-
Name
Flupirtine
- EINECS 260-503-8
- CAS No. 56995-20-1
- Article Data7
- CAS DataBase
- Density 1.35 g/cm3
- Solubility 1g/L(temperature not stated)
- Melting Point 115-116°
- Formula C15H17FN4O2
- Boiling Point 434.9 °C at 760 mmHg
- Molecular Weight 304.324
- Flash Point 216.8 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Carbamicacid, [2-amino-6-[[(4-fluorophenyl)methyl]amino]-3-pyridinyl]-, ethyl ester(9CI);D 9998;Flupirtine;Katadolon;Trancopal Dolo;
- PSA 89.27000
- LogP 3.71060
Synthetic route
-
-
541-41-3
chloroformic acid ethyl ester

-
-
112523-78-1
N6-(4-fluorobenzyl)pyridine-2,3,6-triamine

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 20℃; Inert atmosphere; Darkness; | 82% |
| In 1,4-dioxane at 20℃; for 1h; | 75% |
| In tetrahydrofuran Ambient temperature; Yield given; |
-
-
452-58-4
2,3-Diaminopyridine

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 41.8 percent / propan-2-ol / 71 h / Heating 2: 52.8 percent / MnO2 / tetrahydrofuran / 18 h / Ambient temperature 3: Na2S2O4 / tetrahydrofuran; H2O / Ambient temperature 4: tetrahydrofuran / Ambient temperature View Scheme |
-
-
76902-24-4
spiropyridine>

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 52.8 percent / MnO2 / tetrahydrofuran / 18 h / Ambient temperature 2: Na2S2O4 / tetrahydrofuran; H2O / Ambient temperature 3: tetrahydrofuran / Ambient temperature View Scheme |
-
-
161263-86-1
5'-<(4-fluorobenzyl)amino>spiropyridine>

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Na2S2O4 / tetrahydrofuran; H2O / Ambient temperature 2: tetrahydrofuran / Ambient temperature View Scheme |
-
-
16013-85-7
2,6-dicholoro-3-nitropyridine

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: ammonia / methanol / 20 °C / Inert atmosphere 2: triethylamine / isopropyl alcohol / 3 h / Reflux; Inert atmosphere 3: hydrogenchloride; tin(II) chloride dihdyrate / water / 3 h / 70 °C / Inert atmosphere 4: 1,4-dioxane / 20 °C / Inert atmosphere; Darkness View Scheme |
-
-
27048-04-0
2-amino-6-chloro-3-nitropyridine

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: triethylamine / isopropyl alcohol / 3 h / Reflux; Inert atmosphere 2: hydrogenchloride; tin(II) chloride dihdyrate / water / 3 h / 70 °C / Inert atmosphere 3: 1,4-dioxane / 20 °C / Inert atmosphere; Darkness View Scheme | |
| Multi-step reaction with 3 steps 1: triethylamine / isopropyl alcohol / 0.5 h / 5 °C 2: palladium on activated charcoal; hydrogen / methanol / 5 h / 20 °C / 1551.49 Torr 3: 1,4-dioxane / 1 h / 20 °C View Scheme |
-
-
33400-49-6
2-amino-3-nitro-6-(p-fluoro-benzylamino)-pyridine

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogenchloride; tin(II) chloride dihdyrate / water / 3 h / 70 °C / Inert atmosphere 2: 1,4-dioxane / 20 °C / Inert atmosphere; Darkness View Scheme | |
| Multi-step reaction with 2 steps 1: palladium on activated charcoal; hydrogen / methanol / 5 h / 20 °C / 1551.49 Torr 2: 1,4-dioxane / 1 h / 20 °C View Scheme |
-
-
140-75-0
para-fluorobenzylamine

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: triethylamine / isopropyl alcohol / 0.5 h / 5 °C 2: palladium on activated charcoal; hydrogen / methanol / 5 h / 20 °C / 1551.49 Torr 3: 1,4-dioxane / 1 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| In Isopropyl acetate; dimethyl sulfoxide at 55℃; for 1h; Temperature; Solvent; Inert atmosphere; | 96% |
-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| at 200℃; for 0.5h; | 88% |

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol for 16h; Reflux; | 70% |
-
-
15307-86-5
[2-(2,6-dichloroanilino)phenyl]acetic acid

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| In toluene at 60℃; Inert atmosphere; | 66% |
-
-
56995-20-1
Flupirtine

-
-
74-88-4
methyl iodide

| Conditions | Yield |
|---|---|
| Stage #1: Flupirtine With sodium hydride In tetrahydrofuran; hexane at 0℃; for 0.5h; Inert atmosphere; Darkness; Stage #2: methyl iodide In tetrahydrofuran; hexane Inert atmosphere; | 40% |
-
-
56995-20-1
Flupirtine

-
-
76-05-1
trifluoroacetic acid

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at -5 - 30℃; | |
| In isopropyl alcohol at 30℃; |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 17 - 60℃; Product distribution / selectivity; | |
| In isopropyl alcohol at 20 - 60℃; Product distribution / selectivity; | |
| In isopropyl alcohol at 20 - 60℃; Product distribution / selectivity; | |
| In isopropyl alcohol at 60℃; |
-
-
56995-20-1
Flupirtine

-
-
64-19-7
acetic acid

-
-
1198579-27-9
2-amino-3-carbethoxyamino-6-(4-fluoro-benzylamino)-pyridine acetate

| Conditions | Yield |
|---|---|
| In ethanol at -15 - 60℃; Inert atmosphere; | |
| In ethanol at 40 - 60℃; Inert atmosphere; |
-
-
56995-20-1
Flupirtine

