-
Name
Acridin-9-amine hydrochloride
- EINECS 205-145-5
- CAS No. 134-50-9
- Density 1.268g/cm3
- Solubility
- Melting Point
- Formula C13H10 N2 . Cl H
- Boiling Point 413.5°Cat760mmHg
- Molecular Weight 230.71
- Flash Point 233.2°C
- Transport Information
- Appearance Pale yellow crystals. One of the most highly fluorescent substances.
- Safety Poison by ingestion. Mutation data reported. See also AMINES. When heated to decomposition it emits very toxic fumes of HCl and NOx.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 9-Acridinamine,monohydrochloride (9CI); Acridine, 9-amino-, monohydrochloride (8CI);9-Aminoacridine hydrochloride; 9-Aminoacridine monohydrochloride; Acramineyellow; Aminacrine hydrochloride; Monacrin hydrochloride
- PSA 38.91000
- LogP 4.35340
Synthetic route
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
110166-26-2
aminacrine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; ethyl acetate for 0.25h; Concentration; Reagent/catalyst; Solvent; | 100% |

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.25h; Heating; | 99% |
-
-
52824-42-7
N-(7-bromoheptyl)phthalimide

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 74% |
-
-
108-24-7
acetic anhydride

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
23043-52-9
N-(acridin-9-yl)acetamide

| Conditions | Yield |
|---|---|
| In pyridine at 70℃; for 24h; | 67.8% |
-
-
82484-59-1
4-bis-(β-chloroethyl)aminophenyl isocyanate

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
1036724-69-2
1-acridin-9-yl-3-{4-[bis(2-chloroethyl)amino]phenyl}urea

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 20℃; for 18h; | 60% |
-
-
93667-91-5
2-(9-bromononyl)isoindoline-1,3-dione

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 55.5% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
1428587-11-4
C20H15BN2O3

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; triethylamine In dimethyl sulfoxide at 20 - 40℃; for 49h; | 53% |
| Stage #1: acridin-9-amine hydrochloride With triethylamine In water; dimethyl sulfoxide at 20℃; for 1h; Stage #2: 3-Carboxyphenylboronic acid With 1,8-diazabicyclo[5.4.0]undec-7-ene In water; dimethyl sulfoxide at 40℃; for 48h; | 53% |
-
-
17702-83-9
N-8-bromooctylphthalimide

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 18.5% |
-
-
134-50-9
acridin-9-amine hydrochloride

-
A
-
23045-35-4
2-nitro-9-aminoacridine

-
B
-
157996-62-8
9-amino-2,7-dinitroacridine

| Conditions | Yield |
|---|---|
| With sodium nitrate; sulfuric acid at 0℃; for 0.5h; | A 9.6% B 15% |
| With sodium nitrate; sulfuric acid at 0℃; for 0.5h; | A 9.6% B 15% |
-
-
24566-79-8
2-(6-bromohexyl)isoindole-1,3-dione

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 15% |
-
-
134-50-9
acridin-9-amine hydrochloride

-
B
-
157996-60-6
9-amino-2,7-dibromoacridine

| Conditions | Yield |
|---|---|
| With bromine In chloroform for 2h; Ambient temperature; | A 11.1% B 2.4% |
-
-
1111-97-3
3-chloro-3-methylbut-1-yne

-
-
134-50-9
acridin-9-amine hydrochloride

-
A
-
133286-71-2
10-(3-Methyl-1,2-butadienyl)-9(10H)-iminoacridin

-
B
-
133286-69-8
9-(3-Methyl-1,2-butadienyl)-9-aminoacridin

-
C
-
133286-70-1
9-(1,1-Dimethyl-2-propinyl)-9-aminoacridin

-
D
-
133286-72-3
10-(1,1-Dimethyl-2-propinyl)-9(10H)-iminoacridin

| Conditions | Yield |
|---|---|
| With potassium hydroxide; tetrabutylammomium bromide In toluene for 72h; Heating; | A 370 mg B 1.69 g C 40 mg D 490 mg |

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 67.8 percent / pyridine / 24 h / 70 °C 2: 27.2 percent / Br2 / CHCl3 / 2 h / Ambient temperature View Scheme |
-
-
423768-37-0
1-phenyl-1H-pyrazole-5-carbonyl chloride

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
908562-71-0
C23H16N4O

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; for 18h; |
-
-
806608-18-4
4--phenyl-carbamoylchlorid

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
1036724-69-2
1-acridin-9-yl-3-{4-[bis(2-chloroethyl)amino]phenyl}urea

