-
Name
3-Diethylaminopropylamine
- EINECS 203-236-4
- CAS No. 104-78-9
- Article Data49
- CAS DataBase
- Density 0.84 g/cm3
- Solubility miscible with water
- Melting Point -60 °C
- Formula C7H18N2
- Boiling Point 170.7 °C at 760 mmHg
- Molecular Weight 130.233
- Flash Point 58.9 °C
- Transport Information UN 2684 3/PG 3
- Appearance clear colorless to slightly yellowish liquid
- Safety 26-36/37/39-45
- Risk Codes 10-21/22-34-43
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi,  C
C
- Synonyms 1,3-Propanediamine,N,N-diethyl- (6CI,7CI,8CI,9CI);(3-Diethylaminopropan-1-yl)amine;1-Amino-3-(diethylamino)propane;3-(Diethylamino)-n-propylamine;3-(Diethylamino)propanamine;3-(Diethylamino)propylamine;3-(N,N-Diethylamino)-1-propylamine;3-(N,N-Diethylamino)propylamine;3-Diethylamino-1-propylamine;Diethylaminotrimethylenamine;N,N-Diethyl-1,3-diaminopropane;N,N-Diethyl-1,3-propanediamine;N,N-Diethyl-1,3-propylenediamine;N,N-Diethyltrimethylenediamine;N,N-Ethylpropane-1,3-diamine;N-(3-Aminopropyl)-N,N-diethylamine;N-(3-Aminopropyl)diethylamine;N-(3-Diethylaminopropyl)amine;N-(Diethylaminopropyl)amine;N-Aminopropyl-N,N-diethylamine;N1,N1-Diethylpropane-1,3-diamine;NSC 7776;g-(Diethylamino)propylamine;
- PSA 29.26000
- LogP 1.37730
Synthetic route
-
-
20824-58-2
2-(3-diethylaminopropyl)-1H-isoindole-1,3(2H)-dione

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol for 24h; Reflux; | 73% |
| With hydrazine hydrate In ethanol for 3h; Reflux; | 14% |
| With hydrazine |

| Conditions | Yield |
|---|---|
| With ethanol; sodium for 3h; Reflux; | 56% |
| With sodium-potassium alloy; butan-1-ol | |
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With xylene Hydrolyse des entstandenen N-<3-Diaethylamino-propyl>-phthalimids mit konz.HCl bei Siedetemperatur.; | |
| With xylene Hydrolyse des entstandenen N-<3-Diaethylamino-propyl>-phthalimids mit konz.HCl bei Siedetemperatur.; |
-
-
5460-29-7
2-(3-bromopropyl)isoindole-1,3-dione

-
-
109-89-7
diethylamine

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| Hydrolyse des entstandenen N-<3-Diaethylamino-propyl>-phthalimids mit konz.HCl bei Siedetemperatur.; | |
| (i) KOH, MeOH, (ii) aq. KOH, (heating); Multistep reaction; | |
| Hydrolyse des entstandenen N-<3-Diaethylamino-propyl>-phthalimids mit konz.HCl bei Siedetemperatur.; |
-
-
5351-04-2
3-diethylaminopropionitrile

-
A
-
6050-28-8
bis-(3-diethylamino-propyl)-amine

-
B
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With sodium-potassium alloy; butan-1-ol | |
| With nickel at 100℃; under 62518.2 Torr; Hydrogenation; |
-
-
100536-45-6
tetra-N-ethyl-ξ-propenediyldiamine

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With ammonia; nickel at 100℃; under 91938.4 Torr; Hydrogenation; | |
| With ammonia; nickel at 100℃; under 91938.4 Torr; Hydrogenation; |
-
-
109-89-7
diethylamine

-
-
156-87-6
propan-1-ol-3-amine

-
A
-
107-11-9
1-amino-2-propene

-
B
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With thorium dioxide at 270 - 290℃; |

-
-
503-29-7
azetidine

-
-
109-89-7
diethylamine

-
A
-
103502-67-6
1-<(3-aminopropyl)aminopropyl>diethylamine

-
B
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With perchloric acid at 25℃; Rate constant; Thermodynamic data; E(a), ΔS(excit.); |


-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; diethyl ether |
-
-
5351-04-2
3-diethylaminopropionitrile

