-
Name
8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one
- EINECS 700-257-5
- CAS No. 31251-41-9
- Article Data13
- CAS DataBase
- Density 1.314 g/cm3
- Solubility
- Melting Point 90-92 °C
- Formula C14H10ClNO
- Boiling Point 433.614 °C at 760 mmHg
- Molecular Weight 243.692
- Flash Point 216.042 °C
- Transport Information
- Appearance Yellow solid
- Safety
- Risk Codes
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 8-Chloro-10,11-dihydro-4-aza-5H-dibenzo[a,d]cycloheptan-5-one;
- PSA 29.96000
- LogP 3.06460
Synthetic route
-
-
31255-57-9
3-[2-(3-chlorophenyl)ethyl]-2-pyridine-carbonitrile

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Stage #1: 3-[2-(3-chlorophenyl)ethyl]-2-pyridine-carbonitrile With trifluorormethanesulfonic acid at 60℃; for 1h; Stage #2: With hydrogenchloride In water at 20℃; for 1h; Heating / reflux; | 94% |
| With PPA at 200℃; | 45% |
| With PPA Heating; |
-
-
251353-79-4
3-[2-(5-chloro-2-iodo-phenyl)-ethyl]-pyridine-2-carboxylic acid methyl-phenyl-amide

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; cyclohexane at -78℃; for 0.166667h; | 91% |
| With n-butyllithium In tetrahydrofuran; cyclohexane at -78℃; for 0.166667h; | 78% |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With water at 0 - 35℃; for 25 - 35h; | 76.4% |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In carbon disulfide | 40% |
-
-
15396-37-9
2-iodoyl-3,5-dichlorobenzoic acid

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 121 g / (MeO)3B; BH3*Me2S / tetrahydrofuran / 18 h / 20 °C 2.1: PPh3; Br2 / acetonitrile / 1 h / 20 °C 3.1: LDA; t-BuOMe / various solvent(s); tetrahydrofuran / -30 - 0 °C 4.1: NaH / dimethylformamide / 1 h / 0 °C 4.2: 10 g / dimethylformamide / 0.25 h / 0 °C 5.1: 91 percent / n-BuLi / tetrahydrofuran; cyclohexane / 0.17 h / -78 °C View Scheme |
-
-
82386-90-1
5-chloro-2-iodobenzyl alcohol

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: PPh3; Br2 / acetonitrile / 1 h / 20 °C 2.1: LDA; t-BuOMe / various solvent(s); tetrahydrofuran / -30 - 0 °C 3.1: NaH / dimethylformamide / 1 h / 0 °C 3.2: 10 g / dimethylformamide / 0.25 h / 0 °C 4.1: 91 percent / n-BuLi / tetrahydrofuran; cyclohexane / 0.17 h / -78 °C View Scheme |
-
-
82386-91-2
2-(bromomethyl)-4-chloro-1-iodobenzene

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: LDA; t-BuOMe / various solvent(s); tetrahydrofuran / -30 - 0 °C 2.1: NaH / dimethylformamide / 1 h / 0 °C 2.2: 10 g / dimethylformamide / 0.25 h / 0 °C 3.1: 91 percent / n-BuLi / tetrahydrofuran; cyclohexane / 0.17 h / -78 °C View Scheme |
-
-
251353-78-3
3-[2-(5-chloro-2-iodo-phenyl)-ethyl]-pyridine-2-carboxylic acid phenylamide

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: NaH / dimethylformamide / 1 h / 0 °C 1.2: 10 g / dimethylformamide / 0.25 h / 0 °C 2.1: 91 percent / n-BuLi / tetrahydrofuran; cyclohexane / 0.17 h / -78 °C View Scheme |
-
-
32998-95-1
N-tert-butyl-3-methylpyridine-2-carboxamide

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) n-BuLi / 1.) THF, -40 deg C 2: POCl3 / Heating 3: PPA / Heating View Scheme |
-
-
107285-30-3
3-<2-(3-chlorophenyl)ethyl>-N-(1,1-dimethylethyl)-2-pyridinecarboxamide

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: POCl3 / Heating 2: PPA / Heating View Scheme |
-
-
620-20-2
1-Chloro-3-chloromethyl-benzene

