-
Name
Tetrapropylammonium bromide
- EINECS 217-727-6
- CAS No. 1941-30-6
- Article Data8
- CAS DataBase
- Density 1.1949 (rough estimate)
- Solubility water: 100 g/L in water
- Melting Point 266-272 °C
- Formula C12H28NBr
- Boiling Point
- Molecular Weight 266.265
- Flash Point
- Transport Information
- Appearance white crystals or crystalline powder
- Safety 26-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 1-Propanaminium,N,N,N-tripropyl-, bromide (9CI);Ammonium, tetrapropyl-, bromide (8CI);Tetrapropylammonium bromide (6CI);TPABr;Tetra-n-propylammonium bromide;
- PSA 0.00000
- LogP 0.44720
Synthetic route

| Conditions | Yield |
|---|---|
| In ethanol | |
| In butanone at 80℃; for 96h; | |
| In acetonitrile for 24h; Reflux; | |
| In N,N-dimethyl-formamide at 110℃; for 15h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With tri-n-propylamine In N-methyl-acetamide | |
| With tri-n-propylamine | |
| With tri-n-propylamine In nitromethane |
-
-
1392488-20-8
C6H5O(1-)*C6H6O*C12H28N(1+)

-
-
100-39-0
benzyl bromide

-
A
-
946-80-5
(benzyloxy)benzene

-
B
-
1941-30-6
tetra-n-propylammonium bromide

-
C
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; |


| Conditions | Yield |
|---|---|
| In [D3]acetonitrile at 24.84℃; Equilibrium constant; |

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |


-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |


-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |


-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| With bromine for 0.5h; Schlenk technique; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of Cs3KRe6S8(CN)6*2H2O to aq. mixt. of n-Pr4NBr and Ni(CH3COO)2*4H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 97% |

-
-
224769-15-7
[PdCl2((C6H5)2P(S)CH2P(C6H5)2)]

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
224769-16-8
PdBr2((C6H5)2PCH2P(S)(C6H5)2)

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; excess of bromide, stirring for 1 h; pptn. on vol. reduction, collection (filtration), washing (Et2O), drying(vac.); elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of K4Re6Se8(CN)6*3.5H2O to aq. mixt. of n-Pr4NBr and Ni(CH3COO)2*4H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 95% |


-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of K4Re6Se8(CN)6*3.5H2O to aq. mixt. of n-Pr4NBr and Mn(NO3)2*6H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 95% |


| Conditions | Yield |
|---|---|
| In ethanol; water at 30℃; | 95% |
| In ethanol; water soln. (Pr4N)Br in EtOH was combined with aq. soln. Na(dca) and layered on top soln. Ni(NO3)2*6H2O in water, 2 weeks; ppt. was filtered; elem. anal.; |


| Conditions | Yield |
|---|---|
| In ethanol; water at 30℃; | 95% |


-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of Cs3KRe6S8(CN)6*2H2O to aq. mixt. of n-Pr4NBr and Mn(NO3)2*6H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 93% |


-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In water Inert atmosphere; Heating; | 93% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In methanol; water under N2; addn. of N(Pr-n)4Br in H2O to the Ir compound in MeOH; ppt. filtered off, washed with i-PrOH, and dried in vac.; recrystn. from acetone/propan-2-ol; | 90% |

| Conditions | Yield |
|---|---|
| In methanol (N2); stirring (room temp., 30-60 min); filtn., washing (MeOH, Et2O), drying (vac.); elem. anal.; | 90% |

| Conditions | Yield |
|---|---|
| In ethanol | |
| In butanone at 80℃; for 96h; | |
| In acetonitrile for 24h; Reflux; | |
| In N,N-dimethyl-formamide at 110℃; for 15h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With tri-n-propylamine In N-methyl-acetamide | |
| With tri-n-propylamine | |
| With tri-n-propylamine In nitromethane |
-
-
1392488-20-8
C6H5O(1-)*C6H6O*C12H28N(1+)

-
-
100-39-0
benzyl bromide

-
A
-
946-80-5
(benzyloxy)benzene

-
B
-
1941-30-6
tetra-n-propylammonium bromide

-
C
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; |


| Conditions | Yield |
|---|---|
| In [D3]acetonitrile at 24.84℃; Equilibrium constant; |

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |


-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |


-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |


-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
17148-97-9, 20267-56-5
potassium 1,2-dithiooxalate

| Conditions | Yield |
|---|---|
| In water K-salt in water was added to CrCl3*6H2O in water, the soln. was heated for 30 min at 60°C, chloride was added, the soln. was heated at 80°C for 20 min, ammonium salt was added, the soln. was maintained at 80°C, cooled; filtered, washed with water, methanol, diethyl ether; elem. anal.; | 100% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| With bromine for 0.5h; Schlenk technique; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of Cs3KRe6S8(CN)6*2H2O to aq. mixt. of n-Pr4NBr and Ni(CH3COO)2*4H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 97% |

-
-
224769-15-7
[PdCl2((C6H5)2P(S)CH2P(C6H5)2)]

