Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:637-59-2
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:637-59-2
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
CAS No.637-59-2 / 1-Bromo-3-phenylpropane Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,L
Cas:637-59-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:637-59-2
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHangzhou Think Chemical Co. Ltd
Product: 1-Bromo-3-phenylpropane Chemical name:1-Bromo-3-phenylpropane Batch NO: 20210605 Analysis Date: 2021-06-05 Expiry Date: 2023-06-04 HANGZHOU THINK CHEMICAL CO., LTD. (THINKCHEM) is an integrative corporation of trade, rese
Cas:637-59-2
Min.Order:10 Kilogram
FOB Price: $90.0 / 100.0
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:637-59-2
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:637-59-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:637-59-2
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:637-59-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
1-Bromo-3-phenylpropane Chemical Properties Melting point -10°C (estimate) Boiling point 237-238 °C (lit.) density 1.31 g/mL at 25 °C (lit.) refractive index n20/D 1.546(lit.) Fp 215 °F storage temp. Store belo
Cas:637-59-2
Min.Order:1 Gram
FOB Price: $66.0
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:637-59-2
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryWin-Win chemical Co.Ltd
Stock products, own laboratoryAppearance:white to light yellow solid Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:637-59-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:637-59-2
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Hunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as
Hebei Sankai Chemical Technology Co., Ltd
1. Product advantages? High purity, all above 98.5%, no impurities after dissolution? We will test each batch to ensure quality? OEM and private brand services designed for free? Various cap colors available? We can also provide MT1 peptide powder2.
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
BOC Sciences
We are committed to providing our customers with the best products and services at the most competitive prices.Appearance:white crystalline powder Storage:Room temperature with sealed well Package:according to the clients requirement Application:Use
Changchun Artel lmport and Export trade company
Supply top quality products with a reasonable price Application:api
Hangzhou Fandachem Co.,Ltd
1-Bromo-3-phenylpropane cas 637-59-2Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transportation:by air, by sea, by express
Henan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Shanghai Chinqesen Biotechnology Co., Ltd.
Good Quality Package:1kg/bag Application:Medical or chemical Transportation:Air/Train/Sea Port:Shenzhen
Synthetic route

| Conditions | Yield |
|---|---|
| With polystyrene-supported triphenylphosphine dibromide In chloroform at 61℃; for 3h; | 100% |
| Stage #1: 3-Phenyl-1-propanol With diisopropyl-carbodiimide; copper(II) bis(trifluoromethanesulfonate) In tetrahydrofuran at 100℃; for 0.0833333h; microwave irradiation; Stage #2: With Acetyl bromide In tetrahydrofuran at 150℃; for 0.0833333h; microwave irradiation; Further stages.; | 98% |
| With allyl bromide; 1,1'-carbonyldiimidazole In acetonitrile for 1.5h; Heating; | 97% |

| Conditions | Yield |
|---|---|
| With 1-n-butyl-3-methylimidazolim bromide at 50℃; for 1h; Concentration; Temperature; Time; Inert atmosphere; Green chemistry; | 100% |
| With lithium bromide In [(2)H6]acetone at 20℃; for 32h; Inert atmosphere; | 95% |
| With potassium bromide In N,N-dimethyl-formamide at 20℃; for 12h; Product distribution / selectivity; | 94% |
-
-
69804-99-5
3-phenylpropanol methanesulfonate

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With 1-n-butyl-3-methylimidazolim bromide at 50℃; for 1h; Concentration; Temperature; Time; Inert atmosphere; Green chemistry; | 100% |
| With sodium bromide In N,N-dimethyl-formamide at 25 - 30℃; | 80% |
| With sodium tetrahydroborate; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; for 18h; |
-
-
866942-21-4
2-(3-Phenyl-propoxysulfonyl)-benzoic acid 1,4,7,10-tetraoxa-cyclododec-2-ylmethyl ester

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In acetone at 20℃; for 1.3h; Product distribution; Further Variations:; Reagents; Temperatures; reaction time; | 98% |
| With lithium bromide In [(2)H6]acetone at 20℃; for 1.3h; Inert atmosphere; | 95% |
-
-
866942-17-8
2-(3-phenyl-propoxysulfonyl)-benzoic acid 2-methoxy-ethyl ester

