-
Name
AGMATINE SULFATE ENDOGENOUS AGONIST AT
- EINECS 206-187-7
- CAS No. 306-60-5
- Article Data24
- CAS DataBase
- Density 1.2 g/cm3
- Solubility
- Melting Point
- Formula C5H14N4
- Boiling Point 281.4 °C at 760 mmHg
- Molecular Weight 130.193
- Flash Point 124 °C
- Transport Information
- Appearance powder
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Agmatine(6CI);Guanidine, (4-aminobutyl)- (8CI,9CI);(4-Aminobutyl)guanidine;1,4-Butanediamine, N-(aminoiminomethyl)-;1-Amino-4-guanidinobutane;4-Guanidino-1-butanamine;N-(4-Aminobutyl)guanidine;NSC 56332;2-(4-aminobutyl)guanidine;1-(4-Aminobutyl)guanidin;1-Amino-4-guanidinobutane;2-(4-aminobutyl)guanidine;Guanidine, (4-aminobutyl)-;agmatine;
- PSA 87.92000
- LogP 1.09960
Synthetic route

| Conditions | Yield |
|---|---|
| at 80℃; for 0.75h; | 96% |
-
-
23441-10-3
N-Benzyloxycarbonyl-NG-nitroagmatin

-
-
306-60-5
1-(4-aminobutyl)guanidine

| Conditions | Yield |
|---|---|
| With formic acid; palladium on activated charcoal In methanol for 2h; Ambient temperature; | 52% |
| With formic acid; palladium on activated charcoal In methanol at 25℃; for 2h; Yield given; |

| Conditions | Yield |
|---|---|
| durch Escherichia coli; | |
| With Escherichia coli | |
| With E.coli; streptococcus faecalis bei pH 4; | |
| With L-arginine decarboxylase; pyridoxal 5'-phosphate; diothiothreitol In phosphate buffer at 50℃; for 0.166667h; pH=6.5; | |
| With 3,5,5-Trimethylcyclohex-2-en-1-one In isopropyl alcohol at 150℃; for 4h; Autoclave; |

| Conditions | Yield |
|---|---|
| With water | |
| With barium sulfate |
-
-
14527-26-5, 867-44-7
2-methylisothiourea sulphate

-
-
110-60-1
1,4-diaminobutane

-
-
306-60-5
1-(4-aminobutyl)guanidine
-
-
22906-75-8
3,5-Dimethylpyrazole-1-carboxamidine

-
-
18807-73-3
N-benzyloxycarbonyl-1,4-butanediamine hydrochloride

-
-
306-60-5
1-(4-aminobutyl)guanidine

| Conditions | Yield |
|---|---|
| (i) aq. NaOH, EtOH, (ii) aq. HBr; Multistep reaction; |

-
-
306-60-5
1-(4-aminobutyl)guanidine

| Conditions | Yield |
|---|---|
| With saprophytes | |
| With barium dihydroxide; water |

-
-
306-60-5
1-(4-aminobutyl)guanidine

| Conditions | Yield |
|---|---|
| With sulfuric acid Hydrolysis; |

-
-
306-60-5
1-(4-aminobutyl)guanidine

| Conditions | Yield |
|---|---|
| With carbon dioxide; water |

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With tetramethyl ammoniumhydroxide In methanol for 1h; |

| Conditions | Yield |
|---|---|
| In water |
-
-
306-60-5
1-(4-aminobutyl)guanidine


| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In acetonitrile | 95% |

| Conditions | Yield |
|---|---|
| In methanol | 70% |
-
-
87-51-4
indole-3-acetic acid

-
-
306-60-5
1-(4-aminobutyl)guanidine

-
-
66-99-9
β-naphthaldehyde

-
-
76-05-1
trifluoroacetic acid

-
-
949592-43-2
N-(4-guanidinobutyl)-2-(1H-indol-3-yl)-N-((naphthalen-2-yl)methyl)acetamide trifluoroacetic acid salt

| Conditions | Yield |
|---|---|
| Multistep reaction.; | 26% |

-
-
133-32-4
4-indol-3-yl-butyric acid

-
-
306-60-5
1-(4-aminobutyl)guanidine

-
-
66-99-9
β-naphthaldehyde

-
-
76-05-1
trifluoroacetic acid

-
-
949592-47-6
N-(4-guanidinobutyl)-4-(1H-indol-3-yl)-N-((naphthalen-2-yl)methyl)butanamide trifluoroacetic acid salt

| Conditions | Yield |
|---|---|
| Multistep reaction.; | 24% |

-
-
306-60-5
1-(4-aminobutyl)guanidine

-
-
65178-90-7
2-[4-(4-chlorobenzoyl)-phenoxy]-2-methylpropionyl chloride

-
-
1094098-88-0
2-(4-(4-chlorobenzoyl)phenoxy)-N-(4-guanidinobutyl)-2-methylpropanamide

| Conditions | Yield |
|---|---|
| With pyridine | 10% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; 2-hydroxyethanethiol; sodium sulfite In water at 100℃; for 0.166667h; |
-
-
306-60-5
1-(4-aminobutyl)guanidine