-
A
-
6050-28-8
bis-(3-diethylamino-propyl)-amine

-
B
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| Hydrogenation; |

-
-
5351-04-2
3-diethylaminopropionitrile

-
-
71-36-3
butan-1-ol

-
A
-
6050-28-8
bis-(3-diethylamino-propyl)-amine

-
B
-
104-78-9
diethylaminopropylamine

-
-
5351-04-2
3-diethylaminopropionitrile

-
-
64-17-5
ethanol

-
A
-
6050-28-8
bis-(3-diethylamino-propyl)-amine

-
B
-
104-78-9
diethylaminopropylamine

-
-
5351-04-2
3-diethylaminopropionitrile

-
-
71-36-3
butan-1-ol

-
A
-
6050-28-8
bis-(3-diethylamino-propyl)-amine

-
B
-
104-78-9
diethylaminopropylamine

-
-
57796-55-1
6-chloro-5-ethylamino-3-methoxy-pyrazine-2-carboxylic acid 3-diethylamino-propylamide

-
A
-
104-78-9
diethylaminopropylamine

-
B
-
57796-33-5
6-chloro-5-ethylamino-3-methoxy-pyrazine-2-carboxylic acid
-
-
109-89-7
diethylamine

-
-
107-02-8
acrolein

-
A
-
104-78-9
diethylaminopropylamine

-
B
-
626-56-2, 53152-98-0
3-Methylpiperidine

| Conditions | Yield |
|---|---|
| Stage #1: diethylamine; acrolein In tetrahydrofuran at 4℃; Stage #2: With ammonia; hydrogen; Raney cobalt In tetrahydrofuran at 80℃; under 45004.5 Torr; for 6h; Product distribution / selectivity; autoclave; | A 52 %Chromat. B 8 %Chromat. |


| Conditions | Yield |
|---|---|
| Stage #1: diethylamine; acrolein In tetrahydrofuran at 4℃; Stage #2: With ammonia; hydrogen; Raney cobalt In tetrahydrofuran at 80℃; under 45004.5 Torr; for 6h; Product distribution / selectivity; autoclave; | 87 %Chromat. |
-
-
66999-80-2
N-(3-(diethylamino)-propyl)benzamide

-
A
-
104-78-9
diethylaminopropylamine

-
B
-
100-51-6
benzyl alcohol

| Conditions | Yield |
|---|---|
| With Ag/γ-Al2O3 (5 mol%); potassium tert-butylate; hydrogen In 1,4-dioxane at 170℃; under 45004.5 Torr; for 120h; Green chemistry; chemoselective reaction; | A 83 %Spectr. B 84 %Spectr. |
-
-
812699-95-9
N-{3-[4-(6-chloropyridin-3-yl)-pyrimidin-2-ylamino]-4-methylphenyl}-3-trifluoromethylbenzamide

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| at 110℃; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: C36H28Cl2N12O8S2(2-)*2Na(1+); diethylaminopropylamine In water at 70 - 97℃; for 4h; Stage #2: With hydrogenchloride In water pH=4.5; | 100% |

| Conditions | Yield |
|---|---|
| In methanol; sodium hydroxide; dichloromethane; chloroform | 100% |
-
-
1711-09-7
3-bromobenzoyl chloride

-
-
104-78-9
diethylaminopropylamine

-
-
349405-27-2
3-bromo-N-(3-diethylamino-propyl)benzamide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In dichloromethane; water at 0 - 20℃; for 18h; Cooling with ice; Inert atmosphere; | 100% |
| With sodium hydroxide In dichloromethane; water at 0 - 20℃; for 18h; Product distribution / selectivity; Inert atmosphere; | 99% |
-
-
53063-03-9
2-Chloro-3-diisopropylaminocarbonylpyridine

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| at 150℃; for 1h; | 99% |
-
-
104-78-9
diethylaminopropylamine

-
-
173961-02-9
4-(4-Methoxy-phenyl)-3-oxiranylmethoxymethyl-butan-1-ol

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Heating; | 99% |
-
-
108-24-7
acetic anhydride

-
-
104-78-9
diethylaminopropylamine

-
-
32039-09-1
3-diethylaminopropyl-1-acetamide

| Conditions | Yield |
|---|---|
| With pyridine for 18h; Ambient temperature; | 99% |
-
-
104-78-9
diethylaminopropylamine

-
-
173961-03-0
4-Benzo[1,3]dioxol-5-yl-3-oxiranylmethoxymethyl-butan-1-ol

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: C38H32Cl2N12O10S2(2-)*2Na(1+); diethylaminopropylamine In water at 70 - 98℃; for 4h; Stage #2: With hydrogenchloride In water pH=2.0; | 98% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
104-78-9
diethylaminopropylamine

