-
Name
2-Methylquinoxaline
- EINECS 230-664-9
- CAS No. 7251-61-8
- Article Data94
- CAS DataBase
- Density 1.141 g/cm3
- Solubility miscible with water
- Melting Point 180.5°C
- Formula C9H8N2
- Boiling Point 243.9 °C at 760 mmHg
- Molecular Weight 144.176
- Flash Point 107.2 °C
- Transport Information
- Appearance dark red to brown liquid.
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms NSC 65587;
- PSA 25.78000
- LogP 1.93820
Synthetic route

| Conditions | Yield |
|---|---|
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; copper(II) acetate monohydrate In N,N-dimethyl-formamide at 20℃; Temperature; | 88% |
| With triethylamine; palladium diacetate In tetrahydrofuran; toluene at 20℃; for 3h; | 87% |
| With zinc(II) iodide In ethanol; water at 80℃; for 0.5h; | 86% |

| Conditions | Yield |
|---|---|
| In water for 0.0333333h; Isay condensation; microwave irradiation; | 95% |
| Stage #1: 2-oxopropanal With sodium disulfite In water for 0.166667h; Addition; Stage #2: 1,2-diamino-benzene In water at 20℃; for 18h; Condensation; Cyclization; | 91% |
| In water at 20℃; for 0.5h; Green chemistry; | 85% |

| Conditions | Yield |
|---|---|
| With cesiumhydroxide monohydrate In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; | 96% |
| With cesiumhydroxide monohydrate; C17H14Br2CoN4 In toluene at 150℃; for 24h; Reagent/catalyst; Temperature; Sealed tube; Inert atmosphere; | 93% |
| With 1,10-Phenanthroline; cesiumhydroxide monohydrate; nickel dibromide In toluene at 150℃; for 24h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; Sealed tube; | 93% |

| Conditions | Yield |
|---|---|
| With cesiumhydroxide monohydrate In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; | 93% |
| With 1,10-Phenanthroline; cesiumhydroxide monohydrate; nickel dibromide In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; | 93% |
| With C18H24ClIrN3O(1+)*Cl(1-); potassium hydroxide In water for 24h; Mechanism; Schlenk technique; Reflux; Green chemistry; | 87% |

| Conditions | Yield |
|---|---|
| With cesiumhydroxide monohydrate; [(η6-p-cymene)2Ru2(2,7-bis(di-2-pyridinyl)-1,8-naphthyridine)Cl2](PF6)2; oxygen at 120℃; for 23h; Inert atmosphere; Molecular sieve; | 43% |
-
-
6640-55-7, 24463-30-7, 24463-31-8, 49849-47-0
2-methyl-1,2,3,4-tetrahydroquinoxaline

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; potassium tert-butylate; oxygen; nickel dibromide In tert-Amyl alcohol at 95℃; for 48h; | 99% |
| With C21H21ClIrNO2 In tetrahydrofuran; 2,2,2-trifluoroethanol for 20h; Inert atmosphere; | 92% |
| With potassium tert-butylate In o-xylene at 140℃; for 36h; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); caesium carbonate In N,N-dimethyl-formamide at 130℃; for 18h; Inert atmosphere; | 38 mg |
-
-
6935-29-1
quinoxaline-N-oxide

-
-
2065-66-9
methyl-triphenylphosphonium iodide

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 80℃; for 6h; Sealed tube; Inert atmosphere; | 78% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,2-Dinitrobenzene With hydrogen In diethylene glycol dimethyl ether at 80℃; under 7500.75 Torr; for 24h; Stage #2: propylene glycol In diethylene glycol dimethyl ether at 140℃; for 30h; | 83% |
-
-
291536-12-4
N1-(prop-2-yn-1-yl)benzene-1,2-diamine

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With tin(II) chloride hydrate In water; isopropyl alcohol at 82℃; for 12h; | 85% |

| Conditions | Yield |
|---|---|
| With perchloric acid In acetonitrile at 20℃; for 0.25h; | 95% |