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) n-BuLi / 1.) THF, -40 deg C 2: POCl3 / Heating 3: PPA / Heating View Scheme |
-
-
255861-49-5
3-[2-(3-Chlorophenyl)ethyl]-pyridine-2-carboxylic acid hydrochloride

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Stage #1: 3-[2-(3-Chlorophenyl)ethyl]-pyridine-2-carboxylic acid hydrochloride With thionyl chloride at 30 - 60℃; Stage #2: aluminum (III) chloride In 1,2-dichloro-ethane at -5 - 20℃; |
-
-
271252-79-0
N-phenyl-3-[2-(3-chlorophenyl)ethyl]-2-pyridine carboxamide

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
7440-44-0
pyrographite

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| aluminium trichloride In hexane; dichloromethane; toluene | 44.2 g (61%) |

-
-
271252-79-0
N-phenyl-3-[2-(3-chlorophenyl)ethyl]-2-pyridine carboxamide

-
-
7440-44-0
pyrographite

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With trichlorophosphate In hexane; water; chlorobenzene | 44.1 g (50.8%) |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 5%-palladium/activated carbon; hydrogen / ethyl acetate / 4 h / 760.05 Torr 2: sodium hydroxide; water / ethanol / 48 h 3: thionyl chloride 4: aluminum (III) chloride / carbon disulfide View Scheme |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium hydroxide; water / ethanol / 48 h 2: thionyl chloride 3: aluminum (III) chloride / carbon disulfide View Scheme |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: thionyl chloride 2: aluminum (III) chloride / carbon disulfide View Scheme |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 5%-palladium/activated carbon; hydrogen / ethyl acetate / 48 h / 760.05 Torr 2: sodium hydroxide; water / ethanol / 5 h / Reflux 3: thionyl chloride 4: aluminum (III) chloride / carbon disulfide View Scheme |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium hydroxide; water / ethanol / 5 h / Reflux 2: thionyl chloride 3: aluminum (III) chloride / carbon disulfide View Scheme |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In tetrahydrofuran at 80 - 90℃; pH=10-11; |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
133330-72-0
8-Chloro-6,11-Dihydro-11-Hydroxy-5H-Benzo[5,6]-Cyclohepta[1,2-b]Pyridine

| Conditions | Yield |
|---|---|
| Stage #1: 8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one With sodium tetrahydroborate In methanol at 0 - 20℃; for 1h; Stage #2: With hydrogenchloride In methanol; water | 100% |
| With sodium tetrahydroborate In methanol at 10 - 20℃; for 2.75h; | 96.9% |
| With sodium tetrahydroborate In methanol a) 0 deg C, 2.5 h, b) from 0 deg C to 25 deg C, 1 h; |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
63463-36-5
(1-methyl-4-piperidyl)magnesium chloride

-
-
38089-93-9
8-chloro-6,11-dihydro-11-(1-methyl-4-piperidinyl)-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Reflux; | 95% |
| With (trimethylsilyl)methylmagnesium chloride; lithium chloride; zinc(II) chloride In tetrahydrofuran; diethyl ether at 0℃; for 2h; Inert atmosphere; | 90% |
| In tetrahydrofuran at -95 - -15℃; for 2 - 3h; | 71.1% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
3964-73-6
5,6-Dihydro-11H-benzo<5,6>cyclohepta<1,2-b>pyridin-11-one

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen; triethylamine In tetrahydrofuran at 20℃; for 24h; | 89% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
216870-24-5
diethyl N-ethoxycarbonylpiperidine-4-phosphonate

| Conditions | Yield |
|---|---|
| Stage #1: diethyl N-ethoxycarbonylpiperidine-4-phosphonate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -25 - -5℃; for 2h; Stage #2: 8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one In tetrahydrofuran; hexane at -25 - -20℃; for 2h; | 82% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
931-20-4
1-methylpiperidin-2-one

-
-
212390-12-0
3-(8-Chloro-11-hydroxy-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl)-1-methyl-piperidin-2-one

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane 1.) -78 deg C, 30 min; -78 deg C to -5 deg C, 60 min, 2.) -78 deg C, 1 h; -78 deg C to -10 deg C, 1 h; | 76% |
-
-
127406-56-8
2-(4-formylphenyl)pyridine

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 10h; Reflux; | 75% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
1571-08-0
methyl 4-formylbenzoate

