-
Name
N-Acetylneuraminic acid
- EINECS 205-023-1
- CAS No. 131-48-6
- Article Data17
- CAS DataBase
- Density 1.727 g/cm3
- Solubility Water : 50 g/L (20 °C)
- Melting Point 184-186 °C
- Formula C11H19NO9
- Boiling Point 395.6 °C at 760 mmHg
- Molecular Weight 309.273
- Flash Point 193 °C
- Transport Information
- Appearance Crystalline
- Safety 22-24/25-36-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Aceneuramic acid;Acide aceneuramique;Acidium aceneuramicum;Acido aceneuramico;(-)-5-Acetamido-3,5-dideoxy-D-glycero-D-galacto-nonulosonic;Neuraminic acid, N-acetyl-;
- PSA 176.78000
- LogP -3.48090
Synthetic route

-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| Stage #1: 5-(1-acetylamino-2,3,4,5-tetrahydroxy-pentyl)-2-tert-butyl-isoxazolidine-3-carboxylic acid ethyl ester With sodium methylate In methanol at 20℃; Stage #2: With water for 24h; |

| Conditions | Yield |
|---|---|
| With potassium phosphate buffer; magnesium chloride In water; xylene at 28℃; for 32h; pH=8.0; |

| Conditions | Yield |
|---|---|
| With potassium phosphate buffer; D-glucose; magnesium chloride In water; xylene at 28℃; for 32h; pH=8.0; |
-
-
72-87-7, 1136-42-1, 1136-44-3, 6082-29-7, 6730-06-9, 7772-94-3, 10036-64-3, 14131-60-3, 14131-64-7, 14131-68-1, 14215-68-0, 62057-49-2, 62057-50-5, 62057-51-6, 62057-52-7, 62057-53-8, 62057-54-9, 62057-55-0, 62057-56-1, 62075-50-7, 62137-54-6, 134451-97-1, 134522-12-6, 148347-16-4
N-acetyl-D-mannosamine

-
-
312-85-6
sodium lactate

-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| With tris hydrochloride; Pseudomonas stutzeri SDM cells (lactate oxidase) In water; toluene at 30℃; for 20h; pH=7.0; Microbiological reaction; | 5.93 g |

-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: sodium hydrogen carbonate / methanol / 1 h 2.1: dioxane / 336 h / 30 °C 3.1: NaOMe / methanol / 20 °C 3.2: water / 24 h View Scheme |

-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: dioxane / 336 h / 30 °C 2.1: NaOMe / methanol / 20 °C 2.2: water / 24 h View Scheme |

-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: trifluoroacetic acid / ethanol / 16 h 2.1: sodium hydrogen carbonate / methanol / 1 h 3.1: dioxane / 336 h / 30 °C 4.1: NaOMe / methanol / 20 °C 4.2: water / 24 h View Scheme |

-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| With recombinant oxidoreductase from Ruminococcus gnavus ATCC 29149; NADH In aq. phosphate buffer at 37℃; pH=7.5; Catalytic behavior; Solvent; Time; Reagent/catalyst; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With Amberlite IR 120 H(1+) form at 20℃; for 15h; | 100% |
| With Dowex 50 x 8 (H(1+) form) for 2h; Ambient temperature; | |
| With Amberlite IR-120 (H+) ion-exchange resin |
-
-
131-48-6
N-Acetylneuraminic acid


| Conditions | Yield |
|---|---|
| With Neisseria meningitidis CMP-sialic acid synthetase; Pasteurella multocida α2,3-sialyltransferase 3; cytidine triphosphate; magnesium chloride In aq. buffer at 37℃; pH=8.5; Green chemistry; Enzymatic reaction; | 96% |
-
-
131-48-6
N-Acetylneuraminic acid

-
-
20298-35-5, 22900-11-4, 100678-43-1, 109121-98-4, 109122-00-1, 119241-60-0, 119241-61-1, 119241-62-2
N-acetylneuraminic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol | 95% |
-
-
67-56-1
methanol

-
-
131-48-6
N-Acetylneuraminic acid

-
-
20298-35-5, 22900-11-4, 100678-43-1, 109121-98-4, 109122-00-1, 119241-60-0, 119241-61-1, 119241-62-2
N-acetylneuraminic acid methyl ester

| Conditions | Yield |
|---|---|
| With Amberlite IR-120 resin H(1+) form | 92% |
-
-
131-48-6
N-Acetylneuraminic acid

