-
Name
5'-O-Dimethoxytrityl-deoxythymidine
- EINECS 255-003-1
- CAS No. 40615-39-2
- Article Data163
- CAS DataBase
- Density 1.272 g/cm3
- Solubility 3mg/L at 20℃
- Melting Point 114-116 °C (subl.)(lit.)
- Formula C31H32N2O7
- Boiling Point 618.41°C (rough estimate)
- Molecular Weight 544.604
- Flash Point
- Transport Information
- Appearance White powder
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Thymidine,5'-O-[a,a-bis(p-methoxyphenyl)benzyl]- (7CI);5'-O-(4,4'-Dimethoxytrityl)-2'-deoxythymidine;5'-O-(4,4'-Dimethoxytrityl)thymidine;5'-O-(Di-p-methoxytrityl)thymidine;5'-O-(p,p'-Dimethoxytrityl)thymidine;
- PSA 112.01000
- LogP 3.51930
Synthetic route
-
-
40615-36-9
4,4'-dimethoxytrityl chloride

-
-
50-89-5
thymidine

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With pyridine; dmap at 20℃; for 5h; | 97% |
| With pyridine; dmap at 20℃; for 24h; | 96% |
| With pyridine at 20℃; for 12h; | 96% |
-
-
289712-59-0
2-Azidomethyl-benzoic acid (2R,3S,5R)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-5-(5-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-tetrahydro-furan-3-yl ester

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With diphenyl(methyl)phosphine In 1,4-dioxane; water at 20℃; for 0.25h; Product distribution; Further Variations:; Catalysts; Reaction partners; Reagents; Solvents; reaction times; | 96% |

-
-
50-89-5
thymidine

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With 2,6-di-tert-butyl-4-methylpyridine In nitromethane at 20℃; for 17h; or pyridine, other solvents; | 93% |
| With lithium carbonate; N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; Heating; | 74% |
-
-
40615-36-9
4,4'-dimethoxytrityl chloride

-
-
50-89-5
thymidine

-
A
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
B
-
52417-09-1
3'-O-(4,4'-dimethoxytriphenylmethyl)-5'-O-fluorenylmethoxycarbonylthymidine

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 1.5h; | A 93% B n/a |
| With pyridine at 20℃; for 1.5h; | A 93% B n/a |
| With pyridine at 20℃; for 1.5h; | A 93% B n/a |

-
-
110-86-1
pyridine

-
-
40615-36-9
4,4'-dimethoxytrityl chloride

-
-
50-89-5
thymidine

-
A
-
847699-97-2
benzoic acid 5-(4-benzoylamino-5-methyl-2-oxo-2H-pyrimidin-1-yl)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-tetrahydro-furan-3-yl ester

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| at 20℃; for 1.5h; | A n/a B 93% |

-
-
40615-36-9
4,4'-dimethoxytrityl chloride


-
A
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
B
-
52417-09-1
3'-O-(4,4'-dimethoxytriphenylmethyl)-5'-O-fluorenylmethoxycarbonylthymidine

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 0.5h; | A 93% B n/a |
| With pyridine at 20℃; for 1.5h; | A 93% B n/a |

-
-
342890-03-3
1-((2R,4S,5R)-5-[Bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-4-{[(E)-2-(3-hydroxy-naphthalen-2-yl)-vinyl]-diisopropyl-silanyloxy}-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione

-
A
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| In methanol for 0.75h; Irradiation; | A n/a B 92% |
| In methanol for 0.75h; Irradiation; | A 92% B 91% |
-
-
3868-27-7
triethylammonium 5'-O-dimethoxytritylthymidine 3'-H-phosphonate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With pivaloyl chloride; ethylene glycol at 0℃; for 0.333333h; Hydrolysis; | 91% |

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With tri-n-butyl-tin hydride In toluene at 115℃; for 0.0833333h; | 82% |

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran; water for 3h; Ambient temperature; | 73% |
-
-
40615-35-8
4,4'-dimethoxytrityl alcohol

