Wuhan Fortuna Chemical Co.,Ltd
Manufacturer supply 5-O-Allyl-2,3,4-tri-O-benzyl-D-ribitol CAS 111549-97-4 with attractive price Company profile Wuhan Fortuna Chemical Co.,Ltd established in 2006, is a big integrative chemical enterprise being engaged in Pharmaceutical & i
Cas:111549-97-4
Min.Order:10 Gram
FOB Price: $10.0
Type:Trading Company
inquiryDayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:111549-97-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.
Cas:111549-97-4
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:111549-97-4
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:111549-97-4
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryNewCan Biotech Limited
NewCan Biotech Limited was established in 2021 and is primarily engaged in the research, development, production, and sales of sugars, nucleosides, nucleotides, phosphorylated monomers, as well as next-generation antiviral and antitumor drug intermed
Cas:111549-97-4
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:111549-97-4
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:111549-97-4
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquirySuzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryWin-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Manufacturers
inquiryAntimex Chemical Limied
TIANFU-CHEM 5-O-Allyl-2,3,4-tri-O-benzyl-D-ribitolAppearance:white to light yellow crystal powder Storage:room temperature Package:as per buyers Application:Industrial Grade Transportation:By sea , by air or express
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Trading Company
inquiryZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Trading Company
inquiryHENAN SUNLAKE ENTERPRISE CORPORATION
Cas:111549-97-4
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHebei youze Biotechnology Co.,Ltd
1: Fast and guaranteed shipment (TNT;EMS;FEDEX;DHL;UPS;EUB, special line) 2: Various payment terms accepted (Btc;MoneyGram;WU) 3: Valued package (Paraffin coating; Double aluminum foil bag; Vacuum packaging) 4: Efficient delivery (3-10 DAYS fast deli
Cas:111549-97-4
Min.Order:1 Kilogram
FOB Price: $9.0 / 12.0
Type:Trading Company
inquiryWatson International Ltd
Watson International Ltd' has a very strong R&D and technical capacity supported by FCAD's platform. The subsidiaries under FCAD Group have accumulated much know-how of different fine chemical branches. For example, Apnoke Scientific L
Cas:111549-97-4
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryJilin haofei import and export trade Co.,Ltd
Price, service, company and transport advantage: 1. Best service, place of origin China, high quality, and reasonable price. 2. It's customers' right to choose the package (EMS, DHL, FEDEX, UPS). 3. It's customers' right
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Trading Company
inquiryChemical Technology Co.,LTD
1. Production capacity: we have three production base with high-tech production equipment and instruments, equipped with professional production personnel, meet your requirements.2. Quality assurance: we have a first-class testing equipment and testi
Shandong Bolode Bio-Technology Co., LTD
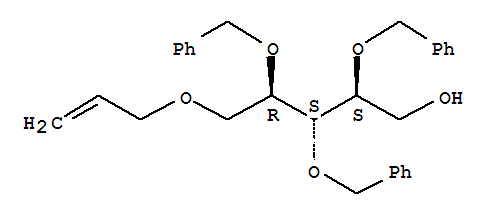
Specification Name 5-O-Allyl-2,3,4-tri-O-benzyl-D-ribitol Cas No, 111549-97-4 Structure Formula Purity 99% Ap
Cas:111549-97-4
Min.Order:1 Gram
Negotiable
Type:Trading Company
inquiryWuhan MoonZY Biological Technology Co.,Ltd
instock with good quality and wholesale price Storage:Keep in a cool & dry place Package:Packing material and QTY as your request Application:Pharma;Industry;other application Transportation:Express or as your request Port:Any port of China
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynova( Tianjin ) Chemical Technology Co,.Ltd
The company have effective management team, professional technical R & D personnel, the service spirit of customer oriented. We have long-term cooperation with famous domestic manufacturer, and excellent customer resources overseas. We are skilled in
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySkyrun Industrial Co.,Ltd
Adequate stocks available,prompt shipment,Strictly control on quality,Timely after-sales Application:Intermediate
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Petrochemical Co.,Ltd
intermediateAppearance:powder Storage:Normal temperature Package:1kg/25kg/customized Application:intermediate Transportation:By Ship/Air
Hangzhou Yierdechem Co. Ltd
intermediateAppearance:powder Storage:Normal temperature Package:1kg/25kg/customized Application:intermediate Transportation:By Ship/Air Port:any port of china
Zhejiang Chemline International Co., Ltd.
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Trading Company
inquiryFinetech Industry Limited
High Quality Best Price Storage:Store in dry, dark and ventilated place Application:Chemical Synthesis Intermediate
Debye Scientific
Debyesci is here who supplied several kinds of chemical products to global pharmaceutical, drug discovery, agrochemical and biotechnology industries for four yearsOur key scientific leadership team has gained experience in top research and developmen
Cas:111549-97-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
118964-50-4
5-O-allyl-2,3,4-tri-O-benzyl-D-ribose

