Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% ----------------------------------------------------------------
Cas:26048-05-5
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:26048-05-5
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryPribolab PTE.LTD
High purity, all the standards are around 99%; NRM, HPLC, Lc-MS/MS methods valued and certified, ensure the accurate purity; All the standards attached COA and MSDS; More than 80 kinds of mycotoxin standards can be provided; Accurate stock; Comp
Cas:26048-05-5
Min.Order:1 Milligram
FOB Price: $5.0 / 10.0
Type:Trading Company
inquiryCOLORCOM LTD.
Colorcom is a global leader in industrial chemical manufacturing and is continuously innovating and transforming to exceed client expectations and industry standards. Colorcom prides itself on superior customer and technical focus, while focusing on
Henan Tianfu Chemical Co., Ltd.
TIANFUCHEM-- 26048-05-5--High purity BEAUVERICIN in stock Our company was built in 2009 with an ISO certificate.In the past 10 years, we have grown up as a famous fine chemicals supplier in China And we had established stable business r
Cas:26048-05-5
Min.Order:1 Metric Ton
FOB Price: $2000.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1, High quality with competitive price: 1) Standard:BP/USP/EP/Enterprise standard 2) All Purity≥99% 3) We are manufacturer and can provide high quality products with factory price. 2, Fast and safe delivery 1) Parcel can be
Cas:26048-05-5
Min.Order:1 Kilogram
FOB Price: $45.0 / 60.0
Type:Trading Company
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine chemicals, ph
Qingdao Beluga Import and Export Co., LTD
BEAUVERICIN CAS:26048-05-5 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates
Cas:26048-05-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Hubei CuiRan Biotechnology Co., Ltd
Hubei CuiRan Biotechnology Co., Ltd is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medicine)
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:26048-05-5
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Beauvericin CAS No.:26048-05-5 Molecule Formula:C45H57N3O9 Molecule Weight:783.95 Purity: 96.0% Package: 25kg/drum Description:White powder Manufacture Standards:Enterprise Standard TESTING ITEMS SPE
Cas:26048-05-5
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:26048-05-5
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:26048-05-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:26048-05-5
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquiryTriumph International Development Limilted
Triumph has the complete production of G- KG - MT service chain,we can make the new technology into productivity quickly in the research and development of new products. Main Business Custom Synthesis:
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:26048-05-5
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
1, High quality with competitive price:2, Fast and safe delivery3.Excellent pre-sales and after-sales service4. Well-trained and professional technologist and sales with rich experience in the field for 5-10 yearsAppearance:see detailed specification
Cas:26048-05-5
Min.Order:0
Negotiable
Type:Trading Company
inquiryResearch Peptide Biotechnology Co., Ltd.
High purity, high success rate, short cycle and moderate priceAppearance:White powder solid Storage:Negative 20 degrees Celsius Package:5mg, 10mg 100mg, 1gram Application:Applied to various scientific research
Cas:26048-05-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHubei Vanz Pharm Co.,Ltd
ISO/factory/goodqualityAppearance:off white Storage:Dry,cool place Package:drum Application:active pharmaceutical ingredients Transportation:by air/sea/express Port:shenzhen/shanghai
GIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:26048-05-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:26048-05-5
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice.pls contact with us freely for getting detailed product spec
Hangzhou Huarong Pharm Co., Ltd.
High qualityAppearance:White powder Storage:Room temperature Package:Aluminum bag Application:Used in producing API Transportation:By air Port:Beijing
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hubei Langyou International Trading Co., Ltd
Beauveria bassiana CAS NO.26048-05-5 Application:Beauveria bassiana CAS NO.26048-05-5
Henan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Synthetic route

-
-
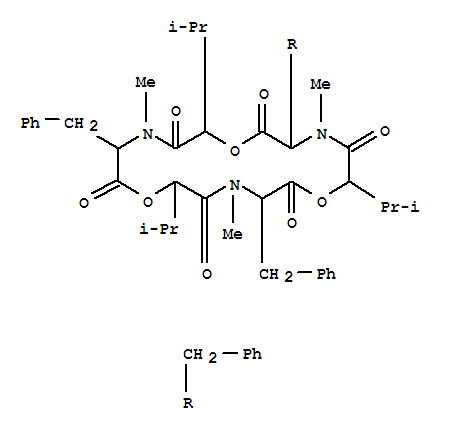
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 2: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 3: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 4: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |
-
-
17407-56-6
(R)-2-Hydroxy-3-methylbutyric acid