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin(II) chloride hydrate In water; isopropyl alcohol Reflux; | 82% |
| Multi-step reaction with 2 steps 1: sodium dithionite; water / ethanol / 1 h / 0 - 20 °C 2: tin(II) chloride hydrate / isopropyl alcohol; water / 12 h / 82 °C View Scheme |
-
-
57-55-6
propylene glycol

-
-
95-54-5
1,2-diamino-benzene

-
A
-
91-19-0
2,3-diethynylquinoxaline

-
B
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With oxygen; zeolite; titanium(IV) oxide In acetonitrile at 20℃; for 10h; Irradiation; | A 12.6% B 22.5% |


| Conditions | Yield |
|---|---|
| With (R)-3,3'-bis(2,4,6-triisopropylphenyl)-1,1'-binaphthyl-2,2'-diylhydrogenphosphate In chloroform; water at 35℃; for 8h; Inert atmosphere; Molecular sieve; enantioselective reaction; | 63% |

| Conditions | Yield |
|---|---|
| With dodecacarbonyl-triangulo-triruthenium; cesiumhydroxide monohydrate; 1,3-bis-(diphenylphosphino)propane In tert-Amyl alcohol at 150℃; for 12h; Schlenk technique; Inert atmosphere; | 36% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In toluene at 150℃; for 12h; Inert atmosphere; Sealed tube; Green chemistry; | 44% |

| Conditions | Yield |
|---|---|
| In ethanol for 1.25h; | 74% |
| With acetic acid | |
| With hydrogenchloride; acetic acid |
-
-
6935-29-1
quinoxaline-N-oxide

-
-
78782-17-9
4,4,5,5-tetramethyl-2-[(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methyl]-1,3,2-dioxaborolane

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With sodium methylate In toluene at 80℃; for 3h; regioselective reaction; | 32% |

| Conditions | Yield |
|---|---|
| In diethyl ether at -70℃; for 120h; | 53% |

| Conditions | Yield |
|---|---|
| With mercury(II) diacetate In tetrahydrofuran for 14h; Ambient temperature; | 51% |
-
-
91-19-0
2,3-diethynylquinoxaline

-
-
110-82-7
cyclohexane

-
A
-
7251-61-8
2-methylquinoxaline

-
B
-
15787-49-2
2-pyridylcyclohexane

-
C
-
13669-35-7
4-cyclohexylpyridine

-
D
-
40115-00-2
2-cyclohexyl-quinoxaline

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iron(II) nitrate; acetic acid In pyridine at 60℃; for 18h; Further byproducts given; | A n/a B n/a C n/a D 15% |


| Conditions | Yield |
|---|---|
| With sodium carbonate; sodium sulfite In water 1.) 20 deg C, 2 h, 2.) 100 deg C, 1 h; | 59% |

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 70 percent / H2 / 10percent Pd/C / ethanol / 760 Torr / Ambient temperature 2: 92 percent / 2) DDQ / multistep reaction thermal rearrangement of other 1,2,3-triazoles View Scheme | |
| Multi-step reaction with 2 steps 1: 70 percent / H2 / 10percent Pd/C / ethanol / 760 Torr / Ambient temperature 2: 27 percent / 2) 10percent Pd/C / 1) toluene, reflux, 1h, 2) ethanol, reflux, 30 min View Scheme |

| Conditions | Yield |
|---|---|
| With sodium sulfite In various solvent(s) at 100℃; pH 10; | 52.3 % Chromat. |

| Conditions | Yield |
|---|---|
| With sodium sulfite In various solvent(s) at 100℃; pH 10; | 54.6 % Chromat. |
-
-
87074-49-5
5-(diethoxymethyl)-3-methylisoxazole

-
-
95-54-5
1,2-diamino-benzene

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With formic acid at 80℃; for 0.0833333h; | 21% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 72h; Ambient temperature; | 80% |