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 10h; Reflux; | 75% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
1208-88-4
4-phenylsulfanyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 8h; Reflux; | 74% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
4707-71-5
Phenanthrene-9-carboxaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 7h; Reflux; | 74% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
122-52-1
triethyl phosphite

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene at 20℃; for 24h; | 73.8% |
-
-
5570-77-4
4-chloro-1-methylpiperidine

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
38089-93-9
8-chloro-6,11-dihydro-11-(1-methyl-4-piperidinyl)-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ol

| Conditions | Yield |
|---|---|
| Stage #1: 4-chloro-1-methylpiperidine With magnesium; ethylene dibromide In tetrahydrofuran at 20 - 48℃; Stage #2: 8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one In tetrahydrofuran at -80 - -70℃; for 2 - 3h; Product distribution / selectivity; | 73.6% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
105-07-7
4-cyanobenzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 6h; Reflux; | 73% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
67713-23-9
2,6-dibromobenzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 6h; Reflux; | 73% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
32555-96-7
4-styrylbenzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 8h; Reflux; | 72% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
30084-90-3
9H-fluorene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 6h; Reflux; | 71% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
110677-45-7
N-(4-formylphenyl)carbazole

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 10h; Reflux; | 70% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
5398-77-6
para-methanesulfonylbenzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 10h; Reflux; | 70% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
69155-76-6
6-fluorochromone-3-carboxaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 8h; Reflux; | 70% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
83-38-5
2,6-dichlorobenzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 6h; Reflux; | 69% |
-
-
4649-09-6
1H-pyrrolo[2,3-b]pyridin-3-carbaldehyde

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 8h; Reflux; | 68% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
3446-89-7
4-(Methylthio)benzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 7h; Reflux; | 68% |
-
-
27395-93-3
4-benzooxazol-2-yl-benzaldehyde

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 7h; Reflux; | 68% |
-
-
3779-27-9
2,2'-bithiophene-5-carboxaldehyde

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 10h; Reflux; | 67% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
4181-05-9
4-(diphenylamino)benzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 10h; Reflux; | 66% |
-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

-
-
121-45-9
phosphorous acid trimethyl ester

| Conditions | Yield |
|---|---|
| With phosphoric acid In chloroform at 20℃; for 12h; | 65% |

-
-
31251-41-9
8-chloro-5,6-dihydro-11H-Benzo[5,6]cyclohepta[1,2-b]pyridin-11-one

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 7h; Reflux; | 65% |
8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one Chemical Properties
Molecular structure of 8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one (CAS NO.31251-41-9) is:
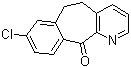
Product Name: 8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one
CAS Registry Number: 31251-41-9
Systematic Name: 8-chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one
Empirical Formula: C14H10ClNO
Molecular Weight: 243.6883
Melting Point: 90-92°C
Index of Refraction: 1.632
Molar Refractivity: 66.136 cm3
Molar Volume: 185.481 cm3
Surface Tension: 53.838 dyne/cm
Density: 1.314 g/cm3
Flash Point: 216.042 °C
Enthalpy of Vaporization: 68.959 kJ/mol
Boiling Point: 433.614 °C at 760 mmHg
Product Categories: Aromatics Compounds;Aromatics;Heterocycles;Intermediates
8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one Uses
8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one (CAS NO.31251-41-9) can be used as loratadine intermediate.
8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one Safety Profile
Hazard Codes:  Xi
Xi
8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one Specification
8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one , its cas register number is 31251-41-9. It also can be called 8-Chloro-10,11-Dihydro-4-Aza-5h-Dibenzo-(A,D)Cycloheptan-5-One ; 8-Chloro-5,6-Dihydro-11h-Benzo-(5,6)-Cyclohepta-(1,2-B)-Pyrimidine-11-On ; 8-Chloro-10,11-dihydro-4-aza-5 ; 4-Aza-8-chloro-10,11-dihydro-5H-benzo[a,d]cyclo-5-heptanone .It is a yellow solid.
Related Products
- 8-Chloro-5,10-dihydrodibenzo[b,e][1,4]diazepin-11-one
- 8-Chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one
- 8-Chloro-5-methylquinoline
- 8-Chloro-5-nitroquinoline
- 312517-86-5
- 31252-42-3
- 312-52-7
- 31253-34-6
- 31255-57-9
- 3125-63-1
- 3125-64-2
- 3125-66-4
- 3125-71-1
- 3125-73-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View