-
-
50-89-5
thymidine

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With ambelyst-15 In dichloromethane for 4h; Reflux; Molecular sieve; regioselective reaction; | 70% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 20℃; for 2h; Inert atmosphere; Schlenk technique; | 61% |
-
-
817623-11-3
2,2'-anhydro-1-[2'-deoxy-5'-O-(4,4'-dimethoxytrityl)-β-D-arabinofuranosyl-5-methyluridine]

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With 15-crown-5; sodium bis(2-methoxyethoxy)aluminium dihydride In 1,2-dimethoxyethane; toluene at 0 - 20℃; for 21h; Product distribution / selectivity; | 35% |
| Stage #1: 2,2'-anhydro-1-[2'-deoxy-5'-O-(4,4'-dimethoxytrityl)-β-D-arabinofuranosyl-5-methyluridine] With sodium bis(2-methoxyethoxy)aluminium dihydride In toluene at 0 - 5℃; for 2.91667h; Stage #2: With water at 5℃; for 0.5h; Product distribution / selectivity; | 21% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 20℃; Product distribution; |
-
-
157793-51-6, 165879-39-0, 165879-40-3
3'-O-(4,4'-dimethoxytrityl)thymidin-5'-yl 5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl ethyl phosphonate

-
A
-
76054-81-4
3'-O-(4,4'-dimethoxytrityl)thymidine

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With ethanolamine In pyridine for 0.25h; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 20℃; for 23h; Product distribution; |
-
-
141-43-5
ethanolamine

-
-
105784-94-9
5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl ethyl phosphonate

-
A
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| In pyridine |

-
-
105784-94-9
5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl ethyl phosphonate

-
A
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
B
-
762-04-9
phosphonic acid diethyl ester

| Conditions | Yield |
|---|---|
| With ethanol In pyridine for 96h; Yield given. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 20℃; Product distribution; |
-
-
95100-66-6
C49H52ClN4O17P2(1-)

-
A
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 20℃; Product distribution; |
-
-
102987-83-7
5′-O-(4,4′-dimethoxytrityl)thymidine-3′-O-[2-cyanoethyl]phosphonate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With 1H-imidazole In methanol for 72h; Ambient temperature; Yield given; |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran for 8h; Product distribution; other bases (piperidine, TBAF, conc. aq. NH3); mechanism of cleavage of succinyl-anchored nucleotides under basic conditions; hydrolysis and ammonolysis with aq. NH3; |
-
-
216568-80-8
6-(2-Methylamino-benzoylamino)-4-oxo-hexanoic acid (2R,3S,5R)-2-hydroxymethyl-5-(5-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-tetrahydro-furan-3-yl ester

-
A
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With pyridine; water; acetic acid; hydrazine for 0.0833333h; Ambient temperature; |
-
-
220928-40-5
5'-O-(4,4'-dimethoxytrityl)-3'-C-(acetyl)thymidine

-
A
-
40615-39-2, 104766-16-7, 131607-26-6, 131607-27-7, 112501-53-8
1-((2R,4R,5R)-5-((bis(4-methoxyphenyl)(phenyl)methoxy)methyl)-4-hydroxytetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)- dione

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With t-butyltin In acetonitrile at 20℃; for 3h; Product distribution; Irradiation; |
-
-
220928-41-6
1-(5-O-(4,4'-dimethoxytrityl)-3-C-acetyl-2-deoxy-β-D-threo-pentofuranosyl)thymine

-
A
-
40615-39-2, 104766-16-7, 131607-26-6, 131607-27-7, 112501-53-8
1-((2R,4R,5R)-5-((bis(4-methoxyphenyl)(phenyl)methoxy)methyl)-4-hydroxytetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)- dione

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With t-butyltin In acetonitrile at 20℃; for 3h; Product distribution; Irradiation; |
-
-
220928-52-9
1-(5-O-(4,4'-dimethoxytrityl)-3-C-(1,1-dimethylpropanoyl)-2-deoxy-β-D-threo-pentofuranosyl)thymine

-
A
-
40615-39-2, 104766-16-7, 131607-26-6, 131607-27-7, 112501-53-8
1-((2R,4R,5R)-5-((bis(4-methoxyphenyl)(phenyl)methoxy)methyl)-4-hydroxytetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)- dione

