Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:185517-21-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:185517-21-9
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:185517-21-9
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:185517-21-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
(R)-2-PROPYLOCTANOIC ACID CAS:185517-21-9 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organi
Cas:185517-21-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:185517-21-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Cas:185517-21-9
Min.Order:0
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:185517-21-9
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:185517-21-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryGolden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Hebei Mojin Biotechnology Co.,Ltd
1, High quality with competitive price:2, Fast and safe delivery3.Excellent pre-sales and after-sales service4. Well-trained and professional technologist and sales with rich experience in the field for 5-10 yearsAppearance:see detailed specification
Research Peptide Biotechnology Co., Ltd.
High purity, high success rate, short cycle and moderate priceAppearance:White powder solid Storage:Negative 20 degrees Celsius Package:5mg, 10mg 100mg, 1gram Application:Applied to various scientific research
Cas:185517-21-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM is one of China's leading providers of integrated fine chemical services including offering, research and development, Custom manufacturing business, as well as other Value-added customer services, for diversified range products of chemicals
Cas:185517-21-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXi`an Eastling Biotech Co., Ltd.
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:Beijing or Guangzhou
Cas:185517-21-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Henan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:185517-21-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers2. Produce products of different specifications and sizes according to your requirements.3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures sta
Cas:185517-21-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
HuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of
wuxi leji biology technology co., LTD
we a state-level key high-tech enterprises in Yixing Environmental Science and Technology High-tech Development Zone. The company, China Agricultural University, Chinese Academy of Agricultural Sciences, Institute of Ecological Science Park, Beijing
MENGNA
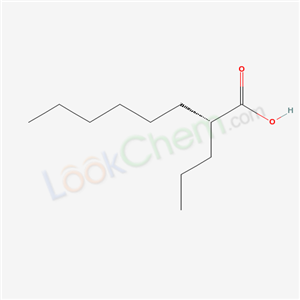
Molecule structure of Arundic acid (CAS NO.185517-21-9): IUPAC Name: (2R)-2-Propyloctanoic acid Molecular Weight: 186.29118 g/mol Molecular Formula: C11H22O2 Density: 0.908 g/cm3 Boiling Point: 289.3 °C at 760 mmHg Flash Point: 154
Cas:185517-21-9
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
213914-70-6
(2S)-2-(2-propenyl)octanoic acid

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With hydrogen; platinum on activated charcoal In isopropyl alcohol under 7600 Torr; for 1.5h; Catalytic hydrogenation; | 99% |
| With hydrogenchloride; sodium chloride; hydrogen; platinum In hexane; ethyl acetate; isopropyl alcohol | |
| With hydrogen; palladium 10% on activated carbon In methanol; ethyl acetate at 20℃; for 1h; | n/a |
| With platinum on carbon; hydrogen In isopropyl alcohol at 30℃; under 3750.38 Torr; for 3.5h; Autoclave; Large scale; | 1045 g |
-
-
1333204-12-8
(R)-2-propyloctan-1-ol

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Stage #1: (R)-2-propyloctan-1-ol With sodium hypochlorite; sodium chlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In water; acetonitrile at 35℃; for 7h; pH=6.7; Inert atmosphere; aq. phosphate buffer; Stage #2: With sodium hydrogencarbonate; sodium sulfite In water; acetonitrile at 0℃; pH=8; Inert atmosphere; Stage #3: With hydrogenchloride In water pH=2; Inert atmosphere; | 98% |
| With sodium hypochlorite solution; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium chlorite In aq. phosphate buffer; acetonitrile at 35℃; for 7h; Inert atmosphere; | 98% |
| With sodium hypochlorite; sodium chlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In aq. phosphate buffer; acetonitrile at 35℃; for 7h; pH=6.7; | 95% |
| With sodium hypochlorite; sodium chlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In aq. phosphate buffer; water; acetonitrile at 35℃; for 7h; pH=6.7; | 95% |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 20℃; for 0.166667h; | 97.1% |
-
-
185463-37-0
(2S)-2-(2-propynyl)octanoic acid

