Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:765-03-7
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product name : DECYL ACETYLENE Purity : 98% MF : C12H22 Application : Pharm/ Chem Industries Appearance : Liquid CAS : 765-03-7 EINECS No : 212-134-9 Type : Agrochemical Intermediates Appearance: Colorless Liquid Storage:RT.
Shanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Shanghai Massive Chemical Technology Co., Ltd.
Shanghai Massive Chemical Technology Co., Ltd. is engaged in development, production and marketing Specialty Chemicals to satisfy the changing needs of the chemical industry. We specialize in manufacturing high quality of Advanced Intermediates, Sp
GIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hangzhou Sartort Biopharma Co., Ltd
Best quality with low priceAppearance:clear colorless to light yellow liquid Storage:ln stock Package:25kg/Barrel Application:Chemicals Transportation:Express/Sea/Air Port:Shanghai
Hunan Russell Chemicals Technology Co.,Ltd
low price and high purityAppearance:solid or liquid Storage:in sealed air resistant place Package:As customer require Application:Pharma;Industry;Agricultural Transportation:by sea or by airplane Port:any port in China
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
HENAN SUNLAKE ENTERPRISE CORPORATION
Tech Data Relevant 1.Certificate Of Analysis (COA) 2..Material Safety Data Sheet (MSDS) 3.Technical Data Sheet(TDS) 4.Route of synthesis (ROS) 5.Nuclear Magnetic Resonance(NMR) 6.Method of Analysis(MOA) Our Service 1. Before Shipmen
Zhengzhou Kingorgchem Chemical Technology Co., Ltd.
Zhengzhou Kingorgchem Chemical Technology Co., Ltd. was founded on the basis of Organophosphorus Chemistry Lab of Institute of Chemistry Henan Academy of Sciences in 2015. The laboratory covers 600 m2 and the pilot plant covers 2000 m2. Kingorgchem i
Cas:765-03-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHebei Ruishun Trade Co.,Ltd
Superiority 1.We have more than 10 years experience in this area. 2.We could offer you competitive prices with satisfied quality. 3.Our company provides after-sale service and technical assistance as the customers requirements. 4.As first c
Cerametek Materials(ShenZhen)Co., Ltd.
sub/micron to mesh powders (near/spheres, whiskers, flake, fiber, porous) and grain/ball, thin/ thick metal / alloys pieces / parts, thin and thick (particle) film materials /target, and custom-made crystals, substrates/parts. In the industries and a
U-Chemo Holding Co.,Limited
U-CHEMO is a high-tech enterprise taking pharmaceutical intermediates and fine chemicals as main products. We pay high attention on product quality and apply quality management in the whole manufacturing process from research to production. In additi
Hangzhou Share Chemical Co., Ltd
At Share Chemical Company, we scrupulously abide by our policy of “Excellent Quality at a Reasonable Price”. We strive to satisfy all of our customers by providing the finest quality products supported by the finest in customer servi
Finetech Industry Limited
FINETECH INDUSTRY LIMITED is a LONDON based CRO company providing drug discovery & development services to worldwide clients. FINETECH INDUSTRY LIMITED supplies the 1-DODECYNE, CAS:765-03-7 with the most competitive price and the best quality. We can
Wuxi Morality Chemical Co., Ltd
Do best quality products, erect the morality model Application:please email us, thanks
Skyrun Industrial Co.,Ltd
Adequate stocks available,prompt shipment,Strictly control on quality,Timely after-sales
Debye Scientific
Debyesci is here who supplied several kinds of chemical products to global pharmaceutical, drug discovery, agrochemical and biotechnology industries for four yearsOur key scientific leadership team has gained experience in top research and developmen
Changzhou Foreign Trade Corp
CFTC?PharmaChem?is?a?service?based?company?in?China,?a?pharmachem?division?of?Changzhou?Foreign?Trade?Corp.,supplying?raw?materials,?intermediates,?APIs?and?fine?chemicals?for?the?pharmaceutical?and?specialty?chemical?industries?worldwide. Applicatio
Sigma-Aldrich Chemie GmbH
Package:5, 25 g in glass bottle Application:1-Dodecyne was used in the synthesis of stable ruthenium nanoparticles.
NovaChemistry
high purity Application:Drug intermediates Materials intermediates and active molecules
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium salt of 1,3-diaminopropane In tetrahydrofuran at 0℃; for 0.5h; other reagent; | 97% |
| With sodium amide; mineral oil at 210℃; |
-
-
86014-21-3
(Z)-1-dimethylphenylsilyl-2-iodo-1-dodecene

-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran; dimethyl sulfoxide at 80℃; for 0.25h; | 95% |
-
-
121134-52-9
1-trimethylsilyldodec-1-yne

