-
Name
Diethyl azodicarboxylate
- EINECS 217-821-7
- CAS No. 1972-28-7
- Article Data40
- CAS DataBase
- Density 1.2 g/cm3
- Solubility
- Melting Point 6°C
- Formula C6H10N2O4
- Boiling Point 226.3 °C at 760 mmHg
- Molecular Weight 174.156
- Flash Point 106.9 °C
- Transport Information UN 3233
- Appearance Clear orange to orange-red liquid
- Safety 26-36/37-62-47-36
- Risk Codes 5-11-20-36/37/38-48/20-63-65-67-48/20/22-40-20/22-20/21/22
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xn,
Xn, Xi
Xi
- Synonyms Diazenedicarboxylicacid, diethyl ester (9CI);Formic acid, azodi-, diethyl ester (6CI,8CI);1,2-Ethoxycarbonyl diazene;Azodicarboxylic acid diethyl ester;DEAD;Diethoxycarbonyldiazene;Diethyl azodicarboxylate;Diethyl diazenedicarboxylate;Diethyl diazodicarboxylate;NSC 3474;NSC 679015;Unifoam AZ-AE 200;
- PSA 77.32000
- LogP 1.75160
Synthetic route

| Conditions | Yield |
|---|---|
| With triphenylphosphine | 95% |

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene In dichloromethane for 0.25h; Ambient temperature; | 88% |
| With N-Bromosuccinimide In dichloromethane 1.) 0 deg C, 0.5 h, 2.) RT, 1h; | 86.6% |
| Stage #1: diethyl hydrazodicarboxylate With chlorine In water; benzene Stage #2: With sodium carbonate | 81% |
-
-
73240-57-0, 77881-55-1
4,7-Dimethyl-15-oxo-5,6-diphenyl-11,12-diazapentacyclo<8.2.2.14,7.02,9.03,8>pentadeca-5,13-dien-11,12-dicarbonsaeure-diethylester

-
A
-
4114-28-7
diethyl hydrazodicarboxylate

-
B
-
13102-23-3
methyl 3',6'-dimethyl[1,1*:2',1"-terphenyl]-4'-carboxylate

-
C
-
86814-76-8
4,9-Dimethyl-6,7-diphenyl-5,8-dioxa-13,14-diazahexacyclo<10.2.2.02,11.03,10.04,6.07,9>hexadec-15-en-13,14-dicarbonsaeure-diethylester

-
D
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| In diethyl ether; acetone at -25 - -20℃; for 6h; Irradiation; | A n/a B 46% C 7% D n/a |

-
-
73240-57-0, 77881-55-1
4,7-Dimethyl-15-oxo-5,6-diphenyl-11,12-diazapentacyclo<8.2.2.14,7.02,9.03,8>pentadeca-5,13-dien-11,12-dicarbonsaeure-diethylester

-
A
-
4114-28-7
diethyl hydrazodicarboxylate

-
B
-
13102-23-3
methyl 3',6'-dimethyl[1,1*:2',1"-terphenyl]-4'-carboxylate

-
C
-
86814-76-8
4,9-Dimethyl-6,7-diphenyl-5,8-dioxa-13,14-diazahexacyclo<10.2.2.02,11.03,10.04,6.07,9>hexadec-15-en-13,14-dicarbonsaeure-diethylester

-
D
-
1972-28-7
diethylazodicarboxylate

-
E
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| In diethyl ether; acetone at -25 - -20℃; for 6h; Product distribution; Irradiation; | A n/a B 46% C 7% D n/a E n/a |


| Conditions | Yield |
|---|---|
| With nitric acid | |
| With water; nitric acid Reagens 4:Aether; |

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| With bromine; benzene |

| Conditions | Yield |
|---|---|
| With water; chlorine |

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 25℃; for 100h; Rate constant; Irradiation; other solvents; |

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| With iodine |
-
-
17696-93-4
diethyl 3-phenyltriazane-1,2-dicarboxylate

-
-
71-43-2
benzene

-
A
-
108673-54-7
[1,4]benzoquinon-[4-(4-amino-anilino)-phenylimine]-phenylimine

-
B
-
62-53-3
aniline

-
C
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| at 26℃; | |
| at 95℃; Geschwindigkeit; |

-
-
4114-28-7
diethyl hydrazodicarboxylate

-
-
7732-18-5
water

-
-
7782-50-5
chlorine

-
-
71-43-2
benzene

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| Explosionsgefahr !; |


-
-
1972-28-7
diethylazodicarboxylate
-
-
7726-95-6
bromine

-
-
71-43-2
benzene

-
A
-
17759-76-1
ethyl 1-bromoformate

-
B
-
1972-28-7
diethylazodicarboxylate

-
-
24009-91-4, 73285-36-6
(1R,2R,5S,6S)-7,8-Diaza-tricyclo[4.2.2.02,5]deca-3,9-diene-7,8-dicarboxylic acid diethyl ester

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 77 percent / benzene / 48 h / Heating 2: diethyl ether; acetone / 6 h / -25 - -20 °C / Irradiation View Scheme |
-
-
1316208-44-2
C11H18N3O6(1-)*C16H36N(1+)

-
B
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| In chloroform Equilibrium constant; |
-
-
542-92-7
cyclopenta-1,3-diene

-
-
1972-28-7
diethylazodicarboxylate

-
-
14011-60-0
diethyl 2,3-diazanorbornene-2,3-dicarboxylate

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; | 100% |
| In diethyl ether at 0℃; for 12h; | 100% |
| In diethyl ether; toluene at 20℃; for 21.5h; Reflux; | 55% |
| With diethyl ether |
-
-
110-83-8
cyclohexene