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With t-butyltin In acetonitrile at 20℃; for 1h; Product distribution; Irradiation; |
-
-
220928-57-4
5'-O-(4,4'-dimethoxytrityl)-3'-C-(2,2-dimethylpropanoyl)thymidine

-
A
-
40615-39-2, 104766-16-7, 131607-26-6, 131607-27-7, 112501-53-8
1-((2R,4R,5R)-5-((bis(4-methoxyphenyl)(phenyl)methoxy)methyl)-4-hydroxytetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)- dione

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With t-butyltin In acetonitrile at 20℃; for 1h; Product distribution; Irradiation; |
-
-
220928-53-0
1-(5-O-(4,4'-dimethoxytrityl)-3-C-benzoyl-2-deoxy-β-D-threo-pentofuranosyl)thymine

-
A
-
40615-39-2, 104766-16-7, 131607-26-6, 131607-27-7, 112501-53-8
1-((2R,4R,5R)-5-((bis(4-methoxyphenyl)(phenyl)methoxy)methyl)-4-hydroxytetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)- dione

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With t-butyltin In acetonitrile at 20℃; for 2h; Product distribution; Irradiation; |
-
-
220928-58-5
5'-O-(4,4'-dimethoxytrityl)-3'-C-(benzoyl)thymidine

-
A
-
40615-39-2, 104766-16-7, 131607-26-6, 131607-27-7, 112501-53-8
1-((2R,4R,5R)-5-((bis(4-methoxyphenyl)(phenyl)methoxy)methyl)-4-hydroxytetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)- dione

-
B
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With t-butyltin In acetonitrile at 20℃; for 2h; Product distribution; Irradiation; |

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With pyridine for 8h; Hydrolysis; |
-
-
123-76-2
levulinic acid

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
93134-37-3
5'-O-(4,4'-dimethoxytrityl)-3'-O-levulinoyl-2'-deoxythymidine

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In tetrahydrofuran at 20℃; for 1.5h; | 100% |
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In 1,4-dioxane at 20℃; for 12h; | 99% |
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In 1,4-dioxane for 2.5h; | 98% |
-
-
108554-72-9
(allyloxy)bis(diisopropylamino)phosphine

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
104655-82-5
5'-O-(p,p'-dimethoxytrityl)thymidine 3'-(allyl N,N-diisopropylphosphoramidite)

| Conditions | Yield |
|---|---|
| With 1H-tetrazole; diisopropylamine In acetonitrile at 25℃; for 1.5h; | 100% |
| With N,N-diisopropylamine tetrazolide In dichloromethane for 0.166667h; Ambient temperature; |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
50-89-5
thymidine

| Conditions | Yield |
|---|---|
| With zinc dibromide In nitromethane for 0.0166667h; Ambient temperature; | 100% |
| With zinc dibromide In nitromethane Kinetics; Ambient temperature; in various solvent:CH2Cl2, THF, Acetone, Dioxane; other 5'-deoxytritylnucleosides; | 100% |
| With β-naphthol In dichloromethane for 0.166667h; Product distribution / selectivity; Irradiation with mercury arc lamp; | 97% |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
83467-48-5
1-[(2R,4S,5R)-4-(tert-butyldiphenylsilanyloxy)-5-hydroxymethyltetrahydrofuran-2-yl]-5-methyl-1H-pyrimidine-2,4-dione

| Conditions | Yield |
|---|---|
| Stage #1: 5'-O-(4-4'-dimethoxytrityl)thymidine; tert-butylchlorodiphenylsilane With 1H-imidazole In dichloromethane at 20℃; for 2h; Stage #2: With toluene-4-sulfonic acid In methanol | 100% |
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 72h; | 75% |
| With benzoic acid 2) CH2Cl2, MeOH; Multistep reaction; |
-
-
103930-70-7
(i-Pr2N)2POC(CH3)2CH2CN

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
111570-97-9
1,1-dimethyl-2-cyanoethyl 5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl N,N-diisopropylphosphoramidite

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 24h; Inert atmosphere; | 100% |
-
-
108-30-5
succinic acid anhydride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
121-44-8
triethylamine

