-
Name
D-Tagatose
- EINECS 201-772-3
- CAS No. 87-81-0
- Article Data109
- CAS DataBase
- Density 1.589 g/cm3
- Solubility water: 0.1 g/mL, clear, colorless
- Melting Point 130-136 °C
- Formula C6H12O6
- Boiling Point 551.7 °C at 760 mmHg
- Molecular Weight 180.158
- Flash Point 301.5 °C
- Transport Information
- Appearance White crystalline powder
- Safety 24/25
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols Xi
- Synonyms Tagatose,D- (8CI);D-lyxo-2-Hexulose;
- PSA 118.22000
- LogP -3.37720
Synthetic route

-
-
87-81-0
D-tagatose

| Conditions | Yield |
|---|---|
| With acetic acid at 80℃; for 0.166667h; | 100% |
-
-
5989-81-1
alpha-lactose monohydrate

-
A
-
10257-28-0
D-Galactose

-
B
-
802873-15-0
lactulose

-
C
-
87-81-0
D-tagatose

| Conditions | Yield |
|---|---|
| With triethylammonium borate In water at 70℃; for 4h; Yields of byproduct given; | A n/a B 87% C n/a |
| With triethylammonium borate In water at 70℃; for 4h; Product distribution; various pH, tertiary amines; | A n/a B 87% C n/a |


| Conditions | Yield |
|---|---|
| With sodium aluminate In water at 35℃; for 48h; | 78% |
| With Rhodothermus marinus cellobiose 2-epimerase In aq. buffer at 70℃; pH=6.3; Kinetics; | 28% |
| 27.6% |

| Conditions | Yield |
|---|---|
| With magnesium(II) chloride hexahydrate; pyridoxal 5'-phosphate; serine-glyoxylate L-α-transaminase from Thermosinus carboxydivorans; TK (A0A0I9QGZ2) from the thermophilic bacterium Geobacillus stearothermophilus; thiamine pyrophosphate; 2-oxo-propionic acid; sodium hydroxide In water at 60℃; for 72h; pH=7; Enzymatic reaction; diastereoselective reaction; | 52% |

| Conditions | Yield |
|---|---|
| With water In ethanol at 200℃; under 75007.5 Torr; Supercritical conditions; | A 29% B 26% |

| Conditions | Yield |
|---|---|
| With water In ethanol at 200℃; under 75007.5 Torr; Supercritical conditions; | A 29% B 26% |

| Conditions | Yield |
|---|---|
| durch Acetobacter suboxydans; | |
| mit Hilfe von Acetobacter suboxydans; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 25℃; for 336h; Product distribution; Kinetics; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 25℃; for 336h; Product distribution; Kinetics; |

-
A
-
87-81-0
D-tagatose

-
B
-
140-86-3
1,4-dihydroxy-2-butanone

-
C
-
57538-80-4
D-threo-1-deoxy-[2,5]hexodiulose

| Conditions | Yield |
|---|---|
| With water; dinitrogen monoxide for 0.75h; Mechanism; Product distribution; Ambient temperature; Irradiation; |

-
-
5965-66-2
LACTOSE

-
A
-
87-81-0
D-tagatose

-
B
-
50-99-7
D-glucose

-
C
-
59-23-4
D-Galactose

-
D
-
4618-18-2
lactulose

| Conditions | Yield |
|---|---|
| With Zeolite Na-X; water at 85℃; for 10h; Product distribution; var. reag.: var. minerals and zeolites; var. pH, isomerization; |


-
-
87-81-0
D-tagatose

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| at 25℃; Produkt5:Polysacchariden; |


| Conditions | Yield |
|---|---|
| With sodium aluminate In water at 35℃; for 20h; Product distribution; Further Variations:; reaction times; |

| Conditions | Yield |
|---|---|
| In phosphate buffer pH=7.40; Equilibrium constant; |
-
-
56-82-6
Glyceraldehyde

-
A
-
57-48-7
D-Fructose

-
B
-
87-81-0
D-tagatose

-
C
-
3615-56-3
D-Sorbose

-
D
-
96-26-4
dihydroxyacetone

| Conditions | Yield |
|---|---|
| With zinc(II) bis(L-proline) In water at 20℃; for 168h; Further byproducts given. Title compound not separated from byproducts; |


| Conditions | Yield |
|---|---|
| Stage #1: D-Galactose With aluminum oxide In pyridine for 2h; Heating; Stage #2: With sulfuric acid In acetone for 2h; Further stages.; | A 29 % Chromat. B 8 % Chromat. C 5 % Chromat. |


| Conditions | Yield |
|---|---|
| Stage #1: D-talose With aluminum oxide In pyridine for 0.5h; Heating; Stage #2: With sulfuric acid In acetone for 2h; Further stages.; | A 51 % Chromat. B 13 % Chromat. C 13 % Chromat. |