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol; water for 10h; Reduction; | 90% |
| With sodium tetrahydroborate In methanol Yield given; |
-
-
111549-96-3
5-O-allyl-2,3,4-tri-O-benzyl-1-O-trityl-D-ribitol

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| With water; acetic acid at 80℃; for 1.5h; | 90% |
| In water; acetic acid at 80℃; for 1.5h; | 90% |
| With water; acetic acid at 80℃; for 1.5h; |
-
-
100-39-0
benzyl bromide

-
-
116490-75-6
5-O-allyl-2,3-di-O-benzyl-D-ribitol

-
A
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
B
-
116490-76-7
5-O-allyl-1,2,3-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In dichloromethane for 16h; Heating; | A 19% B 56% |

-
-
50-69-1
D-ribose

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 75 percent / conc. aq. HCl / 0.5 h / 0 °C 2.1: BuSnO23 / toluene / 4 h / Heating 2.2: 68 percent / CsF / dimethylformamide / 1 h / 20 °C 3.1: NaH / dimethylformamide / 0.5 h / 0 °C 3.2: 63 percent / dimethylformamide / 3.5 h / 0 °C 4.1: 86 percent / aq. HgCl2; CaCO3 / acetonitrile / 1 h / 20 °C 5.1: 90 percent / NaBH4 / methanol; H2O / 10 h View Scheme |
-
-
7152-47-8
D-ribose diethyl dithioacetal

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: BuSnO23 / toluene / 4 h / Heating 1.2: 68 percent / CsF / dimethylformamide / 1 h / 20 °C 2.1: NaH / dimethylformamide / 0.5 h / 0 °C 2.2: 63 percent / dimethylformamide / 3.5 h / 0 °C 3.1: 86 percent / aq. HgCl2; CaCO3 / acetonitrile / 1 h / 20 °C 4.1: 90 percent / NaBH4 / methanol; H2O / 10 h View Scheme |
-
-
129163-69-5
5-O-allyl D-ribose diethyl dithioacetal

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: NaH / dimethylformamide / 0.5 h / 0 °C 1.2: 63 percent / dimethylformamide / 3.5 h / 0 °C 2.1: 86 percent / aq. HgCl2; CaCO3 / acetonitrile / 1 h / 20 °C 3.1: 90 percent / NaBH4 / methanol; H2O / 10 h View Scheme | |
| Multi-step reaction with 3 steps 1: 91 percent / sodium hydride, tetra-n-butylammonium iodide / tetrahydrofuran / Ambient temperature 2: mercury(II)chloride, mercury(II)oxide / acetone; H2O / 2 h / Ambient temperature 3: sodium borohydride / methanol View Scheme |
-
-
118964-49-1
5-O-allyl 2,3,4-tri-O-benzyl-D-ribose diethyl dithioacetal

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 86 percent / aq. HgCl2; CaCO3 / acetonitrile / 1 h / 20 °C 2: 90 percent / NaBH4 / methanol; H2O / 10 h View Scheme | |
| Multi-step reaction with 2 steps 1: mercury(II)chloride, mercury(II)oxide / acetone; H2O / 2 h / Ambient temperature 2: sodium borohydride / methanol View Scheme |
-
-
30725-00-9
2,3-O-isopropylidene-D-ribonic-γ-lactone