-
-
402944-22-3
5'-O-(4,4'-dimethoxytrityl)-2'-deoxythymidine-3'-O-succinate triethylamine salt

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 16h; Inert atmosphere; | 100% |
| Stage #1: succinic acid anhydride; 5'-O-(4-4'-dimethoxytrityl)thymidine With dmap In dichloromethane at 20℃; for 1h; Inert atmosphere; Stage #2: triethylamine In methanol; dichloromethane | 94% |

-
-
108-30-5
succinic acid anhydride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
402944-22-3
5'-O-(4,4'-dimethoxytrityl)-2'-deoxythymidine-3'-O-succinate triethylamine salt

| Conditions | Yield |
|---|---|
| Stage #1: succinic acid anhydride; 5'-O-(4-4'-dimethoxytrityl)thymidine With triethylamine In dichloromethane at 20℃; for 16h; Inert atmosphere; Stage #2: triethylammonium dihydrogen phosphate In dichloromethane; water | 100% |

-
-
16383-57-6
bis(prop-2-en-1-yl)chlorophosphonate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
112677-90-4
Phosphoric acid diallyl ester (2R,3S,5R)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-5-(5-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-tetrahydro-furan-3-yl ester

| Conditions | Yield |
|---|---|
| With tert-butylmagnesium chloride In tetrahydrofuran for 1h; Ambient temperature; | 99% |
-
-
108-24-7
acetic anhydride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
40615-37-0
3'-O-acetyl-5'-O-[bis(4-methoxyphenyl)(phenyl)methyl]-2'-deoxy-3,4-dihydrothymidine

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; Inert atmosphere; | 99% |
| With pyridine 1.) 0 deg C, 4 h, 2.) RT, 22 h; | 96% |
| In pyridine | 90% |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
191151-90-3
O-(2-cyanoethyl) N,N-diisopropylfluorophosphoramidite

-
-
171081-12-2
O-[5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl] O-(2-cyanoethyl) phosphorofluoridite

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In tetrahydrofuran at 20℃; for 1h; | 99% |
| With chloro-trimethyl-silane In tetrahydrofuran for 1h; Ambient temperature; | 97% |
| With chloro-trimethyl-silane Yield given; |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
289681-49-8
1-[(2R,4S,5R)-5-{[bis(4-methoxyphenyl)(phenyl)methoxy]methyl}-4-{[(1,1-dimethylethyl)diphenylsilyl]oxy}tetrahydrofuran-2-yl]-5-methylpyrimidine-2,4(1H,3H)-dione

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide for 24h; | 99% |
| Stage #1: 5'-O-(4-4'-dimethoxytrityl)thymidine With 1H-imidazole In dichloromethane for 0.0833333h; Stage #2: tert-butylchlorodiphenylsilane In dichloromethane at 20℃; for 3h; | 99% |
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 12h; | 92% |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With pyridine; dmap at 20℃; | 99% |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
2283-11-6
hexaethylphosphoric triamide

-
A
-
104655-76-7
5'-O-dimethoxytrityl thymidine 3'-O-phosphorbisdiethylamidite

| Conditions | Yield |
|---|---|
| 1H-tetrazole; diisopropylamine In dichloromethane; water; acetonitrile for 0.25h; Ambient temperature; var. amine catalysts.; Title compound not separated from byproducts; | A 98.5% B 0.5% |
| 1H-tetrazole In dichloromethane; water; acetonitrile for 0.25h; Ambient temperature; var. amine catalysts.; Title compound not separated from byproducts; | A 71.5% B 25.9% |

