-
Name
N-VINYLACETAMIDE 97
- EINECS 225-989-8
- CAS No. 5202-78-8
- Article Data24
- CAS DataBase
- Density 0.89g/cm3
- Solubility Soluble in water
- Melting Point 51-54 °C(lit.)
- Formula C4H7NO
- Boiling Point 207.7 °C at 760 mmHg
- Molecular Weight 85.1057
- Flash Point 103.6 °C
- Transport Information
- Appearance
- Safety 26-36
- Risk Codes 22-36/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Acetamide,N-vinyl- (6CI,7CI,8CI);N-Vinylacetamide;NVA;Vinylacetamide;N-Ethenylacetamide;
- PSA 29.10000
- LogP 0.65690
Synthetic route

| Conditions | Yield |
|---|---|
| With succinic acid anhydride at 40 - 75℃; for 15h; Time; Inert atmosphere; | 82.2% |
-
-
55204-51-8
N-(1-methoxyethyl)acetamide

-
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| at 550℃; | 70% |
| at 440℃; under 100 Torr; |
-
-
69773-71-3
2-methyl-4-methyl-4H-oxazolin-5-one

-
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| at 550℃; under 0.1 - 0.2 Torr; | 32% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hexane | |
| With sodium hydroxide; hexane | |
| With sodium hydroxide; hexane |

| Conditions | Yield |
|---|---|
| at 220℃; |
-
-
19861-71-3
N-(1-cyanoethyl)acetamide

-
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| at 450℃; under 13 Torr; |

| Conditions | Yield |
|---|---|
| In diphenylether at 250℃; Rate constant; |
-
-
60-35-5
acetamide

-
-
542-10-9
ethylidene diacetate

-
A
-
5335-91-1
1,1-bis-acetylamino-ethane

-
B
-
625-77-4
N-acetylacetamide

-
C
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 50℃; for 3h; | A 12.1 % Chromat. B 17.2 % Chromat. C 23.8 % Chromat. |

-
-
60-35-5
acetamide

-
-
542-10-9
ethylidene diacetate

-
A
-
5335-91-1
1,1-bis-acetylamino-ethane

-
B
-
625-77-4
N-acetylacetamide

-
C
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 50℃; for 3h; Product distribution; reactions in presence of further bases or Lewis acids; | A 12.1 % Chromat. B 17.2 % Chromat. C 23.8 % Chromat. D 1.6 % Chromat. |


-
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| at 330 - 350℃; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium hydrogensulfite In 1,2-dichloro-ethane at 120℃; for 12h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With sodium dodecyl-sulfate In water at 20℃; for 1h; pH=1; Reagent/catalyst; Solvent; Temperature; Time; Green chemistry; diastereospecific reaction; | 99% |
| With cerium(IV) sulphate In acetonitrile at 20℃; under 760.051 Torr; for 6h; Catalytic behavior; Temperature; diastereoselective reaction; | 89% |
-
-
5202-78-8
acetylaminoethylene

-
-
224449-33-6
4.-brorno-thiophene-3-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With palladium diacetate; triethylamine; tris-(o-tolyl)phosphine In N,N-dimethyl-formamide at 80℃; for 12h; Inert atmosphere; | 99% |
-
-
5202-78-8
acetylaminoethylene

-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
1105508-27-7
N-ethenyl-N-(3-phenylpropyl)acetamide

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; (R)-3,3'-bis(2,4,6-triisopropylphenyl)binol phosphoric acid; [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; water In toluene at 25℃; for 15h; Schlenk technique; Inert atmosphere; Sealed tube; Irradiation; enantioselective reaction; | 96% |

-
-
5202-78-8
acetylaminoethylene

-
-
3717-15-5
p-methylbenzaldehyde oxime

-
-
1140531-49-2
(E)-4-methylbenzaldehyde O-1-(N-acetyl)aminoethyloxime

| Conditions | Yield |
|---|---|
| With 2,6-di-tert-butyl-pyridine; tris(4-bromophenyl)aminium hexachloroantimonate In dichloromethane at 20℃; for 0.166667h; | 95% |

| Conditions | Yield |
|---|---|
| With GLUTATHIONE; 2,2’-azobis[2-(2-imidazolin-2-yl)propane] dihydrochloride for 1h; pH=4; Product distribution / selectivity; Acetate buffer; UV-irradiation; | 95% |
| With GLUTATHIONE for 1h; pH=4; aq. acetate buffer; UV-irradiation; |

| Conditions | Yield |
|---|---|
| With nickel(II) triflate; potassium carbonate; 5,5′-bis(trifluoromethyl)-2,2′-bipyridine In 1,4-dioxane at 80℃; for 8h; Inert atmosphere; Schlenk technique; | 92% |