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In acetone for 0.3h; Product distribution; Further Variations:; Reagents; Temperatures; reaction time; Heating; | 97% |
| With lithium bromide In [(2)H6]acetone at 20℃; for 12h; Inert atmosphere; | 95% |
-
-
866942-18-9
3-phenylpropyl 2-(methoxyethoxyethylcarboxy)-1-benzosulfonate

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In acetone for 0.1h; Product distribution; Further Variations:; Reagents; Temperatures; reaction time; Heating; | 97% |
| With lithium bromide In acetone for 0.1h; Product distribution / selectivity; Heating / reflux; | 97% |
| With lithium bromide In [(2)H6]acetone at 20℃; for 5h; Inert atmosphere; | 95% |
-
-
866942-16-7
3-phenylpropyl 2-(methylcarboxy)-1-benzenesulfonate

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In acetone for 0.75h; Product distribution; Further Variations:; Reagents; reaction time; Heating; | 96% |
| With lithium bromide In acetone for 0.75h; Product distribution / selectivity; Heating / reflux; | 96% |
| With sodium bromide In acetone for 3h; Product distribution / selectivity; Heating / reflux; | 94% |
| With potassium bromide In acetone for 15h; Product distribution / selectivity; Heating / reflux; | 92% |

| Conditions | Yield |
|---|---|
| With indium(III) bromide; trimethylsilyl bromide; 1,1,3,3-Tetramethyldisiloxane In chloroform at 20 - 60℃; for 1.08333h; Reagent/catalyst; Solvent; Sealed tube; Inert atmosphere; | 96% |
| With trimethylsilyl bromide; 1,1,3,3-Tetramethyldisiloxane; indium tribromide In chloroform at 60℃; for 1h; Reagent/catalyst; Sealed tube; Inert atmosphere; | 96% |
| With indium(III) bromide; 1,1,3,3-Tetramethyldisiloxane; trimethylsilyl cyanide In chloroform at 60℃; Inert atmosphere; |
-
-
866942-19-0
2-(3-phenyl-propoxysulfonyl)-benzoic acid 2-[2-(2-methoxy-ethoxy)-ethoxy]-ethyl ester

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 3.3h; Inert atmosphere; | 95% |
| With sodium bromide In acetone for 3.6h; Product distribution; Further Variations:; Reagents; Temperatures; reaction time; Heating; | 94% |
-
-
866942-20-3
2-(3-phenyl-propoxysulfonyl)-benzoic acid 2-{2-[2-(2-methoxy-ethoxy)-ethoxy]-ethoxy}-ethyl ester

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With sodium bromide In acetone for 2.5h; Product distribution; Further Variations:; Reagents; Temperatures; reaction time; Heating; | 95% |
| With lithium bromide In [(2)H6]acetone at 20℃; for 2.3h; Inert atmosphere; | 95% |
-
-
14629-60-8
trimethyl(3-phenylpropoxy)silane

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With 4-aminophenyl diphenylphosphinite; N-Bromosuccinimide In dichloromethane for 1.5h; Heating; | 95% |
-
-
112471-51-9
2-(3-phenylpropoxy)tetrahydro-2H-pyran

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With 4-aminophenyl diphenylphosphinite; N-Bromosuccinimide In dichloromethane for 3h; Heating; | 95% |

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 25h; Inert atmosphere; | 95% |
| With lithium bromide In acetone at 20℃; for 4.5h; |

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 10h; Inert atmosphere; | 95% |
| With lithium bromide In acetone at 20℃; for 3.5h; |

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 4h; Inert atmosphere; | 95% |
| With lithium bromide In acetone at 20℃; for 1h; |

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 2.5h; Inert atmosphere; | 95% |
| With lithium bromide In acetone at 20℃; for 0.5h; |
-
-
1082742-89-9
3-phenylpropyl quinoline-8-sulfonate

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 4.5h; Inert atmosphere; | 95% |
| With titanium(IV) bromide In dichloromethane at 20℃; for 5.5h; Inert atmosphere; | 89% |
-
-
1174295-53-4
C20H24F2O6S

