Dayang Chem (Hangzhou) Co.,Ltd.
img _fcksaAppearance:off-white crystal Storage:Store in dry, dark and ventilated place. Package:according to the clients requirement Application:It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemic
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:83-34-1
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; ...... Appearance:Almost white crystal Package:1kg/foil bag; 1kg/tin ; 25kg/dru
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $700.0
Type:Trading Company
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Jinan Finer Chemical Co., Ltd
Product Description Product name 3-Methylindole CAS 83-34-1 Assay 99% Appearance White crystal
Henan Tianfu Chemical Co., Ltd.
3-Methylindole Basic information Product Name: 3-Methylindole Synonyms: FEMA 3019;BETA-METHYLINDOLE;3-MI;3-METHYL-1H-INDOLE;3-METHYLINDOLE;SCATOLE;SKATOL;SKATOLE CAS:
Hebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. High quality and competitive price: 1) Standard: BP/USP/EP/ enterprise standard 2) All purity ≥99% 3) We are manufacturers and can provide high quality products at factory prices. 2. Fast and safe delivery 1) The package c
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:83-34-1
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Chemical properties Uses The standard of allowable maximum amount and residue Production methods Product Name: 3-Methylindole Synonyms: FEMA 3019;BETA-METHYLINDOLE;3-MI;3-METHYL-1H-INDOL
Cas:83-34-1
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China and Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED intermediat
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $3.0 / 4.0
Type:Lab/Research institutions
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:Powder Storage:Store in sealed containers at cool & dry pla
Shanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:83-34-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
Product Name: 3-Methylindole CAS: 83-34-1 MF: C9H9N MW: 131.17 EINECS: 201-471-7 Mol File: 83-34-1.mol 3-Methylindole Structure 3-Methylindole Chemical Properties Melting point 92-97 °C (lit.) Boiling point 265-266 &de
Baoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $60.0 / 80.0
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Hubei CuiRan Biotechnology Co., Ltd
Hubei CuiRan Biotechnology Co., Ltd is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medicine)
Leader Biochemical Group
Overview of Our Factories Our Factories production lines Our Factories R&D ability Our Factories warehouse Our Factorie
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $80.0 / 100.0
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Wuhan Han Sheng New Material Technology Co.,Ltd
Our advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We have s
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hefei Zhaobo Technology Co., Ltd.
3-Methylindole CAS 83-34-1 Description Purity: 99% Min Application: Intermediates Appearance: Powder Package: Bag Delivery: 3-5days Our Advantage & Service 1.Top quality: Using high quality material and establishing a strict qualit
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $10.0 / 50.0
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Hangzhou Lingrui Chemical Co.,Ltd.
advantage: 1. The best price, satisfactory quality; 2. customers have the right to choose the delivery of parcels (EMS, DHL, FedEx, UPS); 3. customers have the right to choose from the recent effective packaging methods of their products packaging
Cas:83-34-1
Min.Order:1 Kilogram
Negotiable
Type:Other
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality Appearance:slightly brown platelets Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:A naturally abundant pneumotoxin, found prima
Siwei Development Group Ltd.
Product name: 3-Methylindole CAS No.:83-34-1 Molecule Formula:C9H9N Molecule Weight:131.17 Purity: 98.0% Package: 25kg/drum Description:White crystalline powder Manufacture Standards:Enterprise Standard TESTING ITE
Cas:83-34-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Henan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
Cas:83-34-1
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:83-34-1
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquirySynthetic route
-
-
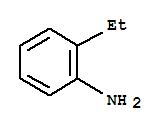
52562-19-3
2-isopropenylaniline