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethyl acetate at 20℃; for 7305h; | 97% |
| With hydrogen; platinum on activated charcoal In isopropyl alcohol under 3800 Torr; for 2.5h; Catalytic hydrogenation; | 94.6% |
| With hydrogen; 5%-palladium/activated carbon In monoethylene glycol diethyl ether; water under 3800.26 Torr; for 5h; | 89% |
| palladium In methanol; ethyl acetate | |
| With hydrogenchloride; sodium chloride; hydrogen; palladium In 1,2-dimethoxyethane; hexane; ethyl acetate |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen In methanol under 3040.2 Torr; for 2h; | 96% |
-
-
1192553-22-2
(S)-2-[(E)-1-propenyl]-(Z)-oct-4-enoic acid

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; under 3040.2 Torr; for 2h; | 96% |
-
-
1087312-90-0
(1R)-2-endo-[(2R)-propyloctanoyl]-1,7,7-trimethylbicyclo[2.2.1]heptan-2-ol

-
A
-
464-49-3
(1R,4R)-1,7,7-trimethylbicyclo[2.2.1]heptan-2-one

-
B
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In water; acetonitrile at 0℃; for 1h; | A n/a B 95% |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With 1‐methyl‐2‐azaadamantane‐N‐oxyl; sodium hypochlorite; sodium chlorite In aq. phosphate buffer; acetone at 20℃; for 3h; pH=6.8; | 92% |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With ferric nitrate In 1,4-dioxane for 20h; Heating; | 60% |
-
-
213914-72-8
N-[(2S)-2-propyloctanoyl]-(1S)-(-)-10,2-camphorsultam

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With tetra(n-butyl)ammonium hydroxide; dihydrogen peroxide In 1,2-dimethoxyethane; water at -20℃; for 0.833333h; | 59% |
| With tetra(n-butyl)ammonium hydroxide; dihydrogen peroxide In tetrahydrofuran at -20℃; for 0.833333h; Hydrolysis; | 59.3% |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Stage #1: N-(2R-(2-propyl)octanoyl)-(1S)-(-)-2,10-camphorsultam With tetra(n-butyl)ammonium hydroxide; dihydrogen peroxide In tetrahydrofuran; water at -20℃; for 0.833333h; Stage #2: With sodium sulfite In tetrahydrofuran; water at 20℃; for 0.5h; | 59.3% |

-
A
-
807363-10-6
(+)-(S)-2-propyloctanoic acid

-
B
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Stage #1: (1,2:5,6-di-O-isopropylidene-α-D-glucofuranos-3-O-yl) 2-hexylpent-3-enoate With hydrogen; platinum(IV) oxide In diethyl ether at 20℃; under 760 Torr; for 12h; Stage #2: With lithium hydroxide; dihydrogen peroxide In methanol; water for 6h; Title compound not separated from byproducts; | |
| Stage #1: (1,2:5,6-di-O-isopropylidene-α-D-glucofuranos-3-O-yl) 2-hexylpent-3-enoate With hydrogen; platinum(IV) oxide In diethyl ether at 20℃; under 760 Torr; for 12h; Stage #2: With titanium(IV) isopropylate; benzyl alcohol In toluene for 7h; Heating; Stage #3: With hydrogen; palladium on activated charcoal In methanol at 20℃; under 760 Torr; for 6h; Title compound not separated from byproducts; |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: H2 / 10 percent Pd/C / methanol / 1 h 2: 60 percent / aq. Fe(NO3)3 / dioxane / 20 h / Heating View Scheme |
-
-
141341-55-1
N-octanoyl-(1S)-(-)-10,2-camphorsultam