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
224769-16-8
PdBr2((C6H5)2PCH2P(S)(C6H5)2)

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; excess of bromide, stirring for 1 h; pptn. on vol. reduction, collection (filtration), washing (Et2O), drying(vac.); elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of K4Re6Se8(CN)6*3.5H2O to aq. mixt. of n-Pr4NBr and Ni(CH3COO)2*4H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 95% |


-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of K4Re6Se8(CN)6*3.5H2O to aq. mixt. of n-Pr4NBr and Mn(NO3)2*6H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 95% |


| Conditions | Yield |
|---|---|
| In ethanol; water at 30℃; | 95% |
| In ethanol; water soln. (Pr4N)Br in EtOH was combined with aq. soln. Na(dca) and layered on top soln. Ni(NO3)2*6H2O in water, 2 weeks; ppt. was filtered; elem. anal.; |


| Conditions | Yield |
|---|---|
| In ethanol; water at 30℃; | 95% |


-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In water gradual addn. of aq. soln. of Cs3KRe6S8(CN)6*2H2O to aq. mixt. of n-Pr4NBr and Mn(NO3)2*6H2O, stirring for 2 h at ambient temp.; filtration, washing with water, drying in air; elem. anal.; | 93% |


-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In water Inert atmosphere; Heating; | 93% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In methanol; water under N2; addn. of N(Pr-n)4Br in H2O to the Ir compound in MeOH; ppt. filtered off, washed with i-PrOH, and dried in vac.; recrystn. from acetone/propan-2-ol; | 90% |

| Conditions | Yield |
|---|---|
| In methanol (N2); stirring (room temp., 30-60 min); filtn., washing (MeOH, Et2O), drying (vac.); elem. anal.; | 90% |

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
33155-11-2
(Pr4N)[PtBr3(trimethylstibine)]

| Conditions | Yield |
|---|---|
| In dichloromethane 2 equiv. of Pr4NBr, stirring (10 min); crystn. on ether addn.; | 90% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| With KOH In potassium hydroxide aq. KOH; Schlenk technique, under Ar; KCN (4.6 mmol) added to Mo compound (0.12 mmol) soln., soln. stirred under heating (80°C) for 3 h, soln. filtered, added to Pr4NBr (0.40 mmol); ppt. filtered, washed (H2O), dried; elem. anal.; | 90% |

-
-
10213-10-2
sodium tungstate (VI) dihydrate

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| With HCl In water W-compd. dissolved in H2O, 6 M HCl added to pH=1.2, refluxed for 3 d, cooled to room temp., (C3H7)4NBr in H2O added; precipitated product collected, washed with EtOH, dried in vac., recrystallized from CH3CN, elem. anal.; | 89% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In acetonitrile at 20℃; | 88% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In methanol; water stoich., aq. soln. of metal salt added slowly to a soln. of S compd. (water/methanol) with vigorous stirring (ca. 1 h), filtered into aq. amine,, stirred for 30 min; ppt. filtered, washed (water/methanol, diethyl ether), dried (vac., CaCl2); elem. anal.; | 88% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In acetonitrile mixt. was stirred in CH3CN for 1 h;; was cooled to -20°C for 48 h, colorless crystalline solid was filtered, washed with ether, and dried;; | 87% |
| In methanol formation of insoluble white products;; | 0% |

-
-
107847-82-5
lithium 2,4,6-tri-isopropylbenzenethiolate

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In methanol mixt. was stirred in CH3OH for 15 min, then cooled to -20°C;; ppt. was collected by vac. filtration and dried;; | 87% |


-
-
1493-13-6
trifluorormethanesulfonic acid

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
124618-92-4
dibromo(tri-tert-butylisonitrile)((tert-butylamino)phenylacetylene)tungsten

| Conditions | Yield |
|---|---|
| In dichloromethane under N2 or Ar, CF3SO3H in CH2Cl2 added to W-complex and N(C3H7)4Br in CH2Cl2 at -30°C, stirred for 0.5 h at 25°C, solvent removed; chromd. (silicagel, 0°C, Et2O), solvent removed, pentane added; elem. anal., IR, NMR, mass spectra; | 86% |

-
-
495-18-1
Benzohydroxamic acid

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
11113-50-1
boric acid

-
-
494754-17-5
tetra-n-propylammonium bis(benzohydroxymato)borate

| Conditions | Yield |
|---|---|
| With Ag2O In benzene byproducts: H2O, AgBr; refluxed for 8 h with azeotropic elimination of water; ppt. filtered, extrd. (CH2Cl2), evapd. (vac.); elem. anal.; | 86% |


-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
77121-86-9
(Pr4N)2Ni(WOS3)2

| Conditions | Yield |
|---|---|
| In water; acetonitrile to suspn. Cs2WOS3 in H2O and CH3CN added dropwise soln. Ni(NO3)2*6H2O in H2O and CH3CN, soln. filtered, soln. Pr4NBr in H2O added; ppt. collected and recrystd. from nitromethane; elem. anal.; | 85% |


| Conditions | Yield |
|---|---|
| With sulfur; potassium hydroxide In water at 140℃; for 14h; Green chemistry; | 85% |
-
-
10025-98-6
potassium tetrachloropalladate(II)