-
-
802294-64-0
propionic acid

-
-
1198579-28-0
2-amino-3-carbethoxyamino-6-(4-fluoro-benzylamino)-pyridine propionate

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at -5 - 60℃; Inert atmosphere; | |
| In isopropyl alcohol at 50 - 60℃; Inert atmosphere; |
-
-
104-15-4
toluene-4-sulfonic acid

-
-
56995-20-1
Flupirtine

-
-
1200119-41-0
2-amino-3-carbethoxyamino-6-(4-fluoro-benzylamino)-pyridine tosilate

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20 - 50℃; | |
| In isopropyl alcohol at 20 - 40℃; |
-
-
75-75-2
methanesulfonic acid

-
-
56995-20-1
Flupirtine

-
-
1200119-31-8
2-amino-3-carbethoxyamino-6-(4-fluoro-benzylamino)-pyridine mesilate

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20 - 60℃; for 0.5h; Inert atmosphere; | |
| In isopropyl alcohol at 20 - 60℃; Inert atmosphere; |
-
-
56995-20-1
Flupirtine

-
-
98-11-3
benzenesulfonic acid

-
-
1200119-39-6
2-amino-3-carbethoxyamino-6-(4-fluoro-benzylamino)-pyridine besilate

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20 - 60℃; for 0.5h; Inert atmosphere; | |
| In isopropyl alcohol at 20 - 60℃; Inert atmosphere; |
-
-
104-15-4
toluene-4-sulfonic acid

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20 - 40℃; |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 30℃; for 0.5h; |
-
-
75-75-2
methanesulfonic acid

-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20 - 60℃; Inert atmosphere; |
-
-
56995-20-1
Flupirtine

-
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| In ethanol at 40 - 60℃; Inert atmosphere; |
-
-
56995-20-1
Flupirtine

-
-
802294-64-0
propionic acid

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 60℃; Inert atmosphere; |
-
-
56995-20-1
Flupirtine

-
-
98-11-3
benzenesulfonic acid

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20 - 60℃; Inert atmosphere; |
-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 0.5 h / 200 °C 2: acetonitrile; N,N-dimethyl-formamide / 75 °C View Scheme |
-
-
56995-20-1
Flupirtine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 0.5 h / 200 °C 2: acetonitrile; N,N-dimethyl-formamide / 75 °C View Scheme |
Flupirtine Specification
The Flupirtine with CAS registry number of 56995-20-1 is also known as Flupirtinum. The IUPAC name is Ethyl N-[2-amino-6-[(4-fluorophenyl)methylamino]pyridin-3-yl]carbamate. Its classification codes are Analgesics; Central Nervous System Agents; Peripheral Nervous System Agents; Sensory System Agents. In addition, the formula is C15H17FN4O2 and the molecular weight is 304.32.
Physical properties about Flupirtine are: (1)ACD/LogP: 1.46 ; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.35; (4)ACD/LogD (pH 7.4): 1.45; (5)ACD/BCF (pH 5.5): 5.88; (6)ACD/BCF (pH 7.4): 7.5; (7)ACD/KOC (pH 5.5): 115.33; (8)ACD/KOC (pH 7.4): 147.07; (9)#H bond acceptors: 6; (10)#H bond donors: 4; (11)#Freely Rotating Bonds: 5; (12)Polar Surface Area: 48.91Å2; (13)Index of Refraction: 1.661; (14)Molar Refractivity: 83.32 cm3; (15)Molar Volume: 225.2 cm3; (16)Polarizability: 33.03×10-24cm3; (17)Surface Tension: 60.6 dyne/cm; (18)Density: 1.35 g/cm3; (19)Flash Point: 216.8 °C ; (20)Enthalpy of Vaporization: 69.12 kJ/mol ; (21)Boiling Point: 434.9 °C at 760 mmHg ; (22)Vapour Pressure: 9.1E-08 mmHg at 25 °C.
Preparation of Flupirtine: it is prepared by reaction of 2-amino-3-nitro-6-chloro pyridine with p-fluorobenzyl amine. Then restore the resulting product, which is acylated by ethyl chloride. Finally, react with maleate in isopropyl alcohol to obtain the product.
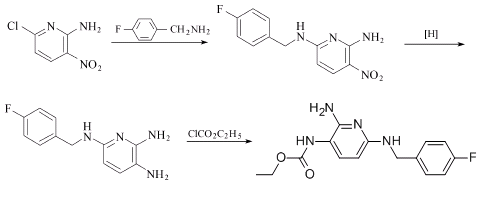
Uses of Flupirtine: it is used as short-term treatment for surgery, trauma, burns and pain caused by toothache. What's more, it can act on the central nervous system without inhibition of respiration or cough.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: CCOC(=O)NC1=C(N=C(C=C1)NCC2=CC=C(C=C2)F)N
2. InChI: InChI=1S/C15H17FN4O2/c1-2-22-15(21)19-12-7-8-13(20-14(12)17)18-9-10-3-5-11(16)6-4-10/h3-8H,2,9H2,1H3,(H,19,21)(H3,17,18,20)
3. InChIKey: JUUFBMODXQKSTD-UHFFFAOYSA-N
Related Products
- Flupirtine
- Flupirtine maleate
- 5699-54-7
- 56996-62-4
- 5699-79-6
- 5700-03-8
- 57000-78-9
- 57-00-1
- 570-02-5
- 5700-49-2
- 57005-04-6
- 5700-56-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View