-
-
536-57-2
p-toluene sulfinic acid

-
-
5202-78-8
acetylaminoethylene

-
-
1578252-28-4
N-[1-(p-toluenesulfonyl)ethyl]acetamide

| Conditions | Yield |
|---|---|
| With photoredox catalyst Ni/TiO2 In acetonitrile at 20℃; for 3.5h; Molecular sieve; Irradiation; | 92% |
-
-
31913-41-4
6-methyl-4-tosyloxy-2(2H)-pyranone

-
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; N-ethyl-N,N-diisopropylamine; tris(dibenzylideneacetone)dipalladium (0) In 1,4-dioxane at 85℃; for 4h; Heck reaction; | 91% |
-
-
869938-32-9
4-methyl-N-(2-methylallyl)-N-(3-phenylprop-2-yn-1-yl)benzenesulfonamide

-
-
5202-78-8
acetylaminoethylene

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; hydrogen; (R)-segphos In dichloromethane at 20℃; for 72h; Schlenk technique; stereoselective reaction; | 91% |
-
-
5202-78-8
acetylaminoethylene

-
-
667-27-6
Ethyl bromodifluoroacetate

-
-
1765-93-1
4-fluoroboronic acid

-
A
-
2262-04-6
ethyl 2,2-difluoro-3-(4-fluorophenyl)-3-oxopropanoate

| Conditions | Yield |
|---|---|
| With carbon monoxide; water; C19H16ClFN2NiO3; potassium carbonate In 1,4-dioxane at 50℃; under 760.051 Torr; for 18h; Time; Schlenk technique; Sealed tube; | A 38 %Spectr. B 91% |


| Conditions | Yield |
|---|---|
| Stage #1: 9H-thioxanthen-9-ol; acetylaminoethylene With acetic acid at 20℃; for 1h; Stage #2: With hydrogenchloride for 1h; | 90% |
-
-
5202-78-8
acetylaminoethylene

-
-
55204-51-8
N-(1-methoxyethyl)acetamide

| Conditions | Yield |
|---|---|
| With acetamide | 90% |
| In methanol |

| Conditions | Yield |
|---|---|
| With GLUTATHIONE; 2,2’-azobis[2-(2-imidazolin-2-yl)propane] dihydrochloride for 2h; pH=4; Acetate buffer; UV-irradiation; | 90% |

| Conditions | Yield |
|---|---|
| With dimethylsulfide; GLUTATHIONE; 2,2’-azobis[2-(2-imidazolin-2-yl)propane] dihydrochloride for 2h; pH=4; Product distribution / selectivity; Acetate buffer; UV-irradiation; | 90% |

| Conditions | Yield |
|---|---|
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate In acetonitrile at 20℃; for 2h; Catalytic behavior; Solvent; Irradiation; regioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate In acetonitrile at 20℃; for 18h; Irradiation; regioselective reaction; | 90% |
-
-
5202-78-8
acetylaminoethylene

-
-
109613-00-5
4-acetophenyl triflate

-
-
823790-71-2
N-acetyl-1-(4'-acetylphenyl)ethenamine

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; 1,1'-bis-(diphenylphosphino)ferrocene; tris(dibenzylideneacetone)dipalladium (0) In 1,4-dioxane at 85℃; for 1.75h; Heck coupling; | 89% |
| With 1,3-bis-(diphenylphosphino)propane; triethylamine; palladium diacetate In N,N-dimethyl-formamide at 100℃; Heck reaction; | 40% |
-
-
5202-78-8
acetylaminoethylene