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 40 percent / silver oxide / dimethylformamide / 48 h / in dark 2: 80 percent / sodium borohydride / tetrahydrofuran / 55 °C / 2.) methanol 45 min and 1h 3: acetic acid; H2O / 4 h / 50 °C 4: pyridine / Ambient temperature 5: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 6: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme | |
| Multi-step reaction with 7 steps 1: 85 percent / pyridine / acetonitrile / 0 °C / 1.) 40 min; 2.) 1 h 2: LiAlH4, tetrakis(triphenylphosphine)palladium / dioxane / 0.25 h 3: 80 percent / sodium borohydride / tetrahydrofuran / 55 °C / 2.) methanol 45 min and 1h 4: acetic acid; H2O / 4 h / 50 °C 5: pyridine / Ambient temperature 6: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 7: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme | |
| Multi-step reaction with 6 steps 1: 85 percent / tetrahydrofuran; pyridine / 2 h / 0 °C 2: 100 percent / palladium tetrakis / dioxane / 0.25 h / Heating 3: 80 percent / NaBH4 / tetrahydrofuran; methanol / 2 h / 55 °C 4: 1.) HOAc, H2O / 1.) 50 degC, 4h 2.) pyridine, roomtemp., overnight 5: 86 percent / NaH / dimethylformamide / 1 h / 0 °C 6: 90 percent / HOAc, H2O / 1.5 h / 80 °C View Scheme |
-
-
111549-94-1
5-O-allyl-2,3-O-isopropylidene-D-ribitol

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: acetic acid; H2O / 4 h / 50 °C 2: pyridine / Ambient temperature 3: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 4: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) HOAc, H2O / 1.) 50 degC, 4h 2.) pyridine, roomtemp., overnight 2: 86 percent / NaH / dimethylformamide / 1 h / 0 °C 3: 90 percent / HOAc, H2O / 1.5 h / 80 °C View Scheme |
-
-
111549-93-0
5'-O-allyl-2',3'-O-isopropylidene-D-ribonic-γ-lactone

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 80 percent / sodium borohydride / tetrahydrofuran / 55 °C / 2.) methanol 45 min and 1h 2: acetic acid; H2O / 4 h / 50 °C 3: pyridine / Ambient temperature 4: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 5: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme | |
| Multi-step reaction with 4 steps 1: 80 percent / NaBH4 / tetrahydrofuran; methanol / 2 h / 55 °C 2: 1.) HOAc, H2O / 1.) 50 degC, 4h 2.) pyridine, roomtemp., overnight 3: 86 percent / NaH / dimethylformamide / 1 h / 0 °C 4: 90 percent / HOAc, H2O / 1.5 h / 80 °C View Scheme |
-
-
111549-92-9
5-O-(allyloxycarbonyl)-2,3-O-isopropylidene-D-ribonolactone

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: LiAlH4, tetrakis(triphenylphosphine)palladium / dioxane / 0.25 h 2: 80 percent / sodium borohydride / tetrahydrofuran / 55 °C / 2.) methanol 45 min and 1h 3: acetic acid; H2O / 4 h / 50 °C 4: pyridine / Ambient temperature 5: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 6: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme | |
| Multi-step reaction with 5 steps 1: 100 percent / palladium tetrakis / dioxane / 0.25 h / Heating 2: 80 percent / NaBH4 / tetrahydrofuran; methanol / 2 h / 55 °C 3: 1.) HOAc, H2O / 1.) 50 degC, 4h 2.) pyridine, roomtemp., overnight 4: 86 percent / NaH / dimethylformamide / 1 h / 0 °C 5: 90 percent / HOAc, H2O / 1.5 h / 80 °C View Scheme |
-
-
111549-95-2
5-O-allyl-1-O-trityl-D-ribitol

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 2: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 86 percent / NaH / dimethylformamide / 1 h / 0 °C 2: 90 percent / HOAc, H2O / 1.5 h / 80 °C View Scheme |

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: pyridine / Ambient temperature 2: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 3: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme |
-
-
5336-08-3
D-Ribono-1,4-lactone