-
-
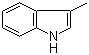
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With 1-fluoro-3,3-dimethyl-1,3-dihydro-1λ3-benzo[d][1,2]iodaoxole In acetonitrile at 20℃; Molecular sieve; | 100% |
| With dipotassium peroxodisulfate; iron(II) fluoride; triethylamine In tetrahydrofuran at 70℃; for 16h; | 54% |
| With copper diacetate In dimethyl sulfoxide at 120℃; for 12h; Wacker Oxidation; | 41% |
| Multi-step reaction with 2 steps 1: 2-Methyl-2-nitropropane; trimethylsilylazide / acetonitrile / 1 h / 0 - 20 °C 2: C58H65N5Ni2O(1+)*F6P(1-) / (2)H8-toluene / 2 h / 110 °C / Inert atmosphere; Glovebox View Scheme |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; water; hydrazine hydrate In ethanol at 100℃; for 24h; Reagent/catalyst; | 99% |
| copper chromite In diethylene glycol dimethyl ether | 97% |
| In diethylene glycol dimethyl ether | 97.5% |
-
-
36797-43-0
3-methyl-N-(p-toluenesulfonyl)indole

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol for 24h; Heating; | 99% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; | 83% |
| With sodium hydride In N,N-dimethyl acetamide at 60℃; for 2h; Inert atmosphere; | 83% |

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran; hexane at 40 - 45℃; for 2h; | 99% |
| With n-butyllithium; diisopropylamine In tetrahydrofuran; hexanes at -78 - 45℃; for 2h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In 1,4-dioxane at 100℃; for 12h; Schlenk technique; | 99% |

| Conditions | Yield |
|---|---|
| With C21H21ClIrNO2 In tetrahydrofuran; 2,2,2-trifluoroethanol for 20h; Inert atmosphere; | 98% |
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate; chloropyridinecobaloxime(III) In ethanol at 30℃; for 24h; Inert atmosphere; Schlenk technique; Irradiation; | 98% |
| With aluminum oxide In N,N-dimethyl-formamide at 120℃; for 7h; Inert atmosphere; | 92% |
-
-
73396-87-9
2-iodo-N-allylaniline

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride; sodium carbonate; palladium diacetate In N,N-dimethyl-formamide at 25℃; for 24h; | 97% |
| With palladium diacetate; tris-(2-(8-sodium sulfonatodibenzofuranyl))phosphine; triethylamine In water; acetonitrile at 40℃; for 19h; | 97% |
| palladium diacetate In acetonitrile at 110℃; | 87% |
-
-
23543-66-0
N-acetyl-3-methylindole

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol for 0.5h; Product distribution; Further Variations:; Solvents; reaction time; Heating; | 97% |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 200℃; Concentration; Temperature; | 96% |
| With zinc(II) chloride at 180℃; |

| Conditions | Yield |
|---|---|
| With 4-methylbenzenesulfonic acid-based ionic liquid supported on silica gel In ethanol at 20℃; for 4h; Fischer Indole Synthesis; | 96% |
| With ammonium cerium (IV) nitrate In methanol for 4h; Reflux; |
-
-
10590-73-5
3-methylindole-2-carboxylic acid

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With formic acid In N,N-dimethyl-formamide at 95 - 100℃; for 6h; Reagent/catalyst; Solvent; Temperature; Green chemistry; | 96% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 100℃; for 14h; Sealed tube; Autoclave; Inert atmosphere; | 95% |
| With C37H36Cl2NPRuS2; potassium hydroxide at 135℃; for 18h; Inert atmosphere; Sealed tube; | 95% |
| With cobalt(II) tetrafluoroborate hexahydrate; tris(2-diphenylphosphinoethyl)phosphine; potassium carbonate at 100℃; for 24h; Autoclave; Glovebox; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| CuO; SiO2 | 94% |

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| at -70℃; for 3h; Irradiation; | 92% |
-
-
51206-73-6
2-chloro-3-methyl-1H-indole

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With hydrogen; triethylamine; palladium on activated charcoal In ethanol | 90% |
-
-
86915-22-2
7-bromo-3-methyl-1H-indole

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In toluene at 110℃; for 12h; | 86% |
| With palladium diacetate; caesium carbonate; isobutyraldehyde In 1,4-dioxane at 80℃; |
-
-
60249-97-0
1-(1-methylethenyl)-2-nitrobenzene