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 2.5h; Inert atmosphere; | 95% |
-
-
1174295-54-5
C22H28F2O7S

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 1.8h; Inert atmosphere; | 95% |
-
-
1174295-48-7
C24H34O6S

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 8.8h; Inert atmosphere; | 95% |
-
-
1174295-49-8
C26H38O7S

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 6.8h; Inert atmosphere; | 95% |
-
-
66950-73-0
3-phenylpropyl 1-trifluoromethanesulfonate

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With lithium bromide In [(2)H6]acetone at 20℃; for 2h; Inert atmosphere; | 95% |
-
-
122-97-4
3-Phenyl-1-propanol

-
-
1229444-43-2
4-(diphenylphosphino)-benzyltrimethylammonium bromide

-
A
-
1268610-56-5
Br(1-)*C22H25NOP(1+)

-
B
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With carbon tetrabromide In 1,2-dichloro-ethane at 60℃; for 2h; | A n/a B 95% |

-
-
122-97-4
3-Phenyl-1-propanol

-
-
1229444-44-3
N-methyl-N-[4-(diphenylphosphino)benzyl]pyrrolidinium bromide

-
A
-
1229444-42-1
Br(1-)*C24H27NOP(1+)

-
B
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With carbon tetrabromide In 1,2-dichloro-ethane at 40℃; for 2h; Inert atmosphere; | A 93% B 95% |

-
-
866942-22-5
2-(3-Phenyl-propoxysulfonyl)-benzoic acid 1,4,7,10,13-pentaoxa-cyclopentadec-2-ylmethyl ester

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With sodium bromide In acetone for 0.37h; Product distribution; Further Variations:; Reagents; Temperatures; reaction time; Heating; | 93% |

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In tetrahydrofuran at 80℃; for 4.5h; | 92% |

-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In acetonitrile at 20℃; Inert atmosphere; Sealed tube; | 82% |
-
-
100-58-3
phenylmagnesium bromide

-
-
109-64-8
1,3-dibromo-propane

-
A
-
92-52-4
biphenyl

-
B
-
1081-75-0
1,3-diphenylpropane

-
C
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With copper(I) bromide In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide for 4h; Product distribution; Heating; Grignard reagent added dropwise to the other reaction partners; | A 3% B 16% C 81% |


| Conditions | Yield |
|---|---|
| With 1,3,5-trichloro-2,4,6-triazine; N,N-dimethyl-formamide; sodium bromide In dichloromethane at 25℃; for 0.25h; | A 80% B n/a |

| Conditions | Yield |
|---|---|
| With boron tribromide; tert-butyl alcohol In dichloromethane at 23℃; for 1h; regioselective reaction; | 77% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
27126-20-1
(3-azidopropyl)benzene

| Conditions | Yield |
|---|---|
| With sodium azide In water; acetone at 20℃; | 100% |
| With sodium azide In N,N-dimethyl-formamide at 80℃; | 100% |
| With sodium azide In dimethyl sulfoxide at 20℃; | 100% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
106-45-6
para-thiocresol

-
-
38644-97-2
(3-phenylpropyl)(p-tolyl)sulfane

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 12h; | 100% |
| With potassium hydroxide | |
| With potassium carbonate In acetone at 45℃; |

| Conditions | Yield |
|---|---|
| With sodium iodide In acetone for 6h; Reflux; | 100% |
| With sodium iodide In acetone Reflux; | 97% |
| With potassium iodide In acetone for 2h; Reflux; | 95% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
845778-49-6
5-azido-5-deoxy-1,2-O-isopropylidene-3-O-methoxymethyl-β-L-idofuranose

-
-
763122-34-5
5-azido-5-deoxy-6-O-(3-phenylpropyl)-1,2-O-isopropylidene-3-O-methoxymethyl-β-L-idofuranose

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; | 100% |
-
-
863642-50-6
N-methyl-N-methoxy-2-benzyloxy-4-hexenamide