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 1.) hydrochloric acid; 2.) pyridine / 1.) 24 h, room temperature; 2.) acetonitrile, 0 deg C 45 min and 0 deg C 1 h 2: LiAlH4, tetrakis(triphenylphosphine)palladium / dioxane / 0.25 h 3: 80 percent / sodium borohydride / tetrahydrofuran / 55 °C / 2.) methanol 45 min and 1h 4: acetic acid; H2O / 4 h / 50 °C 5: pyridine / Ambient temperature 6: 86 percent / 1.) sodium hydride / dimethylformamide / 1.) 0 deg C 30 min; 2.) 0 deg C 30 min; 3.) room temperature overnight 7: 90 percent / acetic acid; H2O / 1.5 h / 80 °C View Scheme | |
| Multi-step reaction with 7 steps 1: 95 percent / H(+) / 24 h / Ambient temperature 2: 85 percent / tetrahydrofuran; pyridine / 2 h / 0 °C 3: 100 percent / palladium tetrakis / dioxane / 0.25 h / Heating 4: 80 percent / NaBH4 / tetrahydrofuran; methanol / 2 h / 55 °C 5: 1.) HOAc, H2O / 1.) 50 degC, 4h 2.) pyridine, roomtemp., overnight 6: 86 percent / NaH / dimethylformamide / 1 h / 0 °C 7: 90 percent / HOAc, H2O / 1.5 h / 80 °C View Scheme |
-
-
114488-50-5
β-5-O-allyl-1-O-methyl-2,3-O,O-isopropylidene-D-ribofuranoside

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 100 percent / aqueous 50percent trifluoroacetic acide / H2O; methanol / 8 h / Heating 2: 58 percent / pulverized potassium hydroxide / dimethylsulfoxide / Ambient temperature 3: 86 percent / 2M hydrochloric acid / dioxane / 8 h / Heating 4: 95 percent / sodium borohydride / ethanol / 2 h / Ambient temperature 5: 19 percent / aqueous 5percent sodium hydroxide, tetrabutylammonium bromide / CH2Cl2 / 16 h / Heating View Scheme | |
| Multi-step reaction with 5 steps 1: water, sulfuric acid / methanol / 2 h / Heating 2: 94 percent / zinc chloride / 0.25 h / Ambient temperature 3: 91 percent / sodium hydride, tetra-n-butylammonium iodide / tetrahydrofuran / Ambient temperature 4: mercury(II)chloride, mercury(II)oxide / acetone; H2O / 2 h / Ambient temperature 5: sodium borohydride / methanol View Scheme |
-
-
4099-85-8
((3aR,4R,6R,6aR)-6-methoxy-2,2-dimethyltetrahydrofuro[3,4-d][1,3]dioxol-4-yl)methanol

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 87 percent / aqueous 50percent sodium hydroxide, tetrabutylammonium bromide / H2O / 6 h 2: 100 percent / aqueous 50percent trifluoroacetic acide / H2O; methanol / 8 h / Heating 3: 58 percent / pulverized potassium hydroxide / dimethylsulfoxide / Ambient temperature 4: 86 percent / 2M hydrochloric acid / dioxane / 8 h / Heating 5: 95 percent / sodium borohydride / ethanol / 2 h / Ambient temperature 6: 19 percent / aqueous 5percent sodium hydroxide, tetrabutylammonium bromide / CH2Cl2 / 16 h / Heating View Scheme | |
| Multi-step reaction with 6 steps 1: 97 percent / sodium hydride, tetra-n-butylammonium iodide / tetrahydrofuran / Ambient temperature 2: water, sulfuric acid / methanol / 2 h / Heating 3: 94 percent / zinc chloride / 0.25 h / Ambient temperature 4: 91 percent / sodium hydride, tetra-n-butylammonium iodide / tetrahydrofuran / Ambient temperature 5: mercury(II)chloride, mercury(II)oxide / acetone; H2O / 2 h / Ambient temperature 6: sodium borohydride / methanol View Scheme |
-
-
116490-68-7, 116490-69-8, 128775-32-6
methyl 5-O-allyl-D-ribofuranoside

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 58 percent / pulverized potassium hydroxide / dimethylsulfoxide / Ambient temperature 2: 86 percent / 2M hydrochloric acid / dioxane / 8 h / Heating 3: 95 percent / sodium borohydride / ethanol / 2 h / Ambient temperature 4: 19 percent / aqueous 5percent sodium hydroxide, tetrabutylammonium bromide / CH2Cl2 / 16 h / Heating View Scheme |
-
-
329006-48-6
5-O-allyl-2,3-di-O-benzyl-β-D-ribofuranose

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 95 percent / sodium borohydride / ethanol / 2 h / Ambient temperature 2: 19 percent / aqueous 5percent sodium hydroxide, tetrabutylammonium bromide / CH2Cl2 / 16 h / Heating View Scheme |
-
-
116490-72-3
(2R,3R,4R,5R)-2-(allyloxymethyl)-3,4-bis(benzyloxy)-5-methoxytetrahydrofuran