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one; iron(II) acetylacetonate In tetrahydrofuran for 0.5h; |

| Conditions | Yield |
|---|---|
| With 1-Phenylethanol; sodium t-butanolate at 100℃; for 8h; Schlenk technique; Irradiation; Green chemistry; chemoselective reaction; | 12% |

| Conditions | Yield |
|---|---|
| In water for 0.0180556h; Isay condensation; microwave irradiation; | 48% |
-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With {[IrH((S)-Difluorophos)]2(μ-Cl)3}+Cl-; hydrogen In toluene at 30℃; under 22502.3 Torr; for 20h; Inert atmosphere; Autoclave; optical yield given as %ee; enantioselective reaction; | 99% |
| With [(Ir(H){(S)-[(4,4-bi-2,2-difluoro-1,3-benzodioxole)-5,5-diyl]bis(diphenylphosphine)})2(μ-Cl)3]Cl; hydrogen In toluene at 30℃; under 22502.3 Torr; for 20h; Autoclave; optical yield given as %ee; enantioselective reaction; | 99% |
| With RuCl[(R)-1-isopropyl-2,2-bis(4-methoxyphenyl)ethylenediamine][(R)-((4,4'-bi-1,3-benzodioxole)-5,5’-diyl)bis(di(3,5-xylyl)phosphine)]; potassium tert-butylate; hydrogen In toluene at 40℃; under 15001.5 Torr; for 13h; Reagent/catalyst; Solvent; Temperature; Autoclave; enantioselective reaction; | 99.9% |

| Conditions | Yield |
|---|---|
| With malononitrile In diethylene glycol dimethyl ether at 120℃; under 2585.81 Torr; for 0.416667h; Microwave irradiation; | 84% |
| In water at 120℃; for 36h; Sealed tube; | 80% |
| With Fe3O(biphenyl-4,4'-dicarboxylate)3; acetic acid In toluene at 120℃; for 24h; Inert atmosphere; | 66% |
-
-
7251-61-8
2-methylquinoxaline

-
-
54804-43-2
2-(bromomethyl)quinoxaline

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; trityl tetrafluoroborate In dichloromethane at 20℃; Irradiation; Inert atmosphere; chemoselective reaction; | 82% |
| With tetra-(n-butyl)ammonium iodide; ethylene dibromide; urea In acetonitrile at 95℃; for 0.5h; Microwave irradiation; | 70% |
| With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In tetrachloromethane at 60℃; for 1h; Irradiation; | 57% |

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; 18-crown-6 ether; potassium tert-butylate In tetrahydrofuran at 120℃; for 20h; Inert atmosphere; Schlenk technique; Sealed tube; | 93% |
| With [(1,5-cyclooctadiene)(OH)iridium(I)]2; potassium tert-butylate; triphenylphosphine In 1,4-dioxane at 130℃; for 24h; Inert atmosphere; | 90% |
| With C17H14Br2CoN4; potassium tert-butylate In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; Green chemistry; | 89% |
-
-
7251-61-8
2-methylquinoxaline

-
-
555-16-8
4-nitrobenzaldehdye

-
-
1450605-01-2
1-(4-nitrophenyl)-2-(quinoxalin-2-yl)ethanol

| Conditions | Yield |
|---|---|
| With H2O4P(1-)*C4H7N2(1+) In 1,4-dioxane; water at 100℃; for 24h; Schlenk technique; Sealed tube; | 87% |
| With morpholin-4-ium hydrogen sulphate; 1-butyl-3-methylimidazolium Tetrafluoroborate at 110℃; for 24h; Sealed tube; | 87% |
| With copper(II) ferrite; tetrabutyl-ammonium chloride In ethylene glycol at 100℃; for 24h; Green chemistry; | 73% |
| In water at 102℃; for 0.416667h; Sealed tube; Microwave irradiation; | 60% |
-
-
7251-61-8
2-methylquinoxaline