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
80971-33-1
5'-O-(4,4'-dimethoxytrityl)-3'-O-(t-butyldimethylsilyl)-2'-deoxythymidine

| Conditions | Yield |
|---|---|
| With 1H-imidazole In tetrahydrofuran silylation; | 98% |
| With 1-methyl-1H-imidazole; iodine In tetrahydrofuran at 20℃; for 1h; | 98% |
| With 1-methyl-1H-imidazole; iodine In tetrahydrofuran at 20℃; for 1h; | 98% |
-
-
67941-87-1
cyclohexylammonium S,S-diphenyl phosphorodithioate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
80585-43-9
Dithiophosphoric acid O-[(2R,3S,5R)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-5-(5-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-tetrahydro-furan-3-yl] ester S,S'-diphenyl ester

| Conditions | Yield |
|---|---|
| With dapsone In pyridine for 4h; Ambient temperature; | 98% |
| In pyridine for 6h; | 87% |
| With 4,6-dimethoxybenzene-1,3-bis(sulfonyl) chloride In pyridine 1.) 1h, 2.) 7h, RT, 3.) ice cubes, 20 min; | 87% |
-
-
75302-66-8
dimorpholinophosphinous chloride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
114360-62-2
C39H47N4O9P

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane for 0.5h; | 98% |
-
-
81087-19-6
p-chlorophenyl N-(p-methoxyphenyl)chlorophosphoramidate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
81087-39-0
(4-Methoxy-phenyl)-phosphoramidic acid (2R,3S,5R)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-5-(5-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-tetrahydro-furan-3-yl ester 4-chloro-phenyl ester

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole In 1,4-dioxane at 25℃; for 2h; | 98% |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
153413-65-1
O,O'-bis-(4-nitrophenyl) N,N-diisopropylphosphoramidite

-
-
153413-66-2, 162681-43-8, 162681-44-9
O-(5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl) O-(4-nitrophenyl) N,N-diisopropylphosphoramidite

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; for 0.166667h; | 98% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; for 0.166667h; | 98% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; | 95% |
-
-
103408-53-3
allyloxybis(N,N-dimethylamino)phosphine

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With 1-methylimidazolium trifluoromethanesulfonate In acetonitrile at 25℃; for 1.5h; | 98% |
-
-
108-30-5
succinic acid anhydride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 3h; | 98% |

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
151782-47-7
O-[5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl] O-methyl (O-4-nitrophenyl)-phosphate

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; Inert atmosphere; | 98% |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
327596-87-2
N,N,N',N'-tetraisopropyl-O-[2-[N-isopropyl-N-(4-methoxybenzoyl)amino]ethyl] phosphordiamidite

-
-
291299-79-1, 291299-99-5, 291300-01-1
5'-O-(4,4'-dimethoxytrityl)-3'-O-(N,N-diisopropylamino)-[2-[N-isopropyl-N-(4-methoxybenzoyl)amino]ethoxy]phosphinyl-2'-deoxythymidine

| Conditions | Yield |
|---|---|
| With 1H-tetrazole In dichloromethane; acetonitrile at 20℃; for 2h; | 97.6% |
-
-
108-30-5
succinic acid anhydride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
74405-40-6
5’-O-(4,4’-dimethoxytriphenylmethyl)thymidine-3’-O-succinate

| Conditions | Yield |
|---|---|
| With acetic acid; 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane at 20℃; for 0.75h; Inert atmosphere; | 97% |
| With pyridine; dmap at 50℃; for 4h; | |
| With pyridine; dmap; triethylamine In dichloromethane for 3h; Ambient temperature; | |
| With pyridine; dmap at 20℃; for 20h; | |
| With pyridine; dmap |
-
-
102691-36-1
N,N,N',N'-tetraisopropyl 2-cyanoethylphosphorodiamidite

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
98796-51-1
5'-O-(4,4'-dimethoxytrityl)-2'-deoxythymidine-3'-O-[O-(2-cyanoethyl)-N,N'-diisopropylphosphoramidite]

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In tetrahydrofuran for 1h; Ambient temperature; | 97% |
| With N,N-diisopropylamine tetrazolide In dichloromethane for 4h; Ambient temperature; | 95% |
| With 1H-tetrazole; N-ethyl-N,N-diisopropylamine In dichloromethane for 16h; Ambient temperature; | 95% |

| Conditions | Yield |
|---|---|
| With (iPr)2Et2N In dichloromethane for 0.166667h; Ambient temperature; | 97% |
-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
530-62-1
1,1'-carbonyldiimidazole