-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With silver nitrate In water; acetone at 20 - 55℃; for 48h; | 92% |
-
-
19780-12-2
5-dodecyne

-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With lithium 2-aminoethylamide In ethylenediamine for 0.333333h; amide-hydrocarbon ratio 3.5:1; | 85% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol at 4℃; for 36h; Horner-Wadsworth-Emmons reaction; | 70% |

| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran; benzene at 50 - 65℃; for 4h; | 52.3% |
-
-
1052762-47-6
lithium 1-phenylethenolate

-
-
88841-79-6
1-dodecenyl phenyl selenone

-
A
-
765-03-7
1-dodecyne

-
B
-
73985-89-4
2-decylcyclopropyl phenyl ketone

| Conditions | Yield |
|---|---|
| A 19% B 41% |

-
-
55334-42-4
1,2-Dibromododecane

-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With potassium hydroxide; sodium hydride 1.) water, EtOH, reflux, 2 h, 2.) DMSO, 10 deg C, 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With ammonia; sodium amide under 5884.06 Torr; | |
| With ammonia; N,N-dimethyl-formamide nach Entfernung des Ammoniaks bei 65-70grad; |

| Conditions | Yield |
|---|---|
| With ammonia unter Druck; |

| Conditions | Yield |
|---|---|
| With sodium at 180 - 220℃; |

| Conditions | Yield |
|---|---|
| (i) , (ii) NaNH2, liq. NH3; Multistep reaction; |
-
-
67-56-1
methanol

-
-
1501-82-2
cyclododecene

-
A
-
112-41-4
1-dodecene

-
B
-
765-03-7
1-dodecyne

-
C
-
2986-54-1
Cyclododecyl methyl ether

-
D
-
51302-77-3, 134287-08-4
bicyclo<7.3.0>dodecane

| Conditions | Yield |
|---|---|
| for 60h; Irradiation; | A 2 % Chromat. B 4 % Chromat. C 11 % Chromat. D 15 % Chromat. |

-
-
1501-82-2
cyclododecene

-
A
-
112-41-4
1-dodecene

-
B
-
765-03-7
1-dodecyne

-
C
-
294-62-2
cyclododecane

-
D
-
2986-54-1
Cyclododecyl methyl ether

-
E
-
51302-77-3, 134287-08-4
bicyclo<7.3.0>dodecane

-
F
-
286-99-7
1,2-epoxycyclododecane

| Conditions | Yield |
|---|---|
| Product distribution; Mechanism; Irradiation; yields in pentane or methanol or methanol-d1; |

-
-
1501-82-2
cyclododecene

-
A
-
112-41-4
1-dodecene

-
B
-
765-03-7
1-dodecyne

-
C
-
2986-54-1
Cyclododecyl methyl ether

-
D
-
51302-77-3, 134287-08-4
bicyclo<7.3.0>dodecane

| Conditions | Yield |
|---|---|
| In methanol for 60h; Irradiation; | A 2 % Chromat. B 4 % Chromat. C 11 % Chromat. D 15 % Chromat. |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide at -78 - 90℃; for 2h; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; Schwartz's reagent; 2,2,6,6-tetramethylpiperidinyl-lithium Yield given; Multistep reaction; |

| Conditions | Yield |
|---|---|
| at 210℃; |


| Conditions | Yield |
|---|---|
| at 220℃; |

| Conditions | Yield |
|---|---|
| With sodium hydride In dimethyl sulfoxide at 20 - 68℃; | |
| With sodium hydride In dimethyl sulfoxide; mineral oil at 20 - 68℃; Reagent/catalyst; Solvent; |

| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecyne With indium(III) chloride; triethyl borane; diisobutylaluminium hydride In tetrahydrofuran at -78℃; for 2.5h; Stage #2: With hydrogen cation In tetrahydrofuran Further stages.; | 100% |
| Stage #1: 1-dodecyne With triethyl borane; dichloroindium hydride In tetrahydrofuran; hexane at -78℃; for 2.5h; Stage #2: With hydrogenchloride In tetrahydrofuran; hexane | 98% |
| With pyridine; Dimethylphenylsilane; water In acetonitrile at 80℃; for 8h; | 98% |
-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In triethylamine; N,N-dimethyl-formamide at 20℃; for 36h; Heck reaction; | 100% |
-
-
1032413-36-7
2-bromo-6-(3-hydroxy-3-methyl-1-butynyl)-4-octyloxypyridine

-
-
765-03-7
1-dodecyne

-
-
1032413-72-1
2-(1-dodecynyl)-6-(3-hydroxy-3-methyl-1-butynyl)-4-octoxypyridine

| Conditions | Yield |
|---|---|
| With copper(l) iodide; diethylamine; bis-triphenylphosphine-palladium(II) chloride at 20℃; for 2.5h; Sonogashira reaction; | 100% |