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 16h; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
88-99-3
benzene-1,2-dicarboxylic acid

-
-
1272425-79-2
9-aminoacridiniun ortho-phthalate monohydrate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
65-85-0
benzoic acid

-
-
1272425-77-0
9-aminoacridiniun benzoate monohydrate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
69-72-7
salicylic acid

-
-
109653-41-0
9-aminoacridiniun salicylate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
74-11-3
para-chlorobenzoic acid

-
-
1302149-89-8
9-aminoacridinium 4-chlorobenzoate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
535-80-8
3-chlorobenzoate

-
-
1302149-88-7
9-aminoacridinium 3-chlorobenzoate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
934991-54-5
(acridin-9-ylamino)acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water; ethyl acetate / 0.25 h 2: N,N-dimethyl-formamide / 5 h / 80 - 85 °C View Scheme |
-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| In aq. phosphate buffer at 24.84℃; pH=7.4; Thermodynamic data; Temperature; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
110166-26-2
aminacrine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; ethyl acetate for 0.25h; Concentration; Reagent/catalyst; Solvent; | 100% |

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.25h; Heating; | 99% |
-
-
52824-42-7
N-(7-bromoheptyl)phthalimide

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 74% |
-
-
108-24-7
acetic anhydride

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
23043-52-9
N-(acridin-9-yl)acetamide

| Conditions | Yield |
|---|---|
| In pyridine at 70℃; for 24h; | 67.8% |
-
-
82484-59-1
4-bis-(β-chloroethyl)aminophenyl isocyanate

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
1036724-69-2
1-acridin-9-yl-3-{4-[bis(2-chloroethyl)amino]phenyl}urea

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 20℃; for 18h; | 60% |
-
-
93667-91-5
2-(9-bromononyl)isoindoline-1,3-dione

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 55.5% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
1428587-11-4
C20H15BN2O3

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; triethylamine In dimethyl sulfoxide at 20 - 40℃; for 49h; | 53% |
| Stage #1: acridin-9-amine hydrochloride With triethylamine In water; dimethyl sulfoxide at 20℃; for 1h; Stage #2: 3-Carboxyphenylboronic acid With 1,8-diazabicyclo[5.4.0]undec-7-ene In water; dimethyl sulfoxide at 40℃; for 48h; | 53% |
-
-
17702-83-9
N-8-bromooctylphthalimide

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 18.5% |
-
-
134-50-9
acridin-9-amine hydrochloride

-
A
-
23045-35-4
2-nitro-9-aminoacridine

-
B
-
157996-62-8
9-amino-2,7-dinitroacridine

| Conditions | Yield |
|---|---|
| With sodium nitrate; sulfuric acid at 0℃; for 0.5h; | A 9.6% B 15% |
| With sodium nitrate; sulfuric acid at 0℃; for 0.5h; | A 9.6% B 15% |
-
-
24566-79-8
2-(6-bromohexyl)isoindole-1,3-dione

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 12h; | 15% |
-
-
134-50-9
acridin-9-amine hydrochloride

-
B
-
157996-60-6
9-amino-2,7-dibromoacridine

| Conditions | Yield |
|---|---|
| With bromine In chloroform for 2h; Ambient temperature; | A 11.1% B 2.4% |
-
-
1111-97-3
3-chloro-3-methylbut-1-yne

-
-
134-50-9
acridin-9-amine hydrochloride

-
A
-
133286-71-2
10-(3-Methyl-1,2-butadienyl)-9(10H)-iminoacridin

-
B
-
133286-69-8
9-(3-Methyl-1,2-butadienyl)-9-aminoacridin

-
C
-
133286-70-1
9-(1,1-Dimethyl-2-propinyl)-9-aminoacridin

-
D
-
133286-72-3
10-(1,1-Dimethyl-2-propinyl)-9(10H)-iminoacridin

| Conditions | Yield |
|---|---|
| With potassium hydroxide; tetrabutylammomium bromide In toluene for 72h; Heating; | A 370 mg B 1.69 g C 40 mg D 490 mg |

-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 67.8 percent / pyridine / 24 h / 70 °C 2: 27.2 percent / Br2 / CHCl3 / 2 h / Ambient temperature View Scheme |
-
-
423768-37-0
1-phenyl-1H-pyrazole-5-carbonyl chloride

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
908562-71-0
C23H16N4O

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; for 18h; |
-
-
806608-18-4
4--phenyl-carbamoylchlorid