-
-
1246842-76-1
3-azidopropyl (β-D-galactopyranosyl)-(1→4)-O-2-deoxy-2-acetamido-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With Neisseria meningitidis CMP-sialic acid synthetase; Pasteurella multocida α2,3-sialyltransferase 3; cytidine triphosphate; magnesium chloride In aq. buffer at 37℃; pH=8.5; Green chemistry; Enzymatic reaction; | 91% |
-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| With Neisseria meningitidis CMP-sialic acid synthetase; Pasteurella multocida α2,3-sialyltransferase 3; cytidine triphosphate; magnesium chloride In aq. buffer at 37℃; pH=8.5; Green chemistry; Enzymatic reaction; | 91% |
-
-
131-48-6
N-Acetylneuraminic acid


| Conditions | Yield |
|---|---|
| With phospho(enol)pyruvic acid mono potassium salt; cytidine triphosphate; ATP In aq. buffer at 37℃; pH=7.5; Enzymatic reaction; | 90% |
-
-
131-48-6
N-Acetylneuraminic acid


| Conditions | Yield |
|---|---|
| With Neisseria meningitidis CMP-sialic acid synthetase; Pasteurella multocida α2,3-sialyltransferase 3; cytidine triphosphate; magnesium chloride In aq. buffer at 37℃; pH=8.5; Green chemistry; Enzymatic reaction; | 87% |
-
-
131-48-6
N-Acetylneuraminic acid


| Conditions | Yield |
|---|---|
| With phospho(enol)pyruvic acid mono potassium salt; cytidine triphosphate; ATP In aq. buffer at 37℃; pH=7.5; Enzymatic reaction; | 80% |
-
-
131-48-6
N-Acetylneuraminic acid

-
-
1257307-68-8
3-azidopropyl β-D-galactopyranosyl-(1->3)-2-acetamido-2-deoxy-α-D-galactopyranoside

-
-
1282094-51-2
3-azidopropyl O-(5-acetamido-3,5-dideoxy-D-glycero-α-D-galacto-2-nonulopyranosylonic acid)-(2→3)-β-D-galactopyranosyl-(1→3)-2-acetamido-2-deoxy-β-D-galactopyranoside

| Conditions | Yield |
|---|---|
| With Neisseria meningitidis CMP-sialic acid synthetase; Pasteurella multocida α2,3-sialyltransferase1 mutant; cytidine triphosphate; magnesium chloride In aq. buffer at 37℃; pH=8.5; Green chemistry; Enzymatic reaction; | 76% |
-
-
67-56-1
methanol

-
-
131-48-6
N-Acetylneuraminic acid

-
-
6730-43-4
N-Acetyl-β-D-neuraminylsaeure-methyl-methylglycosid

| Conditions | Yield |
|---|---|
| With ion exchange resin Dowex 50x8 H+ for 72h; Reflux; | 71% |
-
-
131-48-6
N-Acetylneuraminic acid

-
-
1061653-82-4
3-azidopropyl β-D-galactopyranosyl-(1->3)-2-acetamido-2-deoxy-β-D-glucopyranoside

-
-
1282094-45-4
3-azidopropyl 5-acetamido-3,5-dideoxy-D-glycero-α-D-galacto-2-nonulopyranosyl-(2→3)-β-D-galactopyranosyl-(1→3)-2-acetamido-2-deoxy-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With Neisseria meningitidis CMP-sialic acid synthetase; Pasteurella multocida α2,3-sialyltransferase1 mutant; cytidine triphosphate; magnesium chloride In aq. buffer at 37℃; pH=8.5; Green chemistry; Enzymatic reaction; | 63% |
-
-
131-48-6
N-Acetylneuraminic acid


-
-
5-aminopentyl 5-acetamido-3,5-dideoxy-D-glycero-α-D-galacto-2-nonulopyranosylonate-(2→6)-β-D-galactopyranosyl-(1→4)-2-acetamido-2-deoxy-β-D-glucopyranosyl-(1→2)-α-D-mannopyranosyl-(1→3)-[di(α-D-mannopyranosyl)-(1→3),(1→6)-α-D-mannopyranosyl]-(1→6)-β-D-mannopyranosyl-(1→4)-2-acetamido-2-deoxy-β-D-glucopyranosyl-(1→4)-2-acetamido-2-deoxy-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With phospho(enol)pyruvic acid mono potassium salt; cytidine triphosphate; ATP In aq. buffer at 37℃; for 48h; pH=7.5; Enzymatic reaction; | 60% |
-
-
131-48-6
N-Acetylneuraminic acid


