-
Name
Triisobutyl phosphate
- EINECS 204-798-3
- CAS No. 126-71-6
- Article Data26
- CAS DataBase
- Density 0.982 g/cm3
- Solubility 264mg/L at 25℃
- Melting Point
- Formula C12H27O4P
- Boiling Point 261.2 ºC at 760 mmHg
- Molecular Weight 266.318
- Flash Point 125.8 ºC
- Transport Information
- Appearance Colorless transparent liquid
- Safety 36/37
- Risk Codes 43
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Isobutylphosphate ((C4H9O)3PO) (6CI,7CI);Phosphoric acid, triisobutyl ester (8CI);Antifoam TIP;Daiguard 400;NSC 62222;Reomol TIBP;
- PSA 54.57000
- LogP 4.11230
Synthetic route
-
-
78-83-1
2-methyl-propan-1-ol

-
A
-
126-71-6
triisobutyl phosphate

-
B
-
3846-81-9
diphosphoric acid tetraisobutyl ester

| Conditions | Yield |
|---|---|
| With phosphorous; tetraethylammonium iodide In water at 50℃; Electrolysis; | A 4% B 81% |
-
-
78-83-1
2-methyl-propan-1-ol

-
A
-
1189-24-8
diisobutyl phosphite

-
B
-
6303-30-6
diisobutyl phosphoric acid

-
C
-
126-71-6
triisobutyl phosphate

| Conditions | Yield |
|---|---|
| With sodium hypophosphite; copper dichloride at 24.9℃; for 148h; | A 5.6% B 66.6% C 27.8% |


| Conditions | Yield |
|---|---|
| With pyridine; trichlorophosphate; benzene | |
| With argon; phosphan; copper(l) chloride; copper dichloride In acetonitrile | |
| With argon; phosphan; copper(l) chloride; copper dichloride In acetonitrile at 24.9℃; Rate constant; Thermodynamic data; Product distribution; E(excit.), ΔS(excit.); |
-
-
13259-29-5
sodium isobutoxide

-
-
126-71-6
triisobutyl phosphate

| Conditions | Yield |
|---|---|
| With diethyl ether; trichlorophosphate | |
| With phosphorus trichloride |

| Conditions | Yield |
|---|---|
| With 2-methyl-propan-1-ol; chlorine |
-
-
1606-96-8
triisobutyl phosphite

-
A
-
126-71-6
triisobutyl phosphate

-
B
-
3846-81-9
diphosphoric acid tetraisobutyl ester

| Conditions | Yield |
|---|---|
| With iodoacetophenone; silver perchlorate In benzene for 0.316667h; Title compound not separated from byproducts; |
-
-
17158-87-1
phosphorochloridic acid diisobutyl ester

-
-
78-83-1
2-methyl-propan-1-ol

-
-
126-71-6
triisobutyl phosphate

| Conditions | Yield |
|---|---|
| at 60℃; Rate constant; Thermodynamic data; activation energy, activation enthalpy, other temperature (40, 50, 70, 80 deg C); |

| Conditions | Yield |
|---|---|
| With phosphan; copper dichloride In N,N-dimethyl-formamide at 59.9℃; |

| Conditions | Yield |
|---|---|
| With phosphan; chlorine at 20℃; Rate constant; Thermodynamic data; Ea, ΔS(excit.); var. temp.; also in the presence of pyridine; |
-
-
13529-77-6
phosphonic acid diisobutyl ester

-
A
-
126-71-6
triisobutyl phosphate

-
B
-
75340-94-2
diphosphorus (IV)-oic acid tetraisobutyl ester

-
C
-
3846-81-9
diphosphoric acid tetraisobutyl ester

| Conditions | Yield |
|---|---|
| With sodium perchlorate In acetonitrile electrochemical oxidation, 2.4 A h; Pt anode, Ni cathode; Ag/AgNO3 reference electrode; Title compound not separated from byproducts; |