-
-
765-03-7
1-dodecyne

-
-
92740-99-3
(E)-1-triphenylgermyl-1-dodecene

| Conditions | Yield |
|---|---|
| With butan-1-ol In tetrahydrofuran 1-dodecyne (0.5 mmol) treated with germylmanganate (0.6 mmol) in THF inthe presence of n-BuOH (2.0 mmol) at 25°C for 4.5 h; | 100% |

| Conditions | Yield |
|---|---|
| With copper(II) sulphate hydrate; sodium L-ascorbate In tetrahydrofuran; water at 20℃; for 48h; Huisgen cycloaddition; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecyne With n-butyllithium In tetrahydrofuran at 5℃; for 0.5h; Inert atmosphere; Stage #2: paracetaldehyde In tetrahydrofuran at 5 - 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; Sonogashira Cross-Coupling; Schlenk technique; Inert atmosphere; Cooling with acetone-dry ice; | 100% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; diisopropylamine; triphenylphosphine; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran Ambient temperature; | 99% |
| With triethylamine In water for 2h; Sonogashira Cross-Coupling; Heating; | 78% |
-
-
765-03-7
1-dodecyne

-
-
24574-07-0
tetracosa-11,13-diyne

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; copper(l) iodide; ethyl bromoacetate; bis-triphenylphosphine-palladium(II) chloride In tetrahydrofuran at 20℃; | 99% |
| With 1,10-Phenanthroline; dicobalt octacarbonyl; carbon monoxide In acetonitrile at 80℃; under 760 Torr; for 18h; | 91% |
| With N,N,N,N,-tetramethylethylenediamine; 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate; copper(l) chloride at 20℃; for 7h; Glaser oxidative coupling; | 90% |
-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In triethylamine at 20℃; for 24h; Heck reaction; | 99% |

| Conditions | Yield |
|---|---|
| With C30H37ClN4ORu; hydrogen; sodium t-butanolate In toluene at 105℃; under 4500.45 Torr; for 20h; Glovebox; Sealed tube; | 99% |
| With diethyl ether for 24h; Milling; | 86% |
| With palladium 10% on activated carbon; hydrogen at 20℃; for 24h; Neat (no solvent); | 83% |
-
-
765-03-7
1-dodecyne

-
-
86014-19-9
1-deuterio-1-dodecyne

| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecyne With potassium carbonate In acetonitrile at 20℃; for 0.5h; Inert atmosphere; Stage #2: With water-d2 In acetonitrile at 20℃; for 1h; Inert atmosphere; | 99% |
| With n-butyllithium; water-d2 In diethyl ether; hexane | 69% |
| With water-d2 at 50℃; for 8h; | 65% |
| With water-d2 at 70℃; | |
| With water-d2; potassium carbonate In acetonitrile at 50℃; for 24.5h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| triethyl borane In hexane; benzene addn. of Et3B in hexane to a soln. of acetylene-compd. and a slight excess of Ph3GeH in benzene, heating at 60°C for 2 h; evapn., purifn. by column chromy. or preparative thin-layer chromy. on silica gel,; elem. anal.; isomer ratio Z/E=<1/20 (detd. by GC, NMR); | 99% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 100℃; for 36h; Sonogashira coupling; Inert atmosphere; | 99% |
-
-
765-03-7
1-dodecyne

-
-
7745-93-9
2-bromo-4-nitrotoluene

-
-
1052216-02-0
2-(dodec-1-yn-1-yl)-1-methyl-4-nitrobenzene

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); triethylamine; XPhos In water at 20℃; Sonogashira coupling; Inert atmosphere; Micellar solution; | 99% |
| With dichloro bis(acetonitrile) palladium(II); SPGS-550-M; NOK; triethylamine; XPhos In water for 24h; Reagent/catalyst; Sonogashira Cross-Coupling; Inert atmosphere; Sealed tube; | 98% |
-
-
765-03-7
1-dodecyne

-
-
117604-05-4
4β-azido-4-deoxy-4′-demethylepipodophyllotoxin

-
-
1310548-44-7
4'-O-demethyl-4β-[(4-decyl)-1,2,3-triazol-1-yl]-4-desoxypodophyllotoxin

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; L-ascorbic acid sodium salt In water; tert-butyl alcohol at 20℃; for 8h; | 99% |
-
-
765-03-7
1-dodecyne

-
-
1372804-17-5
1-(2,6-dibromo-phenyl)-1H-pyrrole

-
-
1423468-70-5
1-(2,6-di(dodec-1-yn-1-yl)phenyl)-1H-pyrrole

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); copper(l) iodide; tri-tert-butyl phosphine; diisopropylamine In 1,4-dioxane; toluene at 20℃; Inert atmosphere; | 99% |
-
-
765-03-7
1-dodecyne