-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
863642-51-7
1-phenyl-5-benzyloxy-7-nonen-4-one

| Conditions | Yield |
|---|---|
| Stage #1: 1-Bromo-3-phenylpropane With magnesium In tetrahydrofuran Heating; Stage #2: N-methyl-N-methoxy-2-benzyloxy-4-hexenamide In tetrahydrofuran at 0℃; for 1.5h; | 100% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
25333-42-0
(R)-quinuclidin-3-ol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 7h; Heating / reflux; | 100% |

| Conditions | Yield |
|---|---|
| at 140℃; Continuous flow; neat (no solvent); | 100% |
| In acetonitrile for 8h; Reflux; |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
105-34-0
methyl 2-cyanoacetate

-
-
1202778-95-7
cyano-di(3-phenyl-prop-1-yl)acetic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: methyl 2-cyanoacetate With sodium methylate In methanol at 20℃; Stage #2: 1-Bromo-3-phenylpropane In methanol Reflux; | 100% |

| Conditions | Yield |
|---|---|
| for 24h; Reflux; | 100% |
| In acetonitrile at 70℃; for 31.5h; | 84% |
| at 140℃; Neat (no solvent); | |
| In N,N-dimethyl-formamide at 100℃; for 4h; Sealed tube; |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
802294-64-0
propionic acid

-
-
143097-60-3
(±)-2-methyl-5-phenylpentanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: propionic acid With N,N,N,N,N,N-hexamethylphosphoric triamide; n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -20 - 20℃; for 0.5h; Stage #2: 1-Bromo-3-phenylpropane In tetrahydrofuran; hexane at 0 - 20℃; for 2h; | 100% |
| Stage #1: propionic acid With n-butyllithium; diisopropylamine In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -20 - 20℃; for 0.5h; Stage #2: 1-Bromo-3-phenylpropane In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at 0 - 20℃; for 2h; | 100% |
| Stage #1: propionic acid With n-butyllithium; diisopropylamine In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at 20℃; for 0.5h; Stage #2: 1-Bromo-3-phenylpropane In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at 0 - 20℃; for 2h; | 100% |
| Stage #1: propionic acid With n-butyllithium; diisopropylamine In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -20 - 20℃; for 0.5h; Stage #2: 1-Bromo-3-phenylpropane In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at 0 - 20℃; for 2h; | 100% |
| Stage #1: propionic acid With N,N,N,N,N,N-hexamethylphosphoric triamide; n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at 0 - 20℃; Inert atmosphere; Stage #2: 1-Bromo-3-phenylpropane In tetrahydrofuran; hexane at 0 - 20℃; Inert atmosphere; | 62% |
-
-
637-59-2
1-Bromo-3-phenylpropane

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl acetamide at 80℃; for 3h; | 100% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
603-35-0
triphenylphosphine

-
-
7484-37-9
(3-phenylpropyl)triphenylphosphonium bromide

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | 99% |
| In toluene for 50h; Reflux; | 92% |
| In toluene for 62h; Heating; | 92% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
120703-53-9
Bis(trimethylsilyl)methyl-methylether

-
-
120703-47-1
(4-Methoxy-4,4-bis-trimethylsilanyl-butyl)-benzene

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane | 99% |
| With n-butyllithium 1) hexane, THF, 0 deg C, 20 min, 2a) -78 deg C, 5 min, 2b) 0 deg C, 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With LiCrH4*2LiCl*2THF In tetrahydrofuran at 25℃; for 12h; | 99% |
| With indium(III) chloride; sodium tetrahydroborate In acetonitrile at 20℃; for 2h; | 95% |
| With sodium tetrahydroborate; water In methanol at 20℃; for 0.5h; | 87% |
-
-
533-58-4
2-Iodophenol

-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
939990-21-3
1-iodo-2-(3-phenylpropoxy)benzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 99% |
-
-
1201582-46-8
1-(3-hydroxy-2,4-dimethoxy-6-methyl-5-nitrophenyl)ethanone

-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
1201582-47-9
1-(2,4-dimethoxy-6-methyl-5-nitro-3-(3-phenylpropoxy)phenyl)ethanone

| Conditions | Yield |
|---|---|
| Stage #1: 1-(3-hydroxy-2,4-dimethoxy-6-methyl-5-nitrophenyl)ethanone With sodium hydride In N,N-dimethyl-formamide; oil at 20℃; for 0.25h; Stage #2: 1-Bromo-3-phenylpropane In N,N-dimethyl-formamide; oil at 20℃; | 99% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
106-48-9
4-chloro-phenol