-
-
7647-01-0
hydrogenchloride

-
-
78-83-1
2-methyl-propan-1-ol

-
-
80937-33-3
oxygen

-
A
-
6303-30-6
diisobutyl phosphoric acid

-
B
-
126-71-6
triisobutyl phosphate

-
C
-
513-36-0
isobutyryl chloride

-
D
-
7646-85-7
zinc(II) chloride

| Conditions | Yield |
|---|---|
| copper dichloride In further solvent(s) Kinetics; |


| Conditions | Yield |
|---|---|
| With O2; CuCl2 In neat (no solvent) Kinetics; byproducts: H2O, HCl; the alcohol was loaded together with CuCl2 into the reactor and purged with an Ar-O2 mixture; then Cu3P was added and the mixt. reacted at 293 K, 323 K or 343 K;; |

-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
126-71-6
triisobutyl phosphate

-
-
10092-77-0
diisobutyl butylphosphonate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; hexane at -78 - 0℃; | 93% |
-
-
126-71-6
triisobutyl phosphate

| Conditions | Yield |
|---|---|
| In acetonitrile (N2 or Ar); ligand added to a soln. of U salt; crystd. overnight at room temp.; elem. anal.; | 87% |
-
-
126-71-6
triisobutyl phosphate

| Conditions | Yield |
|---|---|
| In methanol addn. of phosphorus compd. to a soln. of thorium compd. in methanol; centrifugation, sepn., washing ppt. with methanol, centrifugation, sepn., evapn. of supernatant, drying in vac., dissolving in methanol, filtration, keeping at 4°C for 3 d; elem. anal.; | 75% |

-
-
126-71-6
triisobutyl phosphate

| Conditions | Yield |
|---|---|
| In ethanol soln. of P ligand (2 equiv.) in EtOH was added dropwise to soln. of U compd. in EtOH; evapd.; crystd. (hexane, away from direct sunlight); filtered; dried in air; elem. anal.; | A n/a B 70% |

-
-
302-17-0
chloral hydrate

-
-
126-71-6
triisobutyl phosphate

-
-
38457-66-8
(2,2,2-trichloro-1-hydroxy-ethyl)-phosphonic acid diisobutyl ester

| Conditions | Yield |
|---|---|
| In hexane Heating; | 60% |

| Conditions | Yield |
|---|---|
| In ethanol; water To an aqueous soln. of EuCl3*6H2O was added an ethanolic soln. of pyromellitic acid and triisobutyl phosphate, neutralization with NH3 to pH 6 and warming to 30°C for 30-40 min.; Elem. anal.; | 55% |


-
-
126-71-6
triisobutyl phosphate

| Conditions | Yield |
|---|---|
| In ethanol Irradiation (UV/VIS); allowed to evap. slowly in direct sunlight; decanted; elem. anal.; | 35% |
-
-
126-71-6
triisobutyl phosphate

-
-
134-32-7
1-amino-naphthalene

-
-
109556-56-1
diisobutyl-[1]naphthyl-amine
-
-
126-71-6
triisobutyl phosphate

-
-
91-59-8
naphthalen-2-ylamine

-
-
109554-95-2
diisobutyl-[2]naphthyl-amine

| Conditions | Yield |
|---|---|
| Multistep reaction; |

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; Yield given; | |
| In tetrahydrofuran; hexane at -78 - 0℃; |
-
-
126-71-6
triisobutyl phosphate

-
-
83561-06-2
tris(2-methyl-1-propyl) phosphate radical

| Conditions | Yield |
|---|---|
| In water at 18.9℃; pulse radiolysis, pH 4.5-5; fast hydrolysis of alkyl radicals radiolytically formed; |
-
-
126-71-6
triisobutyl phosphate

-
A
-
2466-73-1
Phosphorsaeure-sec.-butylester

-
B
-
6303-30-6
diisobutyl phosphoric acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 100℃; for 2h; Title compound not separated from byproducts.; |
-
-
126-71-6
triisobutyl phosphate