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: LDA / tetrahydrofuran; hexane / 0.5 h / -78 °C 1.2: 85.3 percent / LiI / tetrahydrofuran; hexane; various solvent(s) / 3.5 h / -78 - -30 °C 2.1: 89.6 percent / 2-methyl-2-butene; H2O2; tetrabutylammonium hydroxide / 1,2-dimethoxy-ethane; H2O / 0.17 h / -10 °C 3.1: 94.6 percent / H2 / Pt/C / propan-2-ol / 2.5 h / 3800 Torr View Scheme | |
| Multi-step reaction with 3 steps 1.1: LDA / tetrahydrofuran; hexane / 0.5 h / -78 °C 1.2: 71.7 percent / LiI / tetrahydrofuran; hexane; various solvent(s) / 6 h / -78 - 0 °C 2.1: 100 percent / H2 / Pd/C / ethyl acetate; methanol / 1 h 3.1: 59.3 percent / aq. H2O2; tetrabutylammonium hydroxide / tetrahydrofuran / 0.83 h / -20 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: LDA / tetrahydrofuran; hexane / 0.5 h / -78 °C 1.2: 71.7 percent / LiI / tetrahydrofuran; hexane; various solvent(s) / 6 h / -78 - 0 °C 2.1: 82.2 percent / 2-methyl-2-butene; H2O2; tetrabutylammonium hydroxide / 1,2-dimethoxy-ethane; H2O / 2 h / -10 °C 3.1: 99 percent / H2 / Pt/C / propan-2-ol / 1.5 h / 7600 Torr View Scheme | |
| Multi-step reaction with 3 steps 1.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.5 h / -60 °C / Large scale 1.2: 4 h / -65 - 5 °C / Large scale 2.1: 2-methyl-but-2-ene; dihydrogen peroxide; tetra(n-butyl)ammonium hydroxide / water; 1,2-dimethoxyethane / -10 - 5 °C 3.1: platinum on carbon; hydrogen / isopropyl alcohol / 3.5 h / 30 °C / 3750.38 Torr / Autoclave; Large scale View Scheme | |
| Multi-step reaction with 3 steps 1.1: lithium cyclohexylisopropylamide / tetrahydrofuran 1.2: -5 - 5 °C 2.1: 2-methyl-but-2-ene; dihydrogen peroxide; tetra(n-butyl)ammonium hydroxide / water; 1,2-dimethoxyethane / -10 - 5 °C 3.1: platinum on carbon; hydrogen / isopropyl alcohol / 3.5 h / 30 °C / 3750.38 Torr / Autoclave; Large scale View Scheme |
-
-
111-64-8
n-octanoic acid chloride

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: 98.6 percent / Et3N; 4-dimethylaminopyridine / tetrahydrofuran / 1 h / 0 °C 2.1: LDA / tetrahydrofuran; hexane / 0.5 h / -78 °C 2.2: 85.3 percent / LiI / tetrahydrofuran; hexane; various solvent(s) / 3.5 h / -78 - -30 °C 3.1: 89.6 percent / 2-methyl-2-butene; H2O2; tetrabutylammonium hydroxide / 1,2-dimethoxy-ethane; H2O / 0.17 h / -10 °C 4.1: 94.6 percent / H2 / Pt/C / propan-2-ol / 2.5 h / 3800 Torr View Scheme | |
| Multi-step reaction with 4 steps 1.1: 98.6 percent / Et3N; 4-dimethylaminopyridine / tetrahydrofuran / 1 h / 0 °C 2.1: LDA / tetrahydrofuran; hexane / 0.5 h / -78 °C 2.2: 71.7 percent / LiI / tetrahydrofuran; hexane; various solvent(s) / 6 h / -78 - 0 °C 3.1: 100 percent / H2 / Pd/C / ethyl acetate; methanol / 1 h 4.1: 59.3 percent / aq. H2O2; tetrabutylammonium hydroxide / tetrahydrofuran / 0.83 h / -20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: 98.6 percent / Et3N; 4-dimethylaminopyridine / tetrahydrofuran / 1 h / 0 °C 2.1: LDA / tetrahydrofuran; hexane / 0.5 h / -78 °C 2.2: 71.7 percent / LiI / tetrahydrofuran; hexane; various solvent(s) / 6 h / -78 - 0 °C 3.1: 82.2 percent / 2-methyl-2-butene; H2O2; tetrabutylammonium hydroxide / 1,2-dimethoxy-ethane; H2O / 2 h / -10 °C 4.1: 99 percent / H2 / Pt/C / propan-2-ol / 1.5 h / 7600 Torr View Scheme |
-
-
213914-74-0
N-[(2S)-2-hexyl-4-pentynoyl]-(1S)-(-)-10,2-camphorsultam

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 89.6 percent / 2-methyl-2-butene; H2O2; tetrabutylammonium hydroxide / 1,2-dimethoxy-ethane; H2O / 0.17 h / -10 °C 2: 94.6 percent / H2 / Pt/C / propan-2-ol / 2.5 h / 3800 Torr View Scheme |
-
-
213914-68-2
N-(2S)-(2-hexyl-4-pentenoyl)-(1S)-(-)-10,2-camphorsultam