-
-
5-aminopentyl 5-acetamido-3,5-dideoxy-D-glycero-α-D-galacto-2-nonulopyranosylonate-(2→3)-β-D-galactopyranosyl-(1→4)-2-acetamido-2-deoxy-β-D-glucopyranosyl-(1→2)-α-D-mannopyranosyl-(1→3)-[di(α-D-mannopyranosyl)-(1→3),(1→6)-α-D-mannopyranosyl]-(1→6)-β-D-mannopyranosyl-(1→4)-2-acetamido-2-deoxy-β-D-glucopyranosyl-(1→4)-2-acetamido-2-deoxy-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With phospho(enol)pyruvic acid mono potassium salt; cytidine triphosphate; ATP In aq. buffer at 37℃; for 192h; pH=7.5; Enzymatic reaction; | 56% |
-
-
131-48-6
N-Acetylneuraminic acid


| Conditions | Yield |
|---|---|
| With phospho(enol)pyruvic acid mono potassium salt; cytidine triphosphate; ATP In aq. buffer at 37℃; for 96h; pH=7.5; Enzymatic reaction; | 52% |

| Conditions | Yield |
|---|---|
| With pyrophosphatase; α-2,6-sialyltransferase; cytidine triphosphate; CMP-sialic acid synthetases; magnesium chloride; manganese(ll) chloride In aq. buffer Enzymatic reaction; | 50% |
-
-
131-48-6
N-Acetylneuraminic acid

-
-
56144-08-2
5-amino-O2-methyl-β-D-glycero-D-galacto-3,5-dideoxy-[2]nonulopyranosonic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; methanol at 105℃; |
-
-
131-48-6
N-Acetylneuraminic acid

-
-
75-08-1
ethanethiol

-
-
99336-25-1
5-Acetamino-3,5-didesoxy-D-glycero-D-galakto-2-keto-nononsaeure-γ-lacton-diethylmercaptal

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With Dowex 50 x 8 (H(1+) form) for 3h; Ambient temperature; |
-
-
131-48-6
N-Acetylneuraminic acid

| Conditions | Yield |
|---|---|
| With hydrogen fluoride |
-
-
133-99-3, 490-37-9, 2152-98-9, 4482-75-1, 5965-66-2, 13299-27-9, 13360-52-6, 14641-93-1, 15548-43-3, 16462-44-5, 16984-36-4, 16984-38-6, 20869-27-6, 22688-72-8, 27452-49-9, 29276-55-9, 30608-12-9, 34481-13-5, 37169-60-1, 37169-64-5, 64234-01-1, 64234-02-2, 75281-88-8, 80446-85-1, 84413-57-0, 92344-56-4, 93221-87-5, 93301-77-0, 94799-29-8, 100427-98-3, 100428-00-0, 101312-82-7, 102046-24-2, 102046-25-3, 109432-00-0, 109432-02-2, 109432-04-4, 109432-08-8, 135268-49-4, 138231-26-2, 140461-59-2, 145414-22-8, 145414-26-2, 146339-75-5, 146339-76-6, 149116-55-2
Lactose

-
-
72-87-7, 1136-42-1, 1136-44-3, 6730-06-9, 7772-94-3, 10036-64-3, 14131-60-3, 14131-64-7, 14131-68-1, 14215-68-0, 62057-49-2, 62057-50-5, 62057-51-6, 62057-52-7, 62057-53-8, 62057-54-9, 62057-55-0, 62057-56-1, 62075-50-7, 62137-54-6, 134451-97-1, 134522-12-6, 148347-16-4, 6082-29-7
N-acetyl-D-galactosamine

-
-
131-48-6
N-Acetylneuraminic acid

-
A
-
117773-04-3
O-(5-acetamido-3,5-dideoxy-α-D-glycero-D-galacto-2-nonulopyranosylonic-acid)-(2-3)-O-β-D-galactopyranosyl-(1-3)-2-acetamido-2-deoxy-β-D-galactopyranose

| Conditions | Yield |
|---|---|
| With Triton-X-100; Na-cacodylate buffer; potassium chloride; phospho(enol)pyruvic acid mono potassium salt; cytidine monophosphate; cytidine triphosphate; magnesium chloride; manganese(ll) chloride In water at 37℃; for 82h; myokinase from porcine muscle, pyruvate kinase, inorganical phosphatase, CMP-Neu5Ac-synthase from calf brain, β-galactosidase from bovine testes, α-2,3-sialyltransferase from rat liver; pH 7.5; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
72-87-7, 1136-42-1, 1136-44-3, 6730-06-9, 7772-94-3, 10036-64-3, 14131-60-3, 14131-64-7, 14131-68-1, 14215-68-0, 62057-49-2, 62057-50-5, 62057-51-6, 62057-52-7, 62057-53-8, 62057-54-9, 62057-55-0, 62057-56-1, 62075-50-7, 62137-54-6, 134451-97-1, 134522-12-6, 148347-16-4, 6082-29-7
N-acetyl-D-galactosamine

-
-
131-48-6
N-Acetylneuraminic acid

-
A
-
117773-04-3
O-(5-acetamido-3,5-dideoxy-α-D-glycero-D-galacto-2-nonulopyranosylonic-acid)-(2-3)-O-β-D-galactopyranosyl-(1-3)-2-acetamido-2-deoxy-β-D-galactopyranose