-
-
945-51-7
1,1'-sulfinylbisbenzene

-
A
-
1193-82-4
racemic methyl phenyl sulfoxide

-
B
-
108604-35-9
1-(dodec-1-yn-1-yl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecyne; 1,1'-sulfinylbisbenzene With dichloro-[1,3-bis(2,6-diisopropylpenyl)-2-imidazolidinyliden]-(3-chloropyridyl)palladium(II); lithium tert-butoxide In tetrahydrofuran at 70℃; for 6h; Sonogashira Cross-Coupling; Schlenk technique; Inert atmosphere; Stage #2: With methyl iodide In tetrahydrofuran at 20℃; for 2h; Reagent/catalyst; Schlenk technique; Inert atmosphere; | A 98% B 99% |

-
-
765-03-7
1-dodecyne

-
-
60705-20-6
1-iodo-dodeca-1-yne

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; silver nitrate In acetone at 20℃; for 1h; Darkness; | 98% |
| With N-iodo-succinimide; silver nitrate In acetone at 20℃; for 1h; Darkness; | 98% |
| With N-iodo-succinimide; acetic acid In acetonitrile at 80℃; for 1.5h; Molecular sieve; | 93% |
-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In triethylamine at 20℃; for 24h; Heck reaction; | 98% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; diethylamine at 20℃; for 3.5h; Substitution; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecyne With n-butyllithium In tetrahydrofuran at -78℃; Stage #2: methyl chloroformate In tetrahydrofuran at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With di-tert-butyl(2,2-diphenyl-1-methyl-1-cyclopropyl)phosphine; palladium diacetate; triethylamine In water at 20℃; Sonogashira coupling; Inert atmosphere; Micellar solution; | 98% |
-
-
765-03-7
1-dodecyne

-
-
607715-89-9
(2R,3R,4S,5R)-3,4,5-Tris-benzyloxy-2-benzyloxymethyl-6-[1-iodo-meth-(Z)-ylidene]-tetrahydro-pyran

-
-
1234178-28-9
C47H56O5

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine at 20℃; for 1h; Sonogashira coupling; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With chloro(1,5-cyclooctadiene)rhodium(I) dimer; 4-trifluoromethylbenzoic acid; bis[2-(diphenylphosphino)phenyl] ether In ethanol; 1,2-dichloro-ethane at 80℃; for 16h; Inert atmosphere; Sealed tube; regioselective reaction; | 98% |
-
-
765-03-7
1-dodecyne


| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecyne With benzo[1,3,2]dioxaborole In tetrahydrofuran at 70℃; for 2h; Stage #2: tert-butyl (2R,4S)-4-((4-bromobenzyl)oxy)-2-(((tert-butyldimethylsilyl)oxy)methyl)pyrrolidine-1-carboxylate With tetrakis(triphenylphosphine) palladium(0); sodium hydrogencarbonate In tetrahydrofuran; 1,2-dimethoxyethane; water at 85℃; for 5h; Suzuki-Miyaura Coupling; | 98% |
-
-
765-03-7
1-dodecyne


| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecyne With benzo[1,3,2]dioxaborole In tetrahydrofuran at 70℃; for 2h; Stage #2: tert-butyl (2S,4R)-4-((4-bromobenzyl)oxy)-2-(((tert-butyldimethylsilyl)oxy)methyl)pyrrolidine-1-carboxylate With tetrakis(triphenylphosphine) palladium(0); sodium hydrogencarbonate In tetrahydrofuran; 1,2-dimethoxyethane; water at 85℃; for 5h; Suzuki-Miyaura Coupling; | 98% |
-
-
765-03-7
1-dodecyne

-
-
1617539-93-1
(Z)-C-6-azido-2,3-O,O-dibenzyl-4,5-didehydro-5,6-dideoxy-L-ascorbic acid

| Conditions | Yield |
|---|---|
| With copper diacetate In methanol at 50℃; Temperature; Flow reactor; Sonication; | 98% |
| With copper diacetate In methanol |
-
-
765-03-7
1-dodecyne

-
-
156642-47-6
1-iodo-2-((2-methylallyl)oxy)benzene

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate In N,N-dimethyl-formamide at 100℃; for 0.25h; Microwave irradiation; Inert atmosphere; Sealed tube; Green chemistry; | 98% |
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 24h; Schlenk technique; | 98% |
-
-
765-03-7
1-dodecyne

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate In N,N-dimethyl-formamide at 100℃; for 0.25h; Microwave irradiation; Inert atmosphere; Sealed tube; Green chemistry; | 98% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

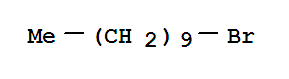














 Xi
Xi