-
-
1044066-84-3
1-chloro-4-(3-phenylpropoxy)benzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 40℃; | 99% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
171414-16-7
N-(4-methoxybenzyl)-2-nitro-N-(prop-2-yn-1-yl)benzenesulfonamide

-
-
704892-79-5
N-(4-methoxybenzyl)-N-(3-phenylpropyl)-2-nitrobenzenesulfonamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 1.25h; | 99% |
-
-
1126-09-6
4-carbethoxypiperidine

-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
21327-50-4
ethyl 1-(3-phenylpropyl)piperidine-4-carboxylate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 2h; | 98.6% |
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; | 87% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
89987-96-2
1,2-bis(3-phenylpropyl)disulfane

| Conditions | Yield |
|---|---|
| With potassium sulfide; nickel(II) chloride hexahydrate; acetylacetone; potassium hydroxide In water; N,N-dimethyl-formamide at 110℃; for 6h; | 98% |
| With potassium 5-methyl-1,3,4-oxadiazole-2-thiolate In water at 40℃; for 1h; | 98% |
| With potassium fluoride on basic alumina; thioacetamide; copper(l) chloride In N,N-dimethyl-formamide at 110℃; for 5h; | 95% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
1068-90-2
2-acetylaminomalonic acid diethyl ester

-
-
52161-73-6
2-acetylamino-2-(3-phenyl-propyl)malonic acid diethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 2-acetylaminomalonic acid diethyl ester With sodium ethanolate In ethanol at 20℃; for 1.5h; Stage #2: 1-Bromo-3-phenylpropane In ethanol for 16h; Heating; | 98% |
| Stage #1: 2-acetylaminomalonic acid diethyl ester With sodium ethanolate In ethanol at 20℃; for 1.5h; Stage #2: 1-Bromo-3-phenylpropane In ethanol for 16.25h; Heating / reflux; | 98% |
| (i) Na, EtOH, (ii) /BRN= 2205527/; Multistep reaction; | |
| With sodium ethanolate; sodium In ethanol; nitrogen | |
| Stage #1: 2-acetylaminomalonic acid diethyl ester With sodium ethanolate In ethanol at 20℃; for 2h; Stage #2: 1-Bromo-3-phenylpropane In ethanol at 20 - 73℃; for 28.25h; |

| Conditions | Yield |
|---|---|
| With silver tetrafluoroborate; 1-(2-bromo-1,1-dimethoxyethyl)benzene at 25℃; for 4h; | 98% |
| With sodium for 2h; Heating; | 51 g |
| With sodium |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
625-36-5
2-chloropropionyl chloride

-
-
253308-01-9
1-(3-chloro-1-oxopropyl)-4-(3-bromopropyl)benzene

| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane at 20℃; for 6h; Friedel-Crafts acylation; | 98% |
-
-
637-59-2
1-Bromo-3-phenylpropane

-
-
609-08-5
Diethyl methylmalonate

-
-
98468-72-5
diethyl methyl(3-phenylpropyl)malonate

| Conditions | Yield |
|---|---|
| Stage #1: Diethyl methylmalonate With lithium hydride In N,N-dimethyl-formamide at 0℃; for 0.5h; Stage #2: 1-Bromo-3-phenylpropane In N,N-dimethyl-formamide at 80℃; for 3h; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 1H-imidazole With sodium hydroxide In dimethyl sulfoxide at 70 - 80℃; for 1.5h; Stage #2: 1-Bromo-3-phenylpropane In dimethyl sulfoxide at 70 - 80℃; for 13h; | 98% |
| Stage #1: 1H-imidazole With potassium carbonate In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: 1-Bromo-3-phenylpropane In tetrahydrofuran Heating; | |
| In N,N-dimethyl-formamide | 16.1 g (86%) |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View







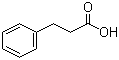








 Xi
Xi