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: caesium carbonate / N,N-dimethyl-formamide / 0.67 h / 0 °C / Inert atmosphere 1.2: 15 h / 0 - 20 °C / Inert atmosphere 2.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 3.1: hydrogenchloride / 1,4-dioxane / 3 h / 20 °C / Inert atmosphere 4.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 5.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 6.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 7.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 8.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 9.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 10.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 11 steps 1.1: caesium carbonate / N,N-dimethyl-formamide / 0.67 h / 0 °C / Inert atmosphere 1.2: 15 h / 0 - 20 °C / Inert atmosphere 2.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 3.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 4.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 5.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 6.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 7.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 8.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 9.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 10.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 11.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 9 steps 1.1: caesium carbonate / N,N-dimethyl-formamide / 0.67 h / 0 °C / Inert atmosphere 1.2: 15 h / 0 - 20 °C / Inert atmosphere 2.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 3.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 4.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 5.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 6.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 7.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 8.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 9.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |
-
-
640-68-6
D-Val-OH

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: sulfuric acid; sodium nitrite / water / 60 °C / 5171.62 Torr / Inert atmosphere 2.1: caesium carbonate / N,N-dimethyl-formamide / 0.67 h / 0 °C / Inert atmosphere 2.2: 15 h / 0 - 20 °C / Inert atmosphere 3.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 4.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 5.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 6.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 7.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 8.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 9.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 10.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 11 steps 1.1: sulfuric acid; sodium nitrite / water / 60 °C / 5171.62 Torr / Inert atmosphere 2.1: caesium carbonate / N,N-dimethyl-formamide / 0.67 h / 0 °C / Inert atmosphere 2.2: 15 h / 0 - 20 °C / Inert atmosphere 3.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 4.1: hydrogenchloride / 1,4-dioxane / 3 h / 20 °C / Inert atmosphere 5.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 6.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 7.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 8.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 9.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 10.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 11.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 12 steps 1.1: sulfuric acid; sodium nitrite / water / 60 °C / 5171.62 Torr / Inert atmosphere 2.1: caesium carbonate / N,N-dimethyl-formamide / 0.67 h / 0 °C / Inert atmosphere 2.2: 15 h / 0 - 20 °C / Inert atmosphere 3.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 4.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 5.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 6.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 7.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 8.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 9.1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 10.1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 11.1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 12.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |
-
-
2441-07-8, 65138-05-8, 14487-24-2
benzyl (R)-2-hydroxy-3-methylbutanoate

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 2: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 3: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 4: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 5: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 6: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 7: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 8: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 9: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 10: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 8 steps 1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 2: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 3: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 4: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 5: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 6: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 7: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 8: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 9 steps 1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap / dichloromethane / 0 - 20 °C / Inert atmosphere 2: hydrogenchloride / 1,4-dioxane / 3 h / 20 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 4: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 5: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 6: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 7: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 8: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 9: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 2: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 4: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 5: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 6: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 7: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 2: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 3: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 4: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 5: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 6: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 8 steps 1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 2: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 3: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 4: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 5: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 6: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 7: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 8: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 2: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 3: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 4: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 5: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 2: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 2: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 2: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 3: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 4: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 5: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 7 steps 1: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 2: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 4: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 5: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 6: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 7: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 2: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 4: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 5: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 6: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 7: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 8 steps 1: hydrogenchloride / 1,4-dioxane / 3 h / 20 °C / Inert atmosphere 2: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 3: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 4: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 5: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 6: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 7: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 8: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 9 steps 1: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 2: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.33 h / 0 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / dichloromethane / 17 h / 0 - 20 °C / Inert atmosphere 4: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 5: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 6: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 7: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 8: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 9: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 18.5h; Inert atmosphere; | 46 mg |

-
-
26048-05-5
beauvericin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 2: N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere 3: hydrogen; palladium 10% on activated carbon / tetrahydrofuran / 6 h / 20 °C / Inert atmosphere 4: hydrogenchloride / 1,4-dioxane / 6 h / 20 °C / Inert atmosphere 5: 1-chloro-1-(dimethylamino)-2-methyl-1-propene / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 6: N-ethyl-N,N-diisopropylamine / dichloromethane / 18.5 h / 0 - 20 °C / Inert atmosphere View Scheme |
-
-
26048-05-5
beauvericin

-
-
1398380-27-2
C45H57N3O9*Na(1+)

| Conditions | Yield |
|---|---|
| With sodium picrate In water; nitrobenzene at 25℃; for 2h; |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View