-
-
6640-55-7, 24463-30-7, 24463-31-8, 49849-47-0
2-methyl-1,2,3,4-tetrahydroquinoxaline

| Conditions | Yield |
|---|---|
| With lithium borohydride; methyl iodide In tetrahydrofuran at 20℃; for 0.25h; | 99% |
| With ammonium borohydride In methanol at 60℃; for 21h; Schlenk technique; | 99% |
| Stage #1: 2-methylquinoxaline With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; N-(2-aminoethyl)-4-(trifluoromethyl)benzenesulfonamide; sodium formate; sodium acetate; acetic acid In water at 80℃; for 0.25h; pH=5.5; Stage #2: With potassium hydroxide In water regioselective reaction; | 96% |
-
-
7251-61-8
2-methylquinoxaline

-
-
41035-18-1
2-(methyl-d3)quinoxaline

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); [bis(acetoxy)iodo]benzene; water-d2 In N,N-dimethyl-formamide at 80℃; for 12h; Schlenk technique; Inert atmosphere; | 97% |
| With water-d2; benzoic acid at 90℃; for 8h; Inert atmosphere; Schlenk technique; | |
| With potassium tert-butylate; water-d2 In dimethyl sulfoxide at 20℃; for 0.166667h; Sealed tube; |
-
-
7251-61-8
2-methylquinoxaline

-
-
106435-53-4
2-chloromethyl-quinoxaline

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; 1,2-dichloro-ethane; urea at 110℃; for 0.5h; Microwave irradiation; | 87% |
| With trichloroisocyanuric acid In chloroform at 60℃; for 1h; | 76.2% |
| With trichloroisocyanuric acid In chloroform for 0.5h; Chlorination; Heating; | |
| Multi-step reaction with 3 steps 1: 60 percent / KOH, Ac2O 2: 1.) O3, 2.) NaBH4 / 1.) MeOH, CH2Cl2, -78 deg C, 2.) MeOH, CH2Cl2, from 0 deg C to RT, 2 h 3: 67 percent / SOCl2 / benzene / 2 h / Ambient temperature View Scheme | |
| With trichloroisocyanuric acid In chloroform for 1.5h; Inert atmosphere; Reflux; |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate In acetonitrile at 20℃; for 2h; Inert atmosphere; Sealed tube; | 94% |
| With oxygen In acetonitrile at 20℃; for 12h; Irradiation; Green chemistry; | 69% |
| With oxygen In neat (no solvent) at 65℃; for 1h; Green chemistry; | 57% |

| Conditions | Yield |
|---|---|
| With bromopentacarbonylmanganese(I); potassium tert-butylate; 1-(pyridin-2-yl)-2-(1-(pyridin-2-yl)ethylidene)hydrazine In tert-butyl alcohol at 140℃; for 14h; Inert atmosphere; Schlenk technique; | 91% |
| With potassium hydroxide In toluene at 120℃; for 18h; Sealed tube; | 87% |
| With potassium tert-butylate; nickel dibromide; N-(8-quinolyl)pyridine-2-carboxamide In tert-butyl alcohol at 140℃; for 48h; Schlenk technique; Inert atmosphere; Sealed tube; | 84% |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; trifluorormethanesulfonic acid; silver trifluoroacetate In dichloromethane; water at 45℃; for 20h; Inert atmosphere; | 76.8% |
| With 4,5,6,7-tetrafluoro-1-hydroxybenzo[d][1,2]iodaoxol-3(1H)-one; Ru(bpy)3Cl2 at 30℃; for 24h; Minisci Aromatic Substitution; Inert atmosphere; Irradiation; regioselective reaction; | 63% |
| With dipotassium peroxodisulfate; sulfuric acid; silver nitrate In water at 50℃; for 24h; Inert atmosphere; Sealed tube; regioselective reaction; | 59% |

| Conditions | Yield |
|---|---|
| With iron(II) acetate In 1,4-dioxane at 120℃; for 24h; Schlenk technique; Inert atmosphere; | 88% |
| With iron(II) acetate In 1,4-dioxane at 120℃; for 24h; Inert atmosphere; Schlenk technique; Green chemistry; | 88% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; diphenyl-phosphinic acid; tetra-(n-butyl)ammonium iodide In dimethyl sulfoxide at 90℃; for 8h; Sealed tube; | 85% |