-
-
87-81-0
D-tagatose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 100 percent / NaBH4 / methanol / 0.25 h / Ambient temperature 2: 100 percent / aq. AcOH / 0.17 h / 80 °C View Scheme |

| Conditions | Yield |
|---|---|
| In water |


| Conditions | Yield |
|---|---|
| With 3-epimerase from Clostridium cellulolyticum H10 pH=8; Kinetics; pH-value; Temperature; Reagent/catalyst; Heating; aq. buffer; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With E. coli ER2566 uridine diphosphogalactose-4-epimerase N179S mutant; uridine diphosphoglucose dehydrogenase; NAD In aq. buffer at 25℃; for 1h; pH=8.5; Catalytic behavior; Reagent/catalyst; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With oxygen; nicotinamide adenine dinucleotide In water at 30℃; for 1h; pH=8; Reagent/catalyst; Time; Green chemistry; Enzymatic reaction; stereoselective reaction; | 91 %Spectr. |
| With nicotinamide adenine dinucleotide; Escherichia coli K12 recombinant galactitol-1-phosphate 5-dehydrogenase; zinc(II) chloride |

-
-
87-81-0
D-tagatose

| Conditions | Yield |
|---|---|
| Stage #1: D-tagatose 1-phosphate With silver nitrate In water Stage #2: With Escherichia coli acid phosphatase; water at 37℃; pH=5.5; Enzymatic reaction; | 495 mg |
-
-
133-99-3, 490-37-9, 2152-98-9, 4482-75-1, 5965-66-2, 13299-27-9, 13360-52-6, 14641-93-1, 15548-43-3, 16462-44-5, 16984-36-4, 16984-38-6, 20869-27-6, 22688-72-8, 27452-49-9, 29276-55-9, 30608-12-9, 34481-13-5, 37169-60-1, 37169-64-5, 64234-01-1, 64234-02-2, 75281-88-8, 80446-85-1, 84413-57-0, 92344-56-4, 93221-87-5, 93301-77-0, 94799-29-8, 100427-98-3, 100428-00-0, 101312-82-7, 102046-24-2, 102046-25-3, 109432-00-0, 109432-02-2, 109432-04-4, 109432-08-8, 135268-49-4, 138231-26-2, 140461-59-2, 145414-22-8, 145414-26-2, 146339-75-5, 146339-76-6, 149116-55-2
Lactose

-
A
-
57-48-7
D-Fructose

-
B
-
802873-15-0
lactulose

-
C
-
3458-28-4
D-Mannose

-
D
-
87-81-0
D-tagatose

-
E
-
50-99-7
D-glucose

-
F
-
2595-98-4
D-talose

-
G
-
59-23-4
D-Galactose

-
H
-
20869-27-6
β-D-galactopyranosyl-(1->4)-D-mannopyranose

| Conditions | Yield |
|---|---|
| With water In ethanol at 200℃; under 75007.5 Torr; Kinetics; Solvent; Supercritical conditions; |

-
-
536-11-8, 645-03-4, 902-54-5, 3031-35-4, 4604-26-6, 5340-95-4, 5995-99-3, 5996-00-9, 13299-20-2, 13299-21-3, 15548-40-0, 16750-26-8, 16967-97-8, 23339-29-9, 24822-33-1, 33428-65-8, 34674-94-7, 35867-21-1, 37169-69-0, 37169-72-5, 38533-27-6, 50271-70-0, 65207-30-9, 82729-73-5, 95463-59-5, 100427-99-4, 100430-37-3, 100483-14-5, 102572-70-3, 110658-40-7, 123809-47-2, 140461-60-5, 143615-15-0
D-melibiose

-
B
-
57-48-7
D-Fructose

-
C
-
536-11-8, 645-03-4, 902-54-5, 3031-35-4, 4604-26-6, 5340-95-4, 5995-99-3, 5996-00-9, 13299-20-2, 13299-21-3, 15548-40-0, 16750-26-8, 16967-97-8, 23339-29-9, 24822-33-1, 33428-65-8, 34674-94-7, 35867-21-1, 37169-69-0, 37169-72-5, 38533-27-6, 50271-70-0, 65207-30-9, 82729-73-5, 95463-59-5, 100427-99-4, 100430-37-3, 100483-14-5, 102572-70-3, 110658-40-7, 123809-47-2, 140461-60-5, 143615-15-0
O-α-D-galactopyranosyl-(1->6)-D-mannopyranose