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
77071-58-0
(Pr4N)2Pd(WOS3)2

| Conditions | Yield |
|---|---|
| In water; acetonitrile soln. K2PdCl4 in H2O and CH3CN added dropwise to stirred suspn. Cs2WOS3in 1:3 H2O-CH3CN, soln. stirred for 2 h, filtered and soln. Pr4NBr in CH3CN added to filtrate; soln. concd. and stored at 0°C overnight, crystals filtered off and washed with anhyd. ethanol and ether; elem. anal.; | 84% |

-
-
1941-30-6
tetra-n-propylammonium bromide

-
-
1261294-87-4
tetrapropylammonium bromopentachlorophosphoride

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride In acetonitrile at 20℃; | 84% |

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| In water byproducts: KBr, HF; stirring (10 min), pptn.; filtn., washing (H2O), drying (air atm.), dissoln. (CH3CN), crystn.; elem. anal.; | 83% |
-
-
17849-38-6
2-Chlorobenzyl alcohol

-
-
1941-30-6
tetra-n-propylammonium bromide

| Conditions | Yield |
|---|---|
| Stage #1: 2-Chlorobenzyl alcohol With pyridine; sulfur trioxide pyridine complex In dichloromethane at 20 - 40℃; for 19h; Stage #2: tetra-n-propylammonium bromide In water for 1h; | 83% |
Tetrapropylammonium bromide Chemical Properties
Molecular Structure of Tetrapropylammonium bromide (CAS NO.1941-30-6):
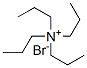 .
.
IUPAC Name: tetrapropylazanium bromide
CAS Registry Number: 1941-30-6
EINECS: 217-727-6
Empirical Formula: C12H28BrN
Molecular Weight: 266.2614
ACD/LogP: -2.64
of Rule of 5 Violations: 0
ACD/LogD (pH 5.5): -2.64
ACD/LogD (pH 7.4): -2.64
ACD/BCF (pH 5.5): 1
ACD/BCF (pH 7.4): 1
ACD/KOC (pH 5.5): 1
ACD/KOC (pH 7.4): 1
H bond acceptors: 1
H bond donors: 0
Freely Rotating Bonds: 8
Polar Surface Area: 0Å2
Melting point: 266-272 oC
Water solubility: 100 g/L
Sensitive: Hygroscopic
Melting point: 266-272 °C
solubility H2O: 0.1 g/mL
Water Solubility: 100 g/L
Sensitive: Hygroscopic
BRN: 3567846
InChI
InChI=1/C12H28N.BrH/c1-5-9-13(10-6-2,11-7-3)12-8-4;/h5-12H2,1-4H3;1H/q+1;/p-1
Smiles
[N+](CCC)(CCC)(CCC)CCC.[BrH-]
Product Categories: quarternary ammonium salts; Ammonium Bromides (Quaternary);Quaternary Ammonium Compounds; Ammonium SaltsAnalytical Reagents; Electrochemistry; Supporting Electrolytes for Electrochemistry; Ammonium Salts; Greener Alternatives: Catalysis; Phase Transfer Catalysts; AnionicHPLC; Chromatography/CE Reagents; Ion Pair; Ion Pair Reagents; Ion Pair Reagents - Anionic
Tetrapropylammonium bromide Toxicity Data With Reference
| 1. | dnd-mam:lym 50 mmol/L | CBINA8 Chemico-Biological Interactions. 19 (1977),197. |
Tetrapropylammonium bromide Consensus Reports
Reported in EPA TSCA Inventory.
Tetrapropylammonium bromide Safety Profile
Mutation data reported. When heated to decomposition it emits very toxic fumes of NOx, NH3, and Br−. See also BROMIDES.
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S37/39:Wear suitable gloves and eye/face protection.
WGK Germany: 3
RTECS: BS8390000
F: 3
HS Code: 29239000
Tetrapropylammonium bromide Specification
Tetrapropylammonium bromide , with CAS number of 1941-30-6, can be called 1-Propanaminium, N,N,N-tripropyl-, bromide ; 1-Propanaminium, N,N,N-tripropyl-, bromide (1:1) ; ammonium compounds, quaternary, tetrapropyl-, bromide . It is a white crystals or crystalline powder. Tetrapropylammonium bromide is hygroscopic. Be incompatible with strong oxidizing agents.
Related Products
- Tetrapropylammonium bisulfate
- Tetrapropylammonium bromide
- Tetrapropylammonium hydroxide
- Tetrapropylammonium iodide
- Tetrapropylammonium perchlorate
- Tetrapropylammonium tetrafluoroborate
- Tetrapropylammonium tetraoxoruthenate
- Tetrapropylammonium trifluoromethanesulphonate
- 194145-29-4
- 1941-52-2
- 194154-40-0
- 19415-51-1
- 194159-22-3
- 194163-91-2
- 19416-70-7
- 19417-00-6
- 194205-01-1
- 19420-57-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View