-
-
105-13-5
4-Methoxybenzyl alcohol

| Conditions | Yield |
|---|---|
| With C17H14Br2CoN4; potassium tert-butylate In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; Green chemistry; | 88% |
| With potassium hydroxide In toluene at 140℃; for 12h; | 86% |
-
-
7251-61-8
2-methylquinoxaline

-
-
1539-59-9
4-cyclohexyl-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylquinoxaline; 4-cyclohexyl-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylic acid diethyl ester With trifluoroacetic acid In 2,2,2-trifluoroethanol at 20℃; for 4h; Sealed tube; Irradiation; Inert atmosphere; Stage #2: In 2,2,2-trifluoroethanol at 20℃; for 4h; | 66% |
| Stage #1: 2-methylquinoxaline; 4-cyclohexyl-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylic acid diethyl ester With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 4h; Irradiation; Inert atmosphere; Sealed tube; Stage #2: In 2,2,2-trifluoroethanol for 4h; Irradiation; Inert atmosphere; | 66% |
-
-
7251-61-8
2-methylquinoxaline

-
-
95-54-5
1,2-diamino-benzene

-
-
2215-43-2
2-(1H-benzo[d]imidazol-2-yl)quinoxaline

| Conditions | Yield |
|---|---|
| With iodine; oxygen In dimethyl sulfoxide at 110℃; | 98% |
-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; iodine In acetonitrile at 70℃; for 3h; regioselective reaction; | 81% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; diphenyl-phosphinic acid; tetra-(n-butyl)ammonium iodide In dimethyl sulfoxide at 90℃; for 8h; Sealed tube; | 69% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; diphenyl-phosphinic acid; tetra-(n-butyl)ammonium iodide In dimethyl sulfoxide at 90℃; for 8h; Sealed tube; | 74% |

| Conditions | Yield |
|---|---|
| With C17H14Br2CoN4; potassium tert-butylate In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; Green chemistry; | 86% |
| With potassium hydroxide In toluene at 140℃; for 12h; | 67% |

| Conditions | Yield |
|---|---|
| With tert-Butyl peroxybenzoate; tetra-(n-butyl)ammonium iodide In water at 100℃; for 10h; Sealed tube; Green chemistry; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylquinoxaline With sodium hydride In tetrahydrofuran at 0℃; for 0.5h; Knoevenagel Condensation; Inert atmosphere; Stage #2: 9-anthracene aldehyde In tetrahydrofuran at 20℃; for 4h; Knoevenagel Condensation; Inert atmosphere; | 88% |
-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With C25H30N2O2RuS(1+)*C32H12BF24(1-); hydrogen In 1,1-dichloroethane at 40℃; under 38002.6 Torr; for 8h; Autoclave; optical yield given as %ee; enantioselective reaction; | 95% |
| With C71H73ClN2O2P2Ru; potassium tert-butylate; hydrogen In isopropyl alcohol at 25℃; under 15201 Torr; for 22h; Autoclave; enantioselective reaction; | 89 %Spectr. |
-
-
946005-73-8
4-(N,N-di-(pyridine-2-ylmethyl)amino)benzaldehyde

-
-
7251-61-8
2-methylquinoxaline

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 20℃; for 15h; Aldol Condensation; Reflux; | 84% |
-
-
7251-61-8
2-methylquinoxaline