-
-
5798-75-4
Ethyl 4-bromobenzoate

-
-
1028431-82-4
ethyl 4-(1-acetamidovinyl)benzoate

| Conditions | Yield |
|---|---|
| With 1,3-bis-(diphenylphosphino)propane; triethylamine; palladium diacetate In isopropyl alcohol for 12h; Heck arylation; Heating; | 89% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 18h; Green chemistry; regioselective reaction; | 88% |
| With 10-methyl-9-(2,4,6-trimethylphenyl) acridinium tetrafluoroborate In acetonitrile at 20℃; for 6h; Irradiation; Sealed tube; | 82% |
| With 2,2’-azobis[2-(2-imidazolin-2-yl)propane] dihydrochloride at 20℃; for 0.5h; pH=4; Acetate buffer; UV-irradiation; |

| Conditions | Yield |
|---|---|
| With potassium phosphate; (R)-3,3'-bis(2,4,6-triisopropylphenyl)binol phosphoric acid; [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; water In toluene at 25℃; for 15h; Reagent/catalyst; Schlenk technique; Inert atmosphere; Sealed tube; Irradiation; enantioselective reaction; | 88% |

Acetamide, N-ethenyl- Specification
The Acetamide, N-ethenyl-, with CAS registry number 5202-78-8, has the systematic name of N-ethenylacetamide. Besides this, it is also called N-Vinylacetamide 97. And the chemical formula of this chemical is C4H7NO. What's more, its EINECS is 225-989-8.
Physical properties of Acetamide, N-ethenyl-: (1)ACD/LogP: 0.00; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0; (4)ACD/LogD (pH 7.4): 0; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 23.81; (8)ACD/KOC (pH 7.4): 23.81; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 20.31 Å2; (13)Index of Refraction: 1.414; (14)Molar Refractivity: 23.93 cm3; (15)Molar Volume: 95.5 cm3; (16)Polarizability: 9.48×10-24cm3; (17)Surface Tension: 24.7 dyne/cm; (18)Density: 0.89 g/cm3; (19)Flash Point: 103.6 °C; (20)Enthalpy of Vaporization: 44.4 kJ/mol; (21)Boiling Point: 207.7 °C at 760 mmHg; (22)Vapour Pressure: 0.222 mmHg at 25°C.
Preparation: this chemical can be prepared by N-(1-methoxy-ethyl)-acetamide. This reaction temperature is 550 ℃. The yield is about 70%.
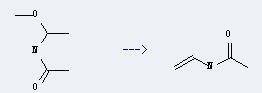
When you are using this chemical, please be cautious about it as the following:
The Acetamide, N-ethenyl- irritates to eyes, respiratory system and skin. And this chemical is harmful if swallowed. When use it, wear suitable protective clothing, gloves and eye/face protection. If contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(N\C=C)C
(2)InChI: InChI=1/C4H7NO/c1-3-5-4(2)6/h3H,1H2,2H3,(H,5,6)
(3)InChIKey: RQAKESSLMFZVMC-UHFFFAOYAE
(4)Std. InChI: InChI=1S/C4H7NO/c1-3-5-4(2)6/h3H,1H2,2H3,(H,5,6)
(5)Std. InChIKey: RQAKESSLMFZVMC-UHFFFAOYSA-N
Related Products
- Acetamide
- Acetamide, 2-(butylamino)-N-(1-phenoxy-2-propyl)-, hydrochloride
- Acetamide, 2-(diethylamino)-N-(1,3-dimethyl-4-(o-fluorobenzoyl)-5-pyrazolyl)-, monohydrochloride
- Acetamide, 2-(dipropylamino)-
- Acetamide, 2-(hydroxyimino)-N-phenyl-
- Acetamide, 2,2,2-trifluoro-N-(2-iodoethyl)-
- Acetamide, 2,2,2-trifluoro-N-(3R)-3-pyrrolidinyl-,hydrochloride (1:1)
- Acetamide, 2,2,2-trifluoro-N-(5-hydroxypentyl)-
- Acetamide, 2,2-dichloro-N-(2-ethylhexyl)-
- Acetamide, 2,2-dimethoxy-N,N-dimethyl-
- 520-28-5
- 5202-85-7
- 5202-86-8
- 5202-89-1
- 5203-01-0
- 52031-09-1
- 5203-14-5
- 520-32-1
- 52032-20-9
- 5203-31-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View