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 100 percent / H2 / Pd/C / ethyl acetate; methanol / 1 h 2: 59.3 percent / aq. H2O2; tetrabutylammonium hydroxide / tetrahydrofuran / 0.83 h / -20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 82.2 percent / 2-methyl-2-butene; H2O2; tetrabutylammonium hydroxide / 1,2-dimethoxy-ethane; H2O / 2 h / -10 °C 2: 99 percent / H2 / Pt/C / propan-2-ol / 1.5 h / 7600 Torr View Scheme | |
| Multi-step reaction with 2 steps 1: 2-methyl-but-2-ene; dihydrogen peroxide; tetra(n-butyl)ammonium hydroxide / water; 1,2-dimethoxyethane / -10 - 5 °C 2: platinum on carbon; hydrogen / isopropyl alcohol / 3.5 h / 30 °C / 3750.38 Torr / Autoclave; Large scale View Scheme |
-
-
807362-94-3
(2R)-2-propyloctanamide

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water; acetic acid at 130℃; for 10h; | n/a |
| With methanesulfonic acid In acetic acid at 105 - 112℃; for 13h; Product distribution / selectivity; | n/a |
| With methanesulfonic acid; acetic acid at 40 - 105℃; for 13h; |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Stage #1: With potassium hydroxide In water at 20℃; for 0.166667h; Stage #2: With hydrogenchloride In n-heptane; Isopropyl acetate; water Product distribution / selectivity; | n/a |
-
-
1333204-09-3
(R)-2-(benzo-1,3-dithiol-2-yl)octan-1-ol

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sodium hydride / tetrahydrofuran; mineral oil / 0 °C / Inert atmosphere 1.2: 18 h / 20 °C / Inert atmosphere 2.1: n-butyllithium / tetrahydrofuran; hexane / 0 °C / Inert atmosphere 2.2: 0.08 h / Inert atmosphere 3.1: hydrogen / ethanol / 3 h / 760.05 Torr / Inert atmosphere 3.2: Inert atmosphere 4.1: sodium chlorite; sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical / water; acetonitrile / 7 h / 35 °C / pH 6.7 / Inert atmosphere; aq. phosphate buffer 4.2: 0 °C / pH 8 / Inert atmosphere 4.3: pH 2 / Inert atmosphere View Scheme |
-
-
1333204-10-6
2-((R)-1-(benzyloxy)octan-2-yl)benzo-1,3-dithiole

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: n-butyllithium / tetrahydrofuran; hexane / 0 °C / Inert atmosphere 1.2: 0.08 h / Inert atmosphere 2.1: hydrogen / ethanol / 3 h / 760.05 Torr / Inert atmosphere 2.2: Inert atmosphere 3.1: sodium chlorite; sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical / water; acetonitrile / 7 h / 35 °C / pH 6.7 / Inert atmosphere; aq. phosphate buffer 3.2: 0 °C / pH 8 / Inert atmosphere 3.3: pH 2 / Inert atmosphere View Scheme |
-
-
1333204-11-7
2-((R)-1-(benzyloxy)octan-2-yl)-2-ethylbenzo-1,3-dithiole

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: hydrogen / ethanol / 3 h / 760.05 Torr / Inert atmosphere 1.2: Inert atmosphere 2.1: sodium chlorite; sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical / water; acetonitrile / 7 h / 35 °C / pH 6.7 / Inert atmosphere; aq. phosphate buffer 2.2: 0 °C / pH 8 / Inert atmosphere 2.3: pH 2 / Inert atmosphere View Scheme |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: sodium tetrahydroborate / methanol / 0.5 h / 0 °C / Inert atmosphere 2.1: sodium hydride / tetrahydrofuran; mineral oil / 0 °C / Inert atmosphere 2.2: 18 h / 20 °C / Inert atmosphere 3.1: n-butyllithium / tetrahydrofuran; hexane / 0 °C / Inert atmosphere 3.2: 0.08 h / Inert atmosphere 4.1: hydrogen / ethanol / 3 h / 760.05 Torr / Inert atmosphere 4.2: Inert atmosphere 5.1: sodium chlorite; sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical / water; acetonitrile / 7 h / 35 °C / pH 6.7 / Inert atmosphere; aq. phosphate buffer 5.2: 0 °C / pH 8 / Inert atmosphere 5.3: pH 2 / Inert atmosphere View Scheme |
-
-
124-13-0
Octanal