-
D
-
3458-28-4
D-Mannose

-
E
-
87-81-0
D-tagatose

-
F
-
50-99-7
D-glucose

-
G
-
2595-98-4
D-talose

-
H
-
59-23-4
D-Galactose

| Conditions | Yield |
|---|---|
| With water In ethanol at 200℃; under 75007.5 Torr; Kinetics; Solvent; Supercritical conditions; |

-
-
57-48-7
D-Fructose

-
A
-
849585-22-4
LACTIC ACID

-
B
-
4573-78-8
2-C-(hydroxymethyl)-D-ribose

-
C
-
87-79-6
L-sorbose

-
D
-
87-81-0
D-tagatose

-
E
-
551-68-8
D-psicose

-
F
-
96-26-4
dihydroxyacetone

-
G
-
56-82-6
Glyceraldehyde

| Conditions | Yield |
|---|---|
| With molybdenum(VI) oxide In water at 100℃; for 4h; | A 26.7 %Spectr. B n/a C n/a D n/a E n/a F n/a G n/a |

-
-
1113-60-6
3-hydroxy-2-oxopropionic acid

-
-
95-43-2
D-threose

-
A
-
512-20-9
α-D-tagatopyranose

-
B
-
20197-42-6
tagatose (β-pyranose)

-
C
-
87-81-0
D-tagatose

| Conditions | Yield |
|---|---|
| Stage #1: 3-hydroxy-2-oxopropionic acid With magnesium(II) chloride hexahydrate; pyridoxal 5'-phosphate; transketolase from geobacillus stearothermophilus; thiamine diphosphate In water at 60℃; for 0.333333h; pH=7; Enzymatic reaction; Stage #2: D-threose With L-serin; sodium hydroxide In water at 60℃; for 96h; pH=7; Overall yield = 52 percent; Overall yield = 93 mg; |


| Conditions | Yield |
|---|---|
| With Gluconobacter cerinus X512 In aq. phosphate buffer at 30℃; Reagent/catalyst; Microbiological reaction; |

| Conditions | Yield |
|---|---|
| With (S)-(+)-5,5’-bis[di(3,5-di-tert-butyl-4-methoxyphenyl)phosphino]-4,4’-bi-1,3-benzodioxole; dichloro(benzene)ruthenium(II) dimer; hydrogen In methanol at 100℃; for 17h; Autoclave; diastereoselective reaction; | 88% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; copper(II) sulfate at 20℃; for 18h; Inert atmosphere; | 87% |
| With copper(I) sulfate; sulfuric acid; copper(II) sulfate at 20℃; for 18h; | 82% |
| With sulfuric acid for 2h; Ambient temperature; |

| Conditions | Yield |
|---|---|
| With phosphoric acid; potassium dihydrogen phosphate In water; iso-butanol at 142℃; under 4500.45 Torr; for 0.5h; Reagent/catalyst; Concentration; Inert atmosphere; | 68% |
| With magnesium(II) chloride hexahydrate; 2-carboxyphenylboronic acid In N,N-dimethyl acetamide at 105℃; for 1h; Reagent/catalyst; Time; | 30% |
-
-
773837-37-9
sodium cyanide

-
-
87-81-0
D-tagatose

-
-
67-64-1
acetone

-
-
70147-48-7
(3aS,6R,6aS)-6-((R)-2,2-dimethyl-1,3-dioxolan-4-yl)-3a-(hydroxymethyl)-2,2-dimethyldihydrofuro[3,4-d][1,3]dioxol-4(3aH)-one

| Conditions | Yield |
|---|---|
| Stage #1: sodium cyanide; D-tagatose In water Kiliani reaction; Stage #2: acetone With sulfuric acid | 51% |


| Conditions | Yield |
|---|---|
| With trifluoroethanol; 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol at 60℃; for 16h; stereoselective reaction; | 47% |

-
-
67-56-1
methanol

-
-
87-81-0
D-tagatose

-
A
-
60504-79-2
methyl β-D-tagatofuranoside

-
B
-
60504-77-0
methyl α-D-tagatofuranoside

-
D
-
60504-80-5
methyl α-D-tagatopyranoside

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20℃; for 1h; | A n/a B 47% C n/a D n/a |

-
-
87-81-0
D-tagatose

-
-
143-33-9
sodium cyanide

-
-
67-64-1
acetone

-
B
-
851984-30-0
2,2':5,6-di-O-isopropylidene-2-C-hydroxymethyl-D-galactono-1,4-lactone