-
-
111462-16-9
5'-O-dimethoxytrityl-3'-O-(1-imidazolylcarbonyl)thymidine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 4h; | 97% |
| In acetonitrile for 1h; | 90% |
| In acetonitrile for 2h; | |
| With dmap In dichloromethane at 20℃; for 14h; | |
| With dmap In dichloromethane |
-
-
130858-91-2
9H-fluoren-9-y1methyl (2-fluoro-2-oxoethyl)carbamate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane for 4h; Ambient temperature; | 97% |
-
-
142061-92-5, 145434-41-9, 130858-92-3
N-(9-fluorenylmethoxycarbonyl)-L-alanyl fluoride

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane for 4h; Ambient temperature; | 97% |
-
-
7693-46-1
4-Nitrophenyl chloroformate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
156939-39-8
Carbonic acid (2R,3S,5R)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-5-(5-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-tetrahydro-furan-3-yl ester 4-nitro-phenyl ester

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane Acylation; | 97% |
| With pyridine In dichloromethane at 20℃; for 16h; | 62% |
| With pyridine In dichloromethane at 0 - 20℃; | 62% |
| With pyridine at 80℃; for 20h; |
5'-O-dimethoxytrityl-deoxythymidine Chemical Properties
Molecular Structure of 5'-O-dimethoxytrityl-deoxythymidine (CAS NO.40615-39-2):
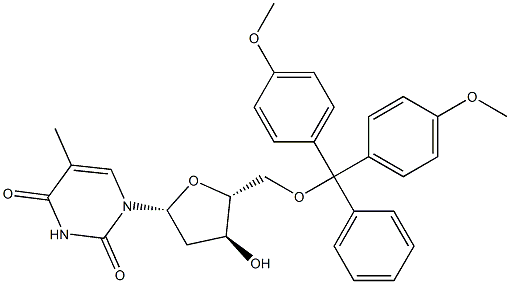
IUPAC Name: 1-[(2R,4S,5R)-5-[[bis(4-methoxyphenyl)-phenylmethoxy]methyl]-4-hydroxyoxolan-2-yl]-5-methylpyrimidine-2,4-dione
Cas register number: 40615-39-2
Molecular formula: C31H32N2O6
Molecular Weight: 528.6
Density: 1.272 g/cm3
Index of Refraction: 1.602
Molar Refractivity: 146.9 cm3
Molar Volume: 427.8 cm3
Polarizability: 58.23×10-24cm3
Surface Tension: 51.6 dyne/cm
Appearance: White Powder
Product Categories: Biochemistry;Nucleosides and their analogs;Nucleosides, Nucleotides & Related Reagents;Bases & Related Reagents;Carbohydrates & Derivatives;Nucleotides.
5'-O-dimethoxytrityl-deoxythymidine Safety Profile
Safety Statements about 5'-O-dimethoxytrityl-deoxythymidine (CAS NO.40615-39-2):
Hazard Codes:
Xi:
R36/37/38: Irritating to eyes, respiratory system and skin.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clothing.
F1: Sensitive to air and humidity.
F3: Hygroscopic.
F10: Keep under argon.
WGK Germany: 3
Hazard Note: Irritant
5'-O-dimethoxytrityl-deoxythymidine Specification
5'-O-dimethoxytrityl-deoxythymidine , its cas register number is 40615-39-2. It also can be called 5’-O-[bis(4-methoxyphenyl)phenylmethyl]-thymidin ; 5'-Dimethoxytrityl thymidine ; 5'-O-(4,4'-dimethoxytrityl)-2'-deoxythymidine ; 5'-O-[Bis(4-methoxyphenyl)benzyl]-2'-deoxythymidine ; 5'-O-(4,4'-dimethoxytrityl)thymidine ; 5-O-(4,4-dimethoxytrityl)thymidine ; 5'-O-dimethoxytrityl-deoxythymidine ; 5'-O-(dimethoxytrityl)-thymidine . Some useful informationabout Handling and Storage: Wash thoroughly after handling. Remove contaminated clothing and wash before reuse.Avoid contact with eyes, skin, and clothing. Avoid ingestion and inhalation.Keep refrigerated. (Store below 4°C/39°F.) Keep container closed when not in use. Store in a dry area.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View