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 86 percent / 2M hydrochloric acid / dioxane / 8 h / Heating 2: 95 percent / sodium borohydride / ethanol / 2 h / Ambient temperature 3: 19 percent / aqueous 5percent sodium hydroxide, tetrabutylammonium bromide / CH2Cl2 / 16 h / Heating View Scheme |
-
-
100-39-0
benzyl bromide

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 86 percent / NaH / dimethylformamide / 1 h / 0 °C 2: 90 percent / HOAc, H2O / 1.5 h / 80 °C View Scheme |
-
-
116490-68-7, 116490-69-8, 128775-32-6
Methyl 5-O-allyl-D-ribofuranoside

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 94 percent / zinc chloride / 0.25 h / Ambient temperature 2: 91 percent / sodium hydride, tetra-n-butylammonium iodide / tetrahydrofuran / Ambient temperature 3: mercury(II)chloride, mercury(II)oxide / acetone; H2O / 2 h / Ambient temperature 4: sodium borohydride / methanol View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
824-94-2
p-methoxybenzyl chloride

-
-
230620-30-1
5-O-allyl-2,3,4-tri-O-benzyl-1-O-p-methoxybenzyl-D-ribitol

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.333333h; Metallation; Stage #2: p-methoxybenzyl chloride In N,N-dimethyl-formamide at 20℃; for 4h; Alkylation; | 89% |
-
-
1227612-68-1
O-(3-O-acetyl-2-azido-4-O-benzyl-6-O-tert-butyl-diphenylsilyl-2-deoxy-D-galactopyranosyl) trichloroacetimidate

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
1227613-04-8
5-O-allyl-2,3,4-tri-O-benzyl-1-O-(3-O-acetyl-2-azido-4-O-benzyl-6-O-tert-butyl-diphenylsilyl-2-deoxy-β-D-galactopyranosyl)-D-ribitol

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In acetonitrile at -40℃; | 88% |
| With trimethylsilyl trifluoromethanesulfonate In acetonitrile at -40℃; for 2h; | 81% |
-
-
6974-32-9
1-O-acetyl-2,3,5-tri-O-benzoyl-β-D-ribofuranose

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
111549-98-5
5-O-allyl-1-O-(2',3',5'-tri-O-benzoyl-β-D-ribofuranosyl)-2,3,4-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In 1,2-dichloro-ethane for 3h; Ambient temperature; | 85% |
| With trimethylsilyl trifluoromethanesulfonate; 4 A molecular sieve 1.) 1,2-dichloroethane, 90 min, room temperature; 2.) 1,2-dichloroethane, room temperature 2 h and 3 h; Yield given. Multistep reaction; |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
118964-41-3
1-O-(2-O-Acetyl-3-O-allyl-5-O-benzyl-β-D-ribofuranosyl)-5-O-allyl-2,3,4-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate; 4 A molecular sieve In dichloromethane for 0.0833333h; | 81% |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
111549-98-5
5-O-allyl-1-O-(2',3',5'-tri-O-benzoyl-β-D-ribofuranosyl)-2,3,4-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate; 4 A molecular sieve In dichloromethane for 0.0833333h; | 81% |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
158895-34-2
1-O-(3-O-Allyl-2,5-di-O-benzoyl-β-D-ribofuranosyl)-5-O-allyl-2,3,4-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve; trimethylsilyl trifluoromethanesulfonate In dichloromethane 1.) r.t., 30 min, 2.) 0 deg C; | 80% |

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In acetonitrile at -30 - 10℃; for 3h; Molecular sieve; | 74% |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
13035-61-5
1,2,3,5-tetraacetylribose

-
-
329006-50-0
1-O-(2,3,5-Tri-O-acetyl-β-D-ribofuranosyl)-5-O-allyl-2,3,4-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In 1,2-dichloro-ethane for 0.25h; | 72% |
| Stage #1: 2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol With 4 A molecular sieve; boron trifluoride diethyl etherate In 1,2-dichloro-ethane at 0 - 25℃; Stage #2: 1,2,3,5-tetraacetylribose In 1,2-dichloro-ethane for 3.33333h; |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
329006-50-0
1-O-(2,3,5-Tri-O-acetyl-β-D-ribofuranosyl)-5-O-allyl-2,3,4-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate; 4 A molecular sieve In dichloromethane for 0.0833333h; | 54% |
-
-
124151-37-7
Acetic acid (3R,4R,5R)-4-allyloxy-5-benzyloxymethyl-2-chloro-tetrahydro-furan-3-yl ester