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With 3,4,7,8-Tetramethyl-o-phenanthrolin; palladium diacetate; molybdenum hexacarbonyl In N,N-dimethyl-formamide at 100℃; for 10h; Inert atmosphere; Sealed tube; | 85% |
| With 4,7-dimethoxy-1,10-phenanthroline; phenylsilane; iron(II) acetate In 1,2-dimethoxyethane at 80℃; | 71% |
| With triethyl phosphite at 180℃; for 0.666667h; | 70% |
-
-
89378-43-8
3-methyl-indole-1-carboxylic acid tert-butyl ester

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol; water for 10h; Heating; | 85% |

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; sodium t-butanolate; tris-(dibenzylideneacetone)dipalladium(0) In toluene at 140℃; for 2h; | 85% |
-
-
108665-95-8
3-methyl-2-(phenylsulfonyl)-1H-indole

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With disodium hydrogenphosphate; sodium amalgam In ethanol at 0℃; Elimination; | 84% |
| With disodium hydrogenphosphate; sodium amalgam In ethanol at 0℃; | 84% |

| Conditions | Yield |
|---|---|
| With carbonylhydridetris(triphenylphosphine)rhodium(I); hydrogen; 1,2-bis-(diphenylphosphino)ethane In toluene at 100℃; under 18751.9 Torr; for 8h; regioselective reaction; | 83% |

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triisopropyl phosphite In 1-methyl-pyrrolidin-2-one at 180℃; for 0.333333h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; Microwave irradiation; | 83% |
-
-
1403266-87-4
3-methyl-1-tosylindoline

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| Stage #1: 3-methyl-1-tosylindoline In tetrahydrofuran at -78℃; for 2h; Stage #2: With oxygen In tetrahydrofuran at 20℃; | 82% |

| Conditions | Yield |
|---|---|
| With di(rhodium)tetracarbonyl dichloride; caesium carbonate In dimethyl sulfoxide at 140℃; for 24h; Inert atmosphere; | 82% |
-
-
65826-93-9
2-(2-aminophenyl)-1-propanol

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; potassium tert-butylate; nickel dibromide In toluene at 130℃; for 48h; chemoselective reaction; | 80% |
| tris(triphenylphosphine)ruthenium(II) chloride In toluene for 6h; Heating; | 79% |
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; potassium carbonate In toluene at 111℃; for 20h; | 73% |
-
-
65783-00-8
2-(2-nitrophenyl)propanenitrile

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With hydrogen at 20℃; under 760.051 Torr; for 24h; Schlenk technique; | 80% |
-
-
64987-77-5
2-(2-nitrophenyl)propanol

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With hydrogen; tris(triphenylphosphine)ruthenium(II) chloride; rhodium In toluene 1.) RT, 11 h, 2.) reflux, 6 h; | 77% |
| Multi-step reaction with 2 steps 1: H2 / Rh/C / 760 Torr 2: 79 percent / RuCl2(PPh3)3 / toluene / 6 h / Heating View Scheme |
-
-
73396-91-5
2-bromo-N-(prop-2-enyl)aniline

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With caesium carbonate; tris(dibenzylideneacetone)dipalladium (0); 1,3-bis(2,6-diisopropylphenyl)imidazolinium In dimethyl amine at 140℃; for 1h; intramolecular Heck reaction; | 77% |
| With phosphine derivative; palladium diacetate In acetonitrile at 110℃; | 60% |
-
-
337966-00-4
2-(ortho-bromophenyl)-2-methyl-ethylamine

-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| Stage #1: 2-(ortho-bromophenyl)-2-methyl-ethylamine With 1,1'-bis-(diphenylphosphino)ferrocene; palladium 10% on activated carbon; sodium t-butanolate In 1,3,5-trimethyl-benzene at 140℃; for 24h; Inert atmosphere; Stage #2: With acetic acid In 1,3,5-trimethyl-benzene at 140℃; for 24h; Inert atmosphere; | 77% |
-
-
83-34-1
3-Methylindole

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
89378-43-8
3-methyl-indole-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With dmap In acetonitrile at 20℃; for 22h; | 100% |
| With dmap In acetonitrile Ambient temperature; | 99% |
| With dmap In acetonitrile at 20℃; for 16h; | 98.5% |
-
-
83-34-1
3-Methylindole