-
-
112292-29-2
diisobutyl (1-methylbutyl)phosphonate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran; hexane / -78 - 0 °C 2: 89 percent / tetrahydrofuran; hexane / -78 - -20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: diethyl ether / 0 °C 2: 83 percent View Scheme |
-
-
126-71-6
triisobutyl phosphate

-
-
10092-77-0
diisobutyl butylphosphonate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: diethyl ether / 0 °C 2: 73 percent / H3O(1+) View Scheme |
-
-
51108-30-6
4-chloro-1H-isoindole-1,3(2H)-dione

-
-
117-21-5
3-chlorophthalic anhydride

-
-
126-71-6
triisobutyl phosphate

-
-
5329-14-6
aminosulfonic acid

-
A
-
4743-17-3
5-Chloroisatoic anhydride

-
B
-
63497-60-9
8-chloro-2H-benzo[d][1,3]oxazine-2,4(1H)-dione

| Conditions | Yield |
|---|---|
| In water; formamide |

Triisobutyl phosphate Chemical Properties
Molecule structure of Isobutyl phosphate (CAS NO.126-71-6):
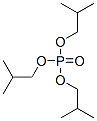
IUPAC Name: Tris(2-methylpropyl) phosphate
Molecular Weight: 266.314141 g/mol
Molecular Formula: C12H27O4P
Density: 0.982 g/cm3
Boiling Point: 261.2 °C at 760 mmHg
Flash Point: 125.8 °C
Index of Refraction: 1.427
Molar Refractivity: 69.62 cm3
Molar Volume: 270.9 cm3
Surface Tension: 29.7 dyne/cm
Enthalpy of Vaporization: 47.88 kJ/mol
Vapour Pressure: 0.0191 mmHg at 25 °C
XLogP3-AA: 3.5
H-Bond Acceptor: 4
Rotatable Bond Count: 9
Exact Mass: 266.164696
MonoIsotopic Mass: 266.164696
Topological Polar Surface Area: 44.8
Heavy Atom Count: 17
Canonical SMILES: CC(C)COP(=O)(OCC(C)C)OCC(C)C
InChI: InChI=1S/C12H27O4P/c1-10(2)7-14-17(13,15-8-11(3)4)16-9-12(5)6/h10-12H,7-9H2,1-6H3
InChIKey: HRKAMJBPFPHCSD-UHFFFAOYSA-N
EINECS: 204-798-3
Classification Code: Skin / Eye Irritant
Triisobutyl phosphate Uses
Isobutyl phosphate (CAS NO.126-71-6) is used as textile auxiliaries, wetting agent and dye auxiliary.
Triisobutyl phosphate Toxicity Data With Reference
| 1. | orl-rat LD50:>5 g/kg | AIHAAP American Industrial Hygiene Association Journal. 34 (1973),286. | ||
| 2. | ihl-rat LC:>122 ppm/6H | AIHAAP American Industrial Hygiene Association Journal. 34 (1973),286. |
Triisobutyl phosphate Consensus Reports
Reported in EPA TSCA Inventory.
Triisobutyl phosphate Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 43
R43:May cause sensitization by skin contact.
Safety Statements: 36/37
S36/37:Wear suitable protective clothing and gloves.
WGK Germany: 1
RTECS: TC9300000
Moderately toxic by inhalation. Low toxicity by ingestion. When heated to decomposition it emits toxic vapors of POx.
Triisobutyl phosphate Specification
Isobutyl phosphate (CAS NO.126-71-6) is also named as 4-01-00-01598 (Beilstein Handbook Reference) ; AI3-07850 ; BRN 1710574 ; NSC 62222 ; Triisobutyl phosphate ; Phosphoric acid, triisobutyl ester (8CI) ; Phosphoric acid, tris(2-methylpropyl) ester .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View