-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
118964-41-3
1-O-(2-O-Acetyl-3-O-allyl-5-O-benzyl-β-D-ribofuranosyl)-5-O-allyl-2,3,4-tri-O-benzyl-D-ribitol

| Conditions | Yield |
|---|---|
| With molecular sieve; silver perchlorate In acetonitrile at -40℃; for 4h; Yield given; |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
98-59-9
p-toluenesulfonyl chloride

| Conditions | Yield |
|---|---|
| In pyridine Ambient temperature; |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
3282-30-2
pivaloyl chloride

-
-
909556-42-9
5-O-allyl-2,3,4-tri-O-benzyl-1-O-pivaloyl-D-ribitol

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 23℃; for 2h; |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1.1: pyridine / 2 h / 0 - 23 °C 2.1: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3.1: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C 4.1: 85 percent / EtN(i-Pr)2 / acetonitrile / 1.5 h / 23 °C 5.1: 90 percent / NaOMe / dioxane; methanol / 72 h / 23 °C 6.1: 61 percent / EtN(i-Pr)2 / 1,2-dichloro-ethane / 2 h / 0 - 23 °C 7.1: tetrazole / acetonitrile / 1 h / 23 °C 7.2: 77 percent / pyridine; iodine / acetonitrile; tetrahydrofuran; H2O 8.1: 88 percent / HOAc; water / CH2Cl2 / 4 h / 23 °C 9.1: NH4OH / methanol / 8 h / Heating 9.2: Dowex 50 x 8-100 (Na(+)) / methanol 9.3: 88 percent / H2 / Pd/C / 2-methyl-propan-2-ol; H2O / 48 h / 23 °C / 10343 Torr View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: pyridine / 2 h / 0 - 23 °C 2.1: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3.1: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C 4.1: 85 percent / EtN(i-Pr)2 / acetonitrile / 1.5 h / 23 °C 5.1: 90 percent / NaOMe / dioxane; methanol / 72 h / 23 °C 6.1: 61 percent / EtN(i-Pr)2 / 1,2-dichloro-ethane / 2 h / 0 - 23 °C 7.1: tetrazole / acetonitrile / 1 h / 23 °C 7.2: 77 percent / pyridine; iodine / acetonitrile; tetrahydrofuran; H2O 8.1: 88 percent / HOAc; water / CH2Cl2 / 4 h / 23 °C 9.1: NH4OH / methanol / 8 h / Heating 9.2: Dowex 50 x 8-100 (Na(+)) / methanol 9.3: 88 percent / H2 / Pd/C / 2-methyl-propan-2-ol; H2O / 48 h / 23 °C / 10343 Torr 10.1: MeOH; Et3N / H2O / 23 °C 10.2: Dowex 50 x 8-100 (Na(+)) / H2O View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
909715-58-8
2,3,4-tri-O-benzyl-1-O-pivaloyl-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: pyridine / 2 h / 0 - 23 °C 2: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: pyridine / 2 h / 0 - 23 °C 2: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
909556-45-2
2,3,4-tri-O-benzyl-5-O-(4,4'-dimethoxytrityl)-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: pyridine / 2 h / 0 - 23 °C 2: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C 4: 85 percent / EtN(i-Pr)2 / acetonitrile / 1.5 h / 23 °C 5: 90 percent / NaOMe / dioxane; methanol / 72 h / 23 °C View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
909556-48-5
[(N-benzyloxycarbonyl)-2-aminoethyl] 2-cyanoethyl (2,3,4-tri-O-benzyl-1-D-ribityl) phosphate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: pyridine / 2 h / 0 - 23 °C 2.1: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3.1: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C 4.1: 85 percent / EtN(i-Pr)2 / acetonitrile / 1.5 h / 23 °C 5.1: 90 percent / NaOMe / dioxane; methanol / 72 h / 23 °C 6.1: 61 percent / EtN(i-Pr)2 / 1,2-dichloro-ethane / 2 h / 0 - 23 °C 7.1: tetrazole / acetonitrile / 1 h / 23 °C 7.2: 77 percent / pyridine; iodine / acetonitrile; tetrahydrofuran; H2O 8.1: 88 percent / HOAc; water / CH2Cl2 / 4 h / 23 °C View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
909556-44-1
2,3,4-tri-O-benzyl-5-O-(4,4'-dimethoxytrityl)-1-O-pivaloyl-D-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: pyridine / 2 h / 0 - 23 °C 2: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C 4: 85 percent / EtN(i-Pr)2 / acetonitrile / 1.5 h / 23 °C View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
909556-46-3
2-cyanoethyl [2,3,4-tri-O-benzyl-5-O-(4,4'-dimethoxytrityl)-1-D-ribityl] N,N-diisopropylphosphoramidite