-
-
14049-14-0
γ-guanidino butyraldehyde

| Conditions | Yield |
|---|---|
| With potassium phosphate buffer (pH: 7.0) at 35℃; for 0.333333h; agmatine oxidase from Penicillium chrysogenum; |
-
-
306-60-5
1-(4-aminobutyl)guanidine

-
A
-
110-15-6
succinic acid

-
B
-
463-00-3
4-guanidinobutyric acid

-
C
-
113-00-8
guanidine nitrate

| Conditions | Yield |
|---|---|
| bei der Oxydation; |


| Conditions | Yield |
|---|---|
| Multistep reaction; |

-
-
23911-26-4
diethylenetriaminepentaacetic dianhydride

-
-
306-60-5
1-(4-aminobutyl)guanidine

-
-
616205-57-3
diethylenetriamine-N,N''-bis(4-guanidinobutyl)-N,N',N''-triacetic acid

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 50℃; for 4h; | |
| In dimethyl sulfoxide at 50℃; for 10h; | |
| In dimethyl sulfoxide at 20 - 50℃; for 24h; |

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; acetic acid In methanol; dichloromethane at 25℃; for 12h; |

| Conditions | Yield |
|---|---|
| Stage #1: 1-(4-aminobutyl)guanidine; Cholesteryl chloroformate In DMF (N,N-dimethyl-formamide) at 20℃; for 72h; Stage #2: With acetic acid |

-
-
306-60-5
1-(4-aminobutyl)guanidine

| Conditions | Yield |
|---|---|
| In water; isopropyl alcohol | |
| In water; isopropyl alcohol |
-
-
306-60-5
1-(4-aminobutyl)guanidine

-
-
52355-54-1
2-(n-octyl)cyclopropanecarbonyl chloride

-
-
117414-73-0
1-(2-n-octylcyclopropanecarboxamido)-4-guanidinobutane

| Conditions | Yield |
|---|---|
| In pyridine; methanol; chloroform |
Agmatine Specification
The Guanidine,N-(4-aminobutyl)-, with the CAS registry number 306-60-5 and EINECS registry number 206-187-7, has the systematic name and IUPAC name of 2-(4-aminobutyl)guanidine. And the molecular formula of the chemical is C5H14N4.
The characteristics of Guanidine,N-(4-aminobutyl)- are as followings: (1)ACD/LogP: -1.04; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): -5.14; (4)ACD/LogD (pH 7.4): -5.13; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 4; (10)#H bond donors: 6; (11)#Freely Rotating Bonds: 5; (12)Polar Surface Area: 22.08 Å2; (13)Index of Refraction: 1.55; (14)Molar Refractivity: 34.38 cm3; (15)Molar Volume: 107.8 cm3; (16)Polarizability: 13.63×10-24cm3; (17)Surface Tension: 51.3 dyne/cm; (18)Density: 1.2 g/cm3; (19)Flash Point: 124 °C; (20)Enthalpy of Vaporization: 52.02 kJ/mol; (21)Boiling Point: 281.4 °C at 760 mmHg; (22)Vapour Pressure: 0.00357 mmHg at 25°C.
Uses of Guanidine,N-(4-aminobutyl)-: It can react with mitomycin A to produce C20H29N7O5. This reaction will need menstruum methanol. And the yield is about 92%.
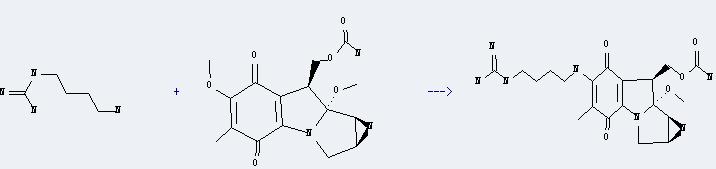
Addtionally, the following datas could be converted into the molecular structure:
(1)SMILES: N(=C(\N)N)\CCCCN
(2)InChI: InChI=1/C5H14N4/c6-3-1-2-4-9-5(7)8/h1-4,6H2,(H4,7,8,9)
(3)InChIKey: QYPPJABKJHAVHS-UHFFFAOYAX
Related Products
- Agmatine
- Agmatine sulfate
- 3066-71-5
- 306-67-2
- 3066-75-9
- 3066-84-0
- 3066-86-2
- 30669-07-9
- 3066-90-8
- 3067-19-4
- 30672-46-9
- 30673-36-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View