-
-
86976-20-7
5-acetoxypyrrolidine-2-one

-
-
86976-21-8
5-(3-Methyl-indol-1-yl)-pyrrolidin-2-one

| Conditions | Yield |
|---|---|
| at 120℃; for 1.5h; | 100% |
-
-
83-34-1
3-Methylindole

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
36797-43-0
3-methyl-N-(p-toluenesulfonyl)indole

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride; sodium hydroxide In dichloromethane at 20℃; for 2h; | 100% |
| With N-benzyl-N,N,N-triethylammonium chloride; sodium hydroxide In dichloromethane at 40℃; Irradiation; | 99% |
| Stage #1: 3-Methylindole With sodium hydride In tetrahydrofuran; mineral oil for 0.5h; Stage #2: p-toluenesulfonyl chloride In tetrahydrofuran; mineral oil | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Methylindole With sodium hydride In tetrahydrofuran; oil at 0℃; for 1.08333h; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran; oil at 0 - 20℃; Inert atmosphere; | 100% |
| Stage #1: 3-Methylindole With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; Stage #2: methyl iodide In N,N-dimethyl-formamide at 20℃; for 6h; | 99% |
| Stage #1: 3-Methylindole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 0.5h; Stage #2: methyl iodide In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 6h; | 99% |

| Conditions | Yield |
|---|---|
| With bismuth(III) nitrate In methanol at 20℃; for 18h; | 100% |
| With sodium tetrachloroaurate dihydrate In ethanol at 60℃; for 6h; | 75% |
| With vanadyl triflate In dichloromethane at 20℃; for 20h; Friedel-Crafts reaction; | 73% |
-
-
83-34-1
3-Methylindole

-
-
135-02-4
ortho-anisaldehyde

-
-
626253-46-1
2-methoxyphenyl(2,2'-bis-3-methylindolyl)methane

| Conditions | Yield |
|---|---|
| In ethanol for 0.05h; Microwave irradiation; Ionic liquid; | 100% |
| With sulfuric acid In ethanol at 20℃; for 24h; | 80% |
-
-
83-34-1
3-Methylindole

-
-
120-14-9
3,4-dimethoxy-benzaldehyde

-
-
1393904-91-0
3,4-dimethoxyphenyl(2,2'-bis-3-methylindolyl)methane

| Conditions | Yield |
|---|---|
| In ethanol for 0.05h; Microwave irradiation; Ionic liquid; | 100% |
-
-
83-34-1
3-Methylindole

-
-
1538-75-6
2,2-dimethylpropanoic anhydride

-
-
89378-43-8
3-methyl-indole-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane for 2h; | 100% |
-
-
83-34-1
3-Methylindole

-
-
1419209-51-0
1-(4-(dimethylamino)phenyl)-3-(3-methyl-1H-indol-2-yl)-1H-imidazolium bromide

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In 1,4-dioxane at 12 - 15℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In 1,4-dioxane for 0.583333h; Cooling with ice; | 100% |
| With N-Bromosuccinimide In 1,4-dioxane at 12 - 15℃; for 0.583333h; | 94% |
-
-
83-34-1
3-Methylindole

-
-
905556-47-0
N-(4-methoxyphenyl)-1-(2-(phenylethynyl)phenyl)methanimine

| Conditions | Yield |
|---|---|
| With silver trifluoromethanesulfonate; acetic acid In acetonitrile at 20℃; Inert atmosphere; | 100% |
-
-
83-34-1
3-Methylindole