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: pyridine / 2 h / 0 - 23 °C 2: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C 4: 85 percent / EtN(i-Pr)2 / acetonitrile / 1.5 h / 23 °C 5: 90 percent / NaOMe / dioxane; methanol / 72 h / 23 °C 6: 61 percent / EtN(i-Pr)2 / 1,2-dichloro-ethane / 2 h / 0 - 23 °C View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
909556-47-4
[(N-benzyloxycarbonyl)-2-aminoethyl] 2-cyanoethyl [2,3,4-tri-O-benzyl-5-O-(4,4'-dimethoxytrityl)-1-D-ribityl] phosphate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: pyridine / 2 h / 0 - 23 °C 2.1: (1,5-cyclooctadiene)-bis(Me-diphenylphosphine)Ir F6phosphate / tetrahydrofuran / 4 h / 23 °C 3.1: 4.65 g / HgO; HgCl2 / acetone; H2O / 0.5 h / 23 °C 4.1: 85 percent / EtN(i-Pr)2 / acetonitrile / 1.5 h / 23 °C 5.1: 90 percent / NaOMe / dioxane; methanol / 72 h / 23 °C 6.1: 61 percent / EtN(i-Pr)2 / 1,2-dichloro-ethane / 2 h / 0 - 23 °C 7.1: tetrazole / acetonitrile / 1 h / 23 °C 7.2: 77 percent / pyridine; iodine / acetonitrile; tetrahydrofuran; H2O View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
230635-00-4
2,3,4-tri-O-benzyl-5-O-p-methoxybenzyl-L-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: NaH / dimethylformamide / 0.33 h / 0 °C 1.2: 89 percent / dimethylformamide / 4 h / 20 °C 2.1: 69 percent / 10 percent Pd/C / methanol; H2O / 10 h / 60 °C View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
230620-43-6
1-O-(2-azido-4,6-O-benzylidene-2-deoxy-3-O-methyl-β-D-galactopyranosyl) 2,3,4-tri-O-benzyl-L-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: NaH / dimethylformamide / 0.33 h / 0 °C 1.2: 89 percent / dimethylformamide / 4 h / 20 °C 2.1: 69 percent / 10 percent Pd/C / methanol; H2O / 10 h / 60 °C 3.1: 16 percent / TMSOTf / acetonitrile / 0.67 h / -50 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: NaH / dimethylformamide / 0.33 h / 0 °C 1.2: 89 percent / dimethylformamide / 4 h / 20 °C 2.1: 69 percent / 10 percent Pd/C / methanol; H2O / 10 h / 60 °C 3.1: 17 percent / TMSOTf / acetonitrile / 0.67 h / -50 °C 4.1: 63 percent / DDQ / CH2Cl2; H2O / 4 h View Scheme |
-
-
111549-97-4
2,3,4-Tris-O-(phenylmethyl)-5-O-(prop-2-enyl)-D-ribitol

-
-
230620-42-5
1-O-(2-azido-4,6-O-benzylidene-2-deoxy-3-O-methyl-β-D-galactopyranosyl) 5-O-p-methoxybenzyl-2,3,4-tri-O-benzyl-L-ribitol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: NaH / dimethylformamide / 0.33 h / 0 °C 1.2: 89 percent / dimethylformamide / 4 h / 20 °C 2.1: 69 percent / 10 percent Pd/C / methanol; H2O / 10 h / 60 °C 3.1: 17 percent / TMSOTf / acetonitrile / 0.67 h / -50 °C View Scheme |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View