
3-Methyl-1H-pyridin-2-one

-
-
29632-74-4
2-fluro-4-iodoaniline

| Conditions | Yield |
|---|---|
| With copper(l) iodide; (R,R)-N,N'-dimethyl-1,2-diaminocyclohexane; potassium carbonate In tert-Amyl alcohol at 100℃; for 5h; Inert atmosphere; Glovebox; | 92% |
| With 8-quinolinol; copper(l) iodide In dimethyl sulfoxide at 130℃; | 78% |
2-Fluoro-4-iodoaniline Chemical Properties
The Molecular Weight of 2-Fluoro-4-iodoaniline(29632-74-4) : 237.01
Molecular Structure :
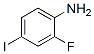
Melting point: 55-57 °C(lit.)
Flash point: >230 °F
WATER Solubility: insoluble
Sensitive: Light Sensitive
Appearaance: light brown to brown Solid
Synonyms: 2-FLUORO-4-IODOBENZENAMINE;2-FLUORO-4-IODO-PHENYLAMINE;2-Fluoro-4-idioaniline;2-Fluoro-4-iodoaniline 98%;2-FLUORO-4-IODOANILINE;2-Fluoro-4-iodoaniline98%;
2-Fluoro-4-iodoaniline Safety Profile
 Xi,Xn ,
Xi,Xn ,  T
TThe Risk Statements information of 2-Fluoro-4-iodoaniline(29632-74-4):
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements information of 2-Fluoro-4-iodoaniline(29632-74-4):
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37/39: Wear suitable gloves and eye/face protection
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
RIDADR: PG3
WGK Germany: 3
Hazard Note: Toxic
Related Products
- 2-Fluoro-4-iodoaniline
- 29635-10-7
- 296-35-5
- 29636-65-5
- 2963-66-8
- 29636-87-1
- 2963-69-1
- 29636-96-2
- 29639-68-7
- 29640-92-4
- 296-41-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View