-
B
-
117249-18-0
O-(5-acetamido-3,5-dideoxy-α-D-glycero-D-galacto-2-nonulopyranosylonic-acid)-(2-3)-O-β-D-galactopyranosyl-(1-3)-2-acetamido-2-deoxy-α-D-galactopyranose

| Conditions | Yield |
|---|---|
| With Triton-X-100; Na-cacodylate buffer; potassium chloride; phospho(enol)pyruvic acid mono potassium salt; cytidine monophosphate; cytidine triphosphate; magnesium chloride; manganese(ll) chloride In water at 37℃; for 82h; myokinase from porcine muscle, pyruvate kinase, inorganical phosphatase, CMP-Neu5Ac-synthase from calf brain, β-galactosidase from bovine testes, α-2,3-sialyltransferase from rat liver; pH 7.5; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
131-48-6
N-Acetylneuraminic acid
-
-
131-48-6
N-Acetylneuraminic acid

-
-
19342-33-7
N-acetyl neuraminic acid

| Conditions | Yield |
|---|---|
| With formic acid; water |
-
-
131-48-6
N-Acetylneuraminic acid

-
-
135754-36-8
4-(acetylamino)-2,4-dideoxy-D-glycero-D-galacto-octonic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In phosphate buffer at 37℃; pH=6 - 8; Kinetics; Further Variations:; pH-values; Solvents; Temperatures; |
N-Acetylneuraminic Acid Chemical Properties
IUPAC Name: (2S,4S,5R,6R)-5-Acetamido-2,4-dihydroxy-6-(1,2,3-trihydroxypropyl)oxane-2-carboxylic acid
Synonyms of N-Acetylneuraminic Acid (CAS NO.131-48-6): Aceneuramic acid ; Acide aceneuramique ; Acidium aceneuramicum ; Acido aceneuramico ; (-)-5-Acetamido-3,5-dideoxy-D-glycero-D-galacto-nonulosonic acid ; Neuraminic acid, N-acetyl-
CAS NO: 131-48-6
Molecular Formula: C11H19NO9
Molecular Weight: 309.27
Molecular Structure: 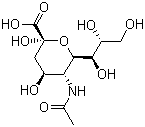
EINECS: 205-023-1
H bond acceptors: 2
H bond donors: 1
Freely Rotating Bonds: 1
Polar Surface Area: 22 Å2
Index of Refraction: 1.752
Molar Refractivity: 52.97 cm3
Molar Volume: 129.7 cm3
Surface Tension: 63.5 dyne/cm
Density: 1.727 g/cm3
Flash Point: 193 °C
Enthalpy of Vaporization: 64.57 kJ/mol
Boiling Point: 395.6 °C at 760 mmHg
Vapour Pressure: 1.82E-06 mmHg at 25°C
Melting point: 184-186 ºC
Alpha: -32 º (c=2,water)
Water solubility: 50 g/L (20 ºC)
Storage temp: −20°C
Sensitive: Air Sensitive
Stability: Stable. Incompatible with strong oxidizing agents.
Appearance: Crystalline
Product Categories of N-Acetylneuraminic Acid (CAS NO.131-48-6): Bioproducts;Sugar derivatives;Miscellaneous Natural Products;Zanamavir;13C & 2H Sugars;Sialic Acids;Biochemistry;Sugar Acids;Sugars;Carbohydrates & Derivatives;Miscellaneous Compounds
N-Acetylneuraminic Acid Uses
Along with involvement in preventing infections, N-Acetylneuraminic Acid (CAS NO.131-48-6) acts as a receptor for influenza viruses, allowing attachment to mucous cells via hemaglutinin.
N-Acetylneuraminic Acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 22-24/25-36-26
S22: Do not breathe dust.
S24/25: Avoid contact with skin and eyes.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clothing.
WGK Germany: 3
F: 3-10-23
Hazard Note: Irritant
N-Acetylneuraminic Acid Specification
General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media: In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam.
Handling: Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Avoid contact with eyes, skin, and clothing. Avoid ingestion and inhalation.
Storage: Store N-Acetylneuraminic Acid (CAS NO.131-48-6) in a cool, dry place. Keep container closed when not in use. Deep freeze (below -20°C). Keep refrigerated. (Store below 4°C/39°F.)
Related Products
- N-Acetylneuraminic Acid
- N-Acetylneuraminic acid methyl ester
- 1314-87-0
- 131488-15-8
- 13149-00-3
- 1314-91-6
- 1314-96-1
- 131-49-7
- 1314-98-3
- 13150-00-0
- 1315-01-1
- 1315-04-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View