-
-
98-88-4
benzoyl chloride

-
-
57826-37-6
(3-methyl-1H-indol-1-yl)(phenyl)methanone

| Conditions | Yield |
|---|---|
| With tetra(n-butyl)ammonium hydrogensulfate; sodium hydroxide In dichloromethane at 20℃; for 1h; Inert atmosphere; | 99% |
| Stage #1: 3-Methylindole With n-butyllithium In tetrahydrofuran at 0℃; for 0.5h; Stage #2: benzoyl chloride In tetrahydrofuran at 0 - 20℃; for 2h; | 95% |
| Stage #1: 3-Methylindole With lithium hexamethyldisilazane In tetrahydrofuran; dichloromethane at 20℃; Stage #2: benzoyl chloride In tetrahydrofuran; dichloromethane at 20℃; for 24h; Further stages.; | 94% |
-
-
83-34-1
3-Methylindole

-
-
5888-29-9
diethyl(methoxymethyl)amine

-
-
1228023-70-8
N-ethyl-N-((3-methyl-1H-indol-1-yl)methyl)ethanamine

| Conditions | Yield |
|---|---|
| With hafnium tetrakis(trifluoromethanesulfonate) In dichloromethane at 20℃; for 0.2h; Inert atmosphere; regioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Methylindole; C10H12Cl2N2O4PS(1+)*Cl(1-) In dichloromethane at 20℃; for 0.166667h; Stage #2: With trichlorophosphate In dichloromethane for 4h; | 99% |
-
-
83-34-1
3-Methylindole

-
-
37517-81-0
3-chloro-3-oxopropanoic acid methyl ester

| Conditions | Yield |
|---|---|
| In 1,2-dichloro-ethane Reflux; | 99% |
| Stage #1: 3-Methylindole With sodium hydride In tetrahydrofuran at 0℃; for 0.5h; Inert atmosphere; Stage #2: 3-chloro-3-oxopropanoic acid methyl ester In tetrahydrofuran at 20℃; for 14h; Inert atmosphere; | 73% |
| Inert atmosphere; Alkaline conditions; |

| Conditions | Yield |
|---|---|
| With tris(2,2'-bipyridyl)ruthenium dichloride; N,N,N,N,-tetramethylethylenediamine In acetonitrile at 20℃; for 0.166667h; Flow reactor; Irradiation; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In acetone at 12 - 15℃; for 0.583333h; Inert atmosphere; | 99% |
| With N-Bromosuccinimide In 1,4-dioxane for 0.583333h; Cooling with ice; | 66% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In dichloromethane at 20℃; for 0.5h; chemoselective reaction; | 99% |
| With toluene-4-sulfonic acid In dichloromethane at 20℃; for 0.5h; | 99% |

-
-
83-34-1
3-Methylindole

-
-
89201-08-1
diethyl 2-(2-oxo-2-phenylethylidene)propanedioate

| Conditions | Yield |
|---|---|
| With copper(II) bis(trifluoromethanesulfonate); 2,2-bis[(4S)-4-isopropyloxazolin-2-yl]propane In dichloromethane at 20℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With C42H33N3O2; zinc trifluoromethanesulfonate In toluene at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With C42H33N3O2; zinc trifluoromethanesulfonate In toluene at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; enantioselective reaction; | 99% |
-
-
83-34-1
3-Methylindole

-
-
15241-32-4, 21298-69-1, 3316-24-3
1-methoxy-2-(2-nitrovinyl)benzene

| Conditions | Yield |
|---|---|
| With C42H33N3O2; zinc trifluoromethanesulfonate In toluene at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With C42H33N3O2; zinc trifluoromethanesulfonate In toluene at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; enantioselective reaction; | 99% |
-
-
83-34-1
3-Methylindole

-
-
58956-40-4
3-hydroxy-1-methyl-3-(1H-indol-3-yl)indolin-2-one

| Conditions | Yield |
|---|---|
| With gallium(III) bromide In dichloromethane at 25℃; for 12h; Solvent; Inert atmosphere; chemoselective reaction; | 99% |
-
-
83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With copper(ll) bromide In chloroform at 20℃; for 0.5h; | 99% |
-
-
83-34-1
3-Methylindole

-
-
1263145-84-1
9-methyl-1-(naphthalen-1-yl)-3-phenyl-9H-pyrrolo[1,2-a]indole

| Conditions | Yield |
|---|---|
| With copper(ll) bromide In chloroform at 20℃; for 1h; | 99% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View















 Xi
Xi