-
-
1440513-46-1
2,3-diphenyl-3,4-dihydroquinazolin-4-ol

| Conditions | Yield |
|---|---|
| With iron(III) chloride In 1,2-dichloro-ethane at 80℃; for 6h; | 84% |
2-Methylquinoxaline Chemical Properties
IUPAC Name: 2-Methylquinoxaline
CAS: 7251-61-8
EINECS: 230-664-9
Product Categories: Quinolines, Quinazolines and Derivatives ; Building Blocks ; Heterocyclic Building Blocks ; Quinoxalines ; Alphabetical Listings ; Flavors and Fragrances ; M-N.
Following is the Molecular Structure of 2-Methylquinoxaline (7251-61-8):
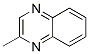
SMILES: n1c2ccccc2nc(c1)C
InChI: InChI=1/C9H8N2/c1-7-6-10-8-4-2-3-5-9(8)11-7/h2-6H,1H3
InChIKey: ALHUXMDEZNLFTA-UHFFFAOYAA
Std. InChI: InChI=1S/C9H8N2/c1-7-6-10-8-4-2-3-5-9(8)11-7/h2-6H,1H3
Std. InChIKey: ALHUXMDEZNLFTA-UHFFFAOYSA-N
Molecular Formula: C9H8N2
Molecular Weight: 144.17
Boiling Point: 243.9℃ at 760mmHg
Flash Point: 107.2℃
Molar Volume: 126.2cm3
Density: 1.141g/cm3
Molar Refractivity: 45.1cm3
Surface Tension: 50.7dyne/cm
Polarizability: 17.88 10-24cm3
Enthalpy of Vaporization: 46.15 kJ/mol
Vapour Pressure: 0.0488mmHg at 25℃
Refractive Index: n20/D 1.613(lit.)
Water Solubility: Miscible
BRN: 113307
2-Methylquinoxaline Safety Profile
Hazard Codes: ![]() Xi Irritant.
Xi Irritant.
Risk Statements: 36/37/38
R36 Irritating to eyes.
R37 Irritating to respiratory system.
R38 Irritating to skin
Safety Statements: 26-36-24/25
S24 Avoid contact with skin.
S25 Avoid contact with eyes.
S26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S27 Take off immediately all contaminated clothing.
S28 After contact with skin, wash immediately with plenty of soap-suds.
S29 Do not empty into drains.
S30 Never add water to this product.
S33 Take precautionary measures against static discharges.
S35 This material and its container must be disposed of in a safe way.
S36 Wear suitable protective clothing.
WGK Germany:3
RTECS:VD3450000
F:10
2-Methylquinoxaline Specification
2-Methylquinoxaline (7251-61-8),which also can be called for 2-Methyl-Quinoxalin ; Quinoxaline,2-Methyl- ; 2-Methylquinoxaline ; 2-Methylquinoxaline 97+% ; 2-Methylquinoxaline,96% ; Methylquinoxaline,2-Methylchinoxaline ; 2-Methylbenzopyrazine ; 3-Methylquinoxaline . 2-Methylquinoxaline (7251-61-8) is liquid,its colour is dark red to brown.It's slightly soluble in water.The followings are the first aid measures about this chemical:Ingestion:If victim is conscious and alert, give 2-4 cupfuls of milk or water. Never give anything by mouth to an unconscious person. Get medical aid immediately.Inhalation:Remove from exposure to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.Skin:Get medical aid. Flush skin with plenty of soap and water for at least 15 minutes while removing contaminated clothing and shoes. Remove contaminated clothing and shoes.Eyes:Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately. 2-Methylquinoxaline(7251-61-8) should be stored in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances. It is stable under normal temperatures and pressures.After decomposition,it turned into nitrogen oxides, carbon monoxide, carbon dioxide and nitrogen.
Related Products
- 2-Methylquinoxaline
- 7251-72-1
- 7251-90-3
- 72519-17-6
- 7251-97-0
- 7252-03-1
- 72520-94-6
- 72521-00-7
- 7252-11-1
- 72522-13-5
- 7252-30-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View