-
-
1360158-43-5
tert-butyl 3-(diethylamino)propylcarbamate

| Conditions | Yield |
|---|---|
| at 20℃; | 98% |
-
-
17669-01-1, 56190-51-3, 15853-98-2
(+)-usnic acid

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| In ethanol for 3h; Reflux; | 98% |
-
-
49627-27-2
sulindac sulfide

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; triethylamine In acetonitrile at 20℃; Inert atmosphere; | 98% |
-
-
21814-48-2
1-chloro-7-methoxy-4-nitro-10H-acridin-9-one

-
-
104-78-9
diethylaminopropylamine

-
-
99140-35-9
1-(3-Diethylamino-propylamino)-7-methoxy-4-nitro-10H-acridin-9-one

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 60℃; for 0.5h; | 97% |
-
-
185408-97-3
2-chloro-N-phenyl-9H-purin-6-amine

-
-
104-78-9
diethylaminopropylamine

-
-
903583-21-1
2-(N,N-diethylamino-3-propylamino)-6-phenylamino-purine

| Conditions | Yield |
|---|---|
| at 110℃; for 17h; | 97% |
-
-
38194-50-2
Sulindac

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; triethylamine In acetonitrile at 20℃; Inert atmosphere; | 97% |
-
-
864244-98-4
4-(2-methyl-4-nitrophenoxy)pyridin-2-amine

-
-
104-78-9
diethylaminopropylamine

-
-
864245-03-4
1-(3-diethylaminopropyl)-3-[4-(2-methyl-4-nitrophenoxy)pyridin-2-yl]urea

| Conditions | Yield |
|---|---|
| Stage #1: 4-(2-methyl-4-nitrophenoxy)pyridin-2-amine; phenyl chloroformate With triethylamine In tetrahydrofuran at 0℃; for 0.5h; Stage #2: diethylaminopropylamine In N,N-dimethyl-formamide at 20℃; for 2h; | 96.9% |
-
-
2150-37-0
methyl 3,5-dimethoxybenzoate

-
-
104-78-9
diethylaminopropylamine

-
-
59741-89-8
methyl 1-(2-bromoethyl)-3,5-dimethoxycyclohexa-2,5-diene-1-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: methyl 3,5-dimethoxybenzoate With ammonia; lithium; tert-butyl alcohol In tetrahydrofuran at -78℃; Birch reduction alkylation; Stage #2: diethylaminopropylamine In tetrahydrofuran | 96% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 0.5h; | 96% |
| In methanol at 20℃; for 72h; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: ortho-nitrofluorobenzene; diethylaminopropylamine Stage #2: With hydrogenchloride In isopropyl alcohol | 96% |
-
-
78676-35-4
benzo[b]thiophene-3-carbonyl azide

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| In benzene Heating; | 95% |
-
-
95072-47-2
1-(chloro(piperidin-1-yl)methylene)piperidin-1-ium chloride

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile Inert atmosphere; Reflux; | 95% |
-
-
56043-45-9, 13829-06-6
N,N,N',N'-tetramethylchlorformamidinium chloride

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile Inert atmosphere; Reflux; | 95% |
| With triethylamine In acetonitrile at 0 - 80℃; for 5h; Inert atmosphere; Schlenk technique; |
-
-
104-78-9
diethylaminopropylamine

-
-
104554-43-0
4-(3,3,4,4,5,5,5-heptafluoro-2,2-bis(trifluoromethyl)pentyl)benzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-(3,3,4,4,5,5,5-heptafluoro-2,2-bis(trifluoromethyl)pentyl)benzoic acid With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane for 0.166667h; Cooling; Stage #2: diethylaminopropylamine In dichloromethane at 20℃; for 5h; | 95% |
| Stage #1: 4-(3,3,4,4,5,5,5-heptafluoro-2,2-bis(trifluoromethyl)pentyl)benzoic acid With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0℃; for 0.166667h; Stage #2: diethylaminopropylamine In dichloromethane at 20℃; for 5h; | 91% |
-
-
10328-92-4
N-Methylisatoic anhydride

-
-
104-78-9
diethylaminopropylamine

-
-
158091-48-6
N-Methyl-N'-(3-diethylaminopropyl)anthranilic amide

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 70℃; for 8h; | 94% |
-
-
104-78-9
diethylaminopropylamine