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: (R)-5-benzyl-2,2,3-trimethylimidazolidin-4-one; sodium dihydrogenphosphate; benzoic acid / water; acetonitrile / 24 h / 0 °C / Inert atmosphere 2.1: sodium tetrahydroborate / methanol / 0.5 h / 0 °C / Inert atmosphere 3.1: sodium hydride / tetrahydrofuran; mineral oil / 0 °C / Inert atmosphere 3.2: 18 h / 20 °C / Inert atmosphere 4.1: n-butyllithium / tetrahydrofuran; hexane / 0 °C / Inert atmosphere 4.2: 0.08 h / Inert atmosphere 5.1: hydrogen / ethanol / 3 h / 760.05 Torr / Inert atmosphere 5.2: Inert atmosphere 6.1: sodium chlorite; sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical / water; acetonitrile / 7 h / 35 °C / pH 6.7 / Inert atmosphere; aq. phosphate buffer 6.2: 0 °C / pH 8 / Inert atmosphere 6.3: pH 2 / Inert atmosphere View Scheme |
-
-
1192553-20-0
(2S,3S,4S)-1-benzyloxy-4-[(E)-1-propenyl]-(Z)-dec-6-ene-2,3-diol

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium periodate; sodium hydrogencarbonate / dichloromethane; water / 6 h / 20 °C 2: sodium chlorite; sodium dihydrogen phosphate monohydrate; cyclohexene / water; tert-butyl alcohol / 12 h / 20 °C 3: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C / 3040.2 Torr View Scheme |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium chlorite; sodium dihydrogen phosphate monohydrate; cyclohexene / water; tert-butyl alcohol / 12 h / 20 °C 2: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C / 3040.2 Torr View Scheme |

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; lithium hydroxide In tetrahydrofuran; water at 0 - 20℃; | 1.65 g |

| Conditions | Yield |
|---|---|
| In 1,2-dimethoxyethane; water at 60℃; for 0.166667 - 0.416667h; Product distribution / selectivity; | 100% |
| In isopropyl alcohol; acetonitrile at 60℃; for 0.166667 - 0.416667h; Product distribution / selectivity; | 85% |
| In water; acetonitrile at 60℃; for 0.166667 - 0.416667h; Product distribution / selectivity; | 79% |
-
-
3886-69-9
(R)-1-phenyl-ethyl-amine

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| In acetonitrile at 70℃; for 0.25h; | 40% |
-
-
185517-21-9
(R)-(-)-arundic acid

-
-
848899-76-3
(2R)-2-propyloctanoic acid, sodium salt

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 20℃; for 2h; | |
| With sodium hydroxide In water | |
| Stage #1: (R)-(-)-arundic acid With sodium hydroxide In tetrahydrofuran; water at 25 - 50℃; for 24h; Stage #2: With calcium chloride In tetrahydrofuran; water at 50℃; for 2h; Solvent; Temperature; |
-
-
70-11-1
α-bromoacetophenone

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 5h; | n/a |
| With triethylamine In dichloromethane | |
| With potassium carbonate In acetone at 20℃; for 1h; |
-
-
185517-21-9
(R)-(-)-arundic acid

-
-
888038-78-6
lithium (2R)-2-propyloctanoate

| Conditions | Yield |
|---|---|
| With lithium hydroxide In ethanol; water for 0.5h; |
-
-
885020-48-4
(2Z)-4-{[tert-butyl(dimethyl)silyl]oxy}-3-(3,4-difluorophenyl)-2-[4-(methylsulfonyl)phenyl]but-2-en-1-ol

-
-
185517-21-9
(R)-(-)-arundic acid

-
-
888010-86-4
(2Z)-4-{[tert-butyl(dimethyl)silyl]oxy}-3-(3,4-difluorophenyl)-2-[4-(methylsulfonyl)phenyl]but-2-en-1-yl (2R)-2-propyloctanoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 3h; |
-
-
2627-86-3
(S)-1-phenyl-ethylamine

-
-
185517-21-9
(R)-(-)-arundic acid

| Conditions | Yield |
|---|---|
| Stage #1: (R)-(-)-arundic acid With oxalyl dichloride In dichloromethane at 0℃; for 1h; Stage #2: (S)-1-phenyl-ethylamine With triethylamine In dichloromethane at 0℃; for 1h; Further stages.; |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View