-
Name
Calcium chromate(VI)
- EINECS 237-366-8
- CAS No. 13765-19-0
- Density g/cm3
- Solubility
- Melting Point 200(-2H2O)ºC
- Formula Ca. Cr H2 O4
- Boiling Point °Cat760mmHg
- Molecular Weight 156.08
- Flash Point °C
- Transport Information
- Appearance
-
Safety
Suspected human carcinogen with experimental carcinogenic, neoplastigenic, and tumorigenic data. Experimental reproductive effects. Mutation data reported. A powerful oxidizer. Mixture with boron burns violently if ignited. See also CHROMIUM COMPOUNDS and CALCIUM COMPOUNDS.
Analytical Methods:
For occupational chemical analysis use NIOSH: Chromium Hexavalent 7024.
- Risk Codes 22-45-50/53
- Molecular Structure
- Hazard Symbols TLV: 0.001 mg(Cr)/m3; suspected human carcinogen.
- Synonyms Calciumchromate(VI) (CaCrO4) (6CI); Calcium chromate; Calcium chromate (CaCrO4);Calcium chrome yellow; Calcium chromium oxide (CaCrO4); Calcium monochromate;Gelbin; Yellow ultramarine
- PSA 80.26000
- LogP -0.47520
Calcium Chromate Chemical Properties
Chemistry informtion about Calcium Chromate (CAS NO.13765-19-0) is:
IUPAC Name: Calcium Dioxido(Dioxo)Chromium
Synonyms: Calciumchromate(Vi ; Calciumchromate(Vi) ; Calciumchromate(Vi)[Qr] ; Calciumchromate,Anhydrous ; Calciumchromeyellow[Qr] ; Calciumchromeyellow901 ; Calciumchromiumoxide(Cacro4) ; Calciummonochromate
MF: CaCrO4
MW: 156.07
EINECS: 237-366-8
Density: 3.12 g/cm3
Melting point: 2710 °C
Solubility in water: 4.5 g/100 mL (0 °C)
Following is the molecular structure of Calcium Chromate (CAS NO.13765-19-0) is:
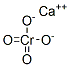
Calcium Chromate Uses
Calcium Chromate (CAS NO.13765-19-0) is used as a pigment, a corrosion inhibitor, and in electroplating, photochemical processing, and industrial waste treatment.
Calcium Chromate Production
Raw materials is Chromic acid
Calcium Chromate Toxicity Data With Reference
Organism Test Type Route Reported Dose (Normalized Dose) Effect Source rat LD50 oral 327mg/kg (327mg/kg) Toxicology and Applied Pharmacology. Vol. 42, Pg. 417, 1977. rat LDLo implant 112mg/kg (112mg/kg) AMA Archives of Industrial Health. Vol. 21, Pg. 530, 1960. Calcium Chromate Consensus Reports
IARC Cancer Review: Group 1 IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 49 , 1990,p. 49.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Human Sufficient Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 23 , 1980,p. 205.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Sufficient Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 23 , 1980,p. 205.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) . Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program. Chromium and its compounds are on the Community Right-To-Know List.
Calcium Chromate Safety Profile
Suspected human carcinogen with experimental carcinogenic, neoplastigenic, and tumorigenic data. Experimental reproductive effects. Mutation data reported. A powerful oxidizer. Mixture with boron burns violently if ignited. See also CHROMIUM COMPOUNDS and CALCIUM COMPOUNDS. Hazard Codes:
 T
T
 N
N
Risk Statements:
R22:Harmful if swallowed.
R45:May cause cancer.
R50/53:Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
Safety Statements:
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S53:Avoid exposure - obtain special instructions before use.
S60:This material and its container must be disposed of as hazardous waste.
S61:Avoid release to the environment. Refer to special instructions / safety data sheets.
RIDADR: 3288
HazardClass: 5.1
PackingGrou: IIICalcium Chromate Standards and Recommendations
OSHA PEL: CL 0.1 mg(CrO3)/m3
ACGIH TLV: TWA 0.001 mg(Cr)/m3; Suspected Human Carcinogen
DFG MAK: DFG TRK: 0.1 mg/m3 calculated as CrO3 in that portion of dust that can possibly be inhaled; 0.2 mg/m3 arc-welding by hand; others 0.1 mg/m3. Animal Carcinogen, Suspected Human Carcinogen
NIOSH REL: (Chromium(VI)) TWA 0.001 mg(Cr(VI))/m3Calcium Chromate Analytical Methods
For occupational chemical analysis use NIOSH: Chromium Hexavalent 7024.
Calcium Chromate Specification
Calcium Chromate (CAS NO.13765-19-0) is a bright yellow solid. It normally occurs as the dihydrate. It is slightly soluble in water. The primary hazard is the threat to the environment. Immediate steps should be taken to prevent its spread to the environment. It insoluble in water. It is a strong oxidizer and reacts with acids and ethanol.Inhalation causes irritation of nose and throat. Ingestion causes severe circulatory collapse and chronic nephritis. Contact with eyes causes irritation. Contact with skin may cause dermatitis and ulcers. Calcium chromate loses water at 200 °C. Calcium chromate reacts with organic matter or reducing agents to form chromium(III). Calcium chromate will react explosively with hydrazine. If mixed with boron and ignited, calcium chromate will burn violently.
Related Products
- Calcium
- Calcium (1)-bis(2-hydroxy-4-methylvalerate)
- Calcium (2S)-2-[(4-chlorobenzoyl)amino]-3-(1H-indol-3-yl)propanoate
- Calcium 2,4-dichlorophenolate
- Calcium 2-ethylhexanoate
- Calcium 2-hydroxypropanoate
- Calcium 2-methoxyethoxide
- Calcium 2-oxoglutarate
- Calcium 3-methyl-2-oxovalerate
- Calcium 4-aminosalicylate
- 13765-24-7
- 13765-25-8
- 13765-26-9
- 137654-20-7
- 137654-21-8
- 13765-44-1
- 137-65-5
- 137658-79-8
- 137661-30-4
- 137661-31-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View