-
-
220822-21-9
3-(2,6-dichlorophenyl)-7-fluoro-1-methyl-1,6-naphthyridin-2(1H)-one

| Conditions | Yield |
|---|---|
| In various solvent(s) for 17h; Amination; Heating; | 94% |
-
-
24066-82-8
ethyl 2-isothiocyanatoacetate

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| In Petroleum ether for 4h; | 94% |

-
-
104-78-9
diethylaminopropylamine

-
-
1335015-53-6
N-(3-(diethylamino)propyl)-9-bromo-5,12-dioxo-5,12-dihydroindolizino[2,3-g]quinoline-6-carboxamide

| Conditions | Yield |
|---|---|
| With dmap In chloroform for 5h; Reflux; | 94% |
-
-
70976-93-1
N,N,N′,N′-tetraethylchloroformamidinium chloride

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile Inert atmosphere; Reflux; | 94% |
| With triethylamine In acetonitrile at 0 - 80℃; for 5h; Inert atmosphere; Schlenk technique; |
-
-
642491-85-8
6,7-difluoro-2,3-dihydropyrrolo[2,1-b]quinazolin-9(1H)-one

-
-
104-78-9
diethylaminopropylamine

| Conditions | Yield |
|---|---|
| at 100℃; for 12h; Sealed tube; | 94% |
| at 100℃; for 12h; Sealed tube; | 74% |
| With sodium carbonate In N,N-dimethyl-formamide at 140℃; for 2h; regioselective reaction; | 71% |
-
-
446-33-3
3-fluoro-6-nitrotoluene

-
-
104-78-9
diethylaminopropylamine

-
-
329943-33-1
N1,N1-diethyl-N3-(3-methyl-4-nitrophenyl)-1,3-propanediamine

| Conditions | Yield |
|---|---|
| With pyridine; sodium hydrogencarbonate for 3h; Heating; | 93% |
3-Diethylaminopropylamine Chemical Properties
Product Name:3-Diethylaminopropylamine
The MF of 3-Diethylaminopropylamine(104-78-9) is C7H18N2.
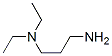
The MW of 3-Diethylaminopropylamine(104-78-9) is 130.23.
Synonyms:1-(Diethylamino)propylamine-3;1,3-Propanediamine,N,N-diethyl-;3-(N,N-Diethylamino)-1-propylamine;N-(3-Diethylaminopropyl)amine;3-Propanediamine,N,N-diethyl-1;Diethylaminotrimethylenamine;N,N-(Diethylamino)propylamine
EINECS: 203-236-4
Mol File: 104-78-9.mol
Surface Tension: 30.8 dyne/cm
Density: 0.84 g/ml
Flash Point: 58.9 °C
Enthalpy of Vaporization: 40.71 kJ/mol
Boiling Point: 170.7 °C at 760 mmHg
Vapour Pressure: 1.45 mmHg at 25°C
Water Solubility: MISCIBLE
Sensitive: Air Sensitive
BRN: 741879
CAS DataBase Reference: 104-78-9(CAS DataBase Reference)
NIST Chemistry Reference: 104-78-9(NIST)
EPA Substance Registry System: 104-78-9(EPA Substance)
3-Diethylaminopropylamine Uses
3-Diethylaminopropylamine Toxicity Data With Reference
| 1. | skn-rbt 100 µg/24H open | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. | ||
| 2. | orl-rat LD50:1410 mg/kg | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 2/27 ,1967. | ||
| 3. | skn-rbt LD50:750 mg/kg | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. |
3-Diethylaminopropylamine Consensus Reports
3-Diethylaminopropylamine Safety Profile
Safety information of 3-Diethylaminopropylamine(104-78-9):
Hazard Codes:
 C,
C, Xi
Xi Risk Statements: 10-21/22-34-43
R10:Flammable ;
R21/22:Harmful in contact with skin and if swallowed ;
R34:Causes burns ;
R43:May cause sensitization by skin contact ;
Safety Statements: 26-36/37/39-45
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice ;
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection ;
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) ;
RIDADR: UN 2684 3/PG 3
WGK Germany: 1
RTECS: TX7350000
HazardClass: 3
PackingGroup: III
HS Code: 29212900
3-Diethylaminopropylamine Standards and Recommendations
3-Diethylaminopropylamine Specification
Related Products
- 3-Diethylaminopropylamine
- 10478-99-6
- 104797-47-9
- 104798-53-0
- 104801-96-9
- 104802-52-0
- 104-80-3
- 10480-34-9
- 1048-05-1
- 1048-08-4
- 104809-67-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View