-
-
134-50-9
acridin-9-amine hydrochloride

-
-
1036724-69-2
1-acridin-9-yl-3-{4-[bis(2-chloroethyl)amino]phenyl}urea

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 16h; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
88-99-3
benzene-1,2-dicarboxylic acid

-
-
1272425-79-2
9-aminoacridiniun ortho-phthalate monohydrate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
65-85-0
benzoic acid

-
-
1272425-77-0
9-aminoacridiniun benzoate monohydrate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
69-72-7
salicylic acid

-
-
109653-41-0
9-aminoacridiniun salicylate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
74-11-3
para-chlorobenzoic acid

-
-
1302149-89-8
9-aminoacridinium 4-chlorobenzoate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
535-80-8
3-chlorobenzoate

-
-
1302149-88-7
9-aminoacridinium 3-chlorobenzoate

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.0166667h; Reflux; |
-
-
134-50-9
acridin-9-amine hydrochloride

-
-
934991-54-5
(acridin-9-ylamino)acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water; ethyl acetate / 0.25 h 2: N,N-dimethyl-formamide / 5 h / 80 - 85 °C View Scheme |
-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| In aq. phosphate buffer at 24.84℃; pH=7.4; Thermodynamic data; Temperature; |
-
-
134-50-9
acridin-9-amine hydrochloride

| Conditions | Yield |
|---|---|
| In aq. phosphate buffer at 24.84℃; pH=7.4; Thermodynamic data; Temperature; |
Acridin-9-amine hydrochloride Chemical Properties
IUPAC Name: Acridin-9-amine hydrochloride
The MF of Acridin-9-amine hydrochloride (CAS NO.134-50-9) is C13H11ClN2.
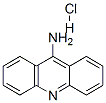
The MW of Acridin-9-amine hydrochloride (CAS NO.134-50-9) is 230.69.
Synonyms of Acridin-9-amine hydrochloride (CAS NO.134-50-9): 9-Aminoacridine hydrochloride ; Acridin-9-amine hydrochloride ; 5-Aminoacridine hydrochloride
Apperance: Pale yellow crystals.One of the most highly fluorescent substances
Flash Point: 233.2 °C
Boiling Point: 413.5 °C
EINECS: 205-145-5
Acridin-9-amine hydrochloride Uses
Acridin-9-amine hydrochloride (CAS NO.134-50-9) is used experimentally as a probe for biological membranes and dna.
Acridin-9-amine hydrochloride Production
Acridin-9-amine hydrochloride forms from treatment of 9-aminoacridine with hydrogen chloride. It is amphoteric (reacts both as a weak acid and as a weak base).
Acridin-9-amine hydrochloride Toxicity Data With Reference
| 1. | mmo-sat 10 µg/plate | KSRNAM Kiso to Rinsho. Clinical Report. 16 (1982),6240. | ||
| 2. | mma-sat 50 µg/plate | KSRNAM Kiso to Rinsho. Clinical Report. 17 (1983),70. | ||
| 3. | dnr-esc 20 µL/disc | MUREAV Mutation Research. 97 (1982),1. | ||
| 4. | dnr-bcs 20 µL/disc | MUREAV Mutation Research. 97 (1982),1. | ||
| 5. | cyt-ham:lng 600 µg/L | GMCRDC Gann Monograph on Cancer Research. 27 (1981),95. | ||
| 6. | dnd-man:lym 10 pph | BIPMAA Biopolymers. 11 (1972),2537. | ||
| 7. | orl-mus LD50:78 mg/kg | 29ZVAB Handbook of Analytical Toxicology ,Sunshine, I., ed., Cleveland, OH.: Chemical Rubber Co.,1969,7 |
Acridin-9-amine hydrochloride Consensus Reports
Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program.
Acridin-9-amine hydrochloride Safety Profile
Poison by ingestion. Mutation data reported. See also AMINES. When heated to decomposition it emits very toxic fumes of HCl and NOx.Safety information of Acridin-9-amine hydrochloride (CAS NO.134-50-9):
Safety Statements
22 Do not breathe dust
24/25 Avoid contact with skin and eyes
WGK Germany 3
Acridin-9-amine hydrochloride Specification
Flash point data for Acridin-9-amine hydrochloride is not available, but Acridin-9-amine hydrochloride is probably combustible.
Related Products
- Acridin-9-amine hydrochloride
- 13450-90-3
- 1345-09-1
- 13450-91-4
- 13450-92-5
- 13450-99-2
- 134510-06-8
- 13451-00-8
- 13451-02-0
- 13451-18-8
- 13451-19-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View