-
C
-
864846-17-3
2,3:5,6-di-O-isopropylidene-2-C-hydroxymethyl-D-talono-1,4-lactone

| Conditions | Yield |
|---|---|
| Stage #1: D-tagatose; sodium cyanide With water for 12h; Heating; Stage #2: With Amberlite IR-120 H(1+); water at 20℃; Stage #3: acetone With copper(I) sulfate; sulfuric acid; copper(II) sulfate at 20℃; for 6h; | A 3% B n/a C 44% |


| Conditions | Yield |
|---|---|
| With trifluoroethanol; 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol at 60℃; for 16h; stereoselective reaction; | 31% |


| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; L-proline In methanol at 64℃; for 24h; | A 8% B 17% |


| Conditions | Yield |
|---|---|
| Stage #1: D-tagatose With 2-aminopyridine; acetic acid at 90℃; Lobry de Bruyn-van Ekenstein transformation; Sealed tube; Stage #2: 2-aminopyridine With acetic acid at 90℃; Sealed tube; Stage #3: With trifluoroacetic acid at 75℃; for 1h; | A 16% B 11% |

-
-
87-81-0
D-tagatose

-
-
143-33-9
sodium cyanide

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
67-64-1
acetone

-
A
-
851984-30-0
2,2':5,6-di-O-isopropylidene-2-C-hydroxymethyl-D-galactono-1,4-lactone

-
B
-
864846-22-0
2-C-tert-butyldimethylsilyloxymethyl-2,3:5,6-di-O-isopropylidene-D-talono-1,4-lactone

| Conditions | Yield |
|---|---|
| Multistep reaction; | A 11% B 4.93 g |

-
-
87-81-0
D-tagatose

-
-
161970-48-5
2-C-(hydroxymethyl)-D-xylose

| Conditions | Yield |
|---|---|
| With molybdic acid In water at 80℃; for 15h; Isomerization; | 0.5% |
-
-
110-86-1
pyridine

-
-
87-81-0
D-tagatose

-
-
108-24-7
acetic anhydride

-
-
898815-21-9
[(2R,3S,4S,5R)-2,3,4,5-tetraacetoxytetrahydropyran-2-yl]methyl acetate

-
-
87-81-0
D-tagatose

-
A
-
14307-14-3
2-amino-D-2-deoxy-talose

| Conditions | Yield |
|---|---|
| With methanol; ammonium chloride; ammonia |

| Conditions | Yield |
|---|---|
| With methanol; ammonium chloride; ammonia |
-
-
87-81-0
D-tagatose

-
-
84414-87-9
O1,O2;O3,O4-diisopropylidene-ξ-D-tagatofuranose

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetone | |
| With sulfuric acid; copper(II) sulfate; acetone |

| Conditions | Yield |
|---|---|
| phenyl-d-tagatosazone; |
-
-
87-81-0
D-tagatose

-
-
589-21-9
(4-bromophenyl)hydrazine

-
-
20603-67-2, 94061-61-7
D-lyxo-[2]hexosulose-bis-(4-bromo-phenylhydrazone)

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid 1) DMF, 0 deg C, 2) acetylation; Multistep reaction; |
-
-
87-81-0
D-tagatose

-
-
685-27-8
2,2,2-trifluoro-N-methyl-N-(2,2,2-trifluoroacetyl)acetamide

-
-
622-33-3
N-benzyloxyamine

| Conditions | Yield |
|---|---|
| 1.) NMP, 75 deg C, 30 min, 2.) NMP, RT, 3 h; Multistep reaction; |


| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 25℃; for 336h; Product distribution; Kinetics; |
D-TAGATOSE Chemical Properties
The MW of D-TAGATOSE(87-81-0): 180.16
EINECS: 201-772-3
Melting point: 130-136 °C
refractive index: -5.5 ° (C=1, H2O)
solubility: H2O: 0.1 g/mL, clear, colorless
The Structure of D-TAGATOSE(87-81-0):
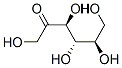
D-TAGATOSE Uses
D-TAGATOSE(87-81-0) is a natural low-calorie bulk sweetener, and it has many physiological functions including anti-hyperglycemia, pre-biotic effects and non-cariogenicity.
D-TAGATOSE Toxicity Data With Reference
D-TAGATOSE Safety Profile
 Xi
Xi Risk Statements: 36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements: 24/25-36-26
24/25: Avoid contact with skin and eyes
36: Wear suitable protective clothing
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
WGK Germany: 3
F: 3-10: Hygroscopic. Keep under argon.
Related Products
- D-TAGATOSE
- 87812-99-5
- 878-17-1
- 87818-06-2
- 87818-31-3
- 878194-88-8
- 878197-87-6
- 87820-16-4
- 878207-28-4
- 878207-82-0
- 878207-92-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View