-
-
1972-28-7
diethylazodicarboxylate

-
-
17833-25-9
Diethyl 1-(2-cyclohexen-1-yl)-1,2-hydrazinedicarboxylate

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In dichloromethane at -78℃; | 100% |
| With tin(IV) chloride In dichloromethane at -60℃; for 0.0833333h; | 77% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In toluene; acetonitrile at 20℃; for 0.166667h; | 100% |
| With [bis(acetoxy)iodo]benzene; hydrazine hydrate In dichloromethane for 3.5h; Ambient temperature; | 90% |
| With tert-Butyl iodide In acetonitrile for 0.166667h; Reflux; | 90% |
-
-
7114-39-8
[1,3,2]dioxaphospholan-2-yl-dimethyl-amine

-
-
1972-28-7
diethylazodicarboxylate

-
-
75386-16-2
dimethylamino-5 ethoxy-7 carboethoxy-9 trioxa-1,4,6diaza-8,9 phospha(V)-5 spiro(4,4)nonene-7,8

| Conditions | Yield |
|---|---|
| In dichloromethane 10 min, 5 deg C, then room t.,; | 100% |
-
-
14274-42-1
dimethyl-(4,4,5,5-tetramethyl-[1,3,2]dioxaphospholan-2-yl)-amine

-
-
1972-28-7
diethylazodicarboxylate

-
-
75386-17-3
tetramethyl-2,2,3,3-dimethylamino-5-ethoxy-7 carboethoxy-9 trioxa-1,4,6 diaza-8,9 phospha(V)-5 spiro(4,4)noneme-7,8

| Conditions | Yield |
|---|---|
| In dichloromethane Product distribution; 10 min, 5 deg C, then room t; reactions of phosphites and cyclis aminophosphites; | 100% |
| In dichloromethane 10 min, 5 deg C, then room t.,; | 100% |
-
-
18389-60-1
2-dimethylamino-1,3,2-benzodioxaphosphole

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| In dichloromethane 10 min, 5 deg C, then room t., 24 h; | 100% |
-
-
603-35-0
triphenylphosphine

-
-
1972-28-7
diethylazodicarboxylate

-
-
58477-00-2
diethylazodicarboxylate - triphenylphosphine (DEAD-Ph3P)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Mechanism; other phosphine; | 100% |
| In chloroform-d1 | |
| In tetrahydrofuran at 0℃; for 1h; |

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 48h; Ambient temperature; | 100% |
-
-
82100-30-9
1-phenyl-2-(tetrahydropyrimidin-2(1H)-ylidene)ethan-1-one

-
-
1972-28-7
diethylazodicarboxylate

-
-
112030-74-7
C18H24N4O5

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 48h; Ambient temperature; | 100% |
-
-
107165-82-2
2-(p-chlorobenzoylmethylene)imidazolidine

-
-
1972-28-7
diethylazodicarboxylate

-
-
112030-73-6
C17H21ClN4O5

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 48h; Ambient temperature; | 100% |
-
-
107165-83-3
1-(4-chlorophenyl)-2-(tetrahydropyrimidin-2(1H)-ylidene)ethan-1-one

-
-
1972-28-7
diethylazodicarboxylate

-
-
112030-75-8
C18H23ClN4O5

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 48h; Ambient temperature; | 100% |
-
-
107165-84-4
1-(4-methoxyphenyl)-2-(tetrahydropyrimidin-2(1H)-ylidene)ethan-1-one

-
-
1972-28-7
diethylazodicarboxylate

-
-
112030-76-9
C19H26N4O6

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 48h; Ambient temperature; | 100% |

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| In dichloromethane 10 min, 5 deg C, then room t., 24 h; | 100% |
-
-
59875-94-4
1,1-dibenzoylpropane

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| With triphenylphosphine In acetonitrile for 24h; Michael addition; Heating; | 100% |
| triphenylphosphine In acetonitrile at 25℃; for 24h; | 100% |
| tris(triphenylphosphine)ruthenium(II) chloride In acetonitrile for 24h; Product distribution; Further Variations:; Catalysts; Reaction partners; Temperatures; reflux, in dark; | 83% |

| Conditions | Yield |
|---|---|
| With lithium perchlorate; bis(tri-n-butyltin)oxide In diethyl ether at 24.85℃; | 100% |
| With 1,3,4-triphenyl-1,2,4-triazolium hexafluorophosphate; potassium tert-butylate In chloroform at 20℃; for 4h; Inert atmosphere; | 75% |

| Conditions | Yield |
|---|---|
| With lithium perchlorate; bis(tri-n-butyltin)oxide In diethyl ether at 24.85℃; | 100% |
-
-
56826-61-0
(2-methylpyridin-3-yl)methanol

-
-
1972-28-7
diethylazodicarboxylate

-
-
362488-31-1
tert-butyl (3S,4S)-4-[(hydroxyamino)carbonyl]-3-({4-[(2-methyl-3-pyridinyl)methoxy]benzoyl}amino)-1-piperidinecarboxylate trifluoroacetate

| Conditions | Yield |
|---|---|
| With ammonium chloride; triphenylphosphine In tetrahydrofuran | 100% |

-
-
14199-15-6
Methyl 4-hydroxyphenylacetate

-
-
1972-28-7
diethylazodicarboxylate

-
-
22545-14-8
2-(2-ethoxyphenyl)ethanol

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran; diethyl ether | 100% |

-
-
35905-85-2
2-bromonicotinic acid

-
-
1972-28-7
diethylazodicarboxylate

-
-
52718-95-3
2-bromonicotinic acid methyl ester

| Conditions | Yield |
|---|---|
| With triphenylphosphine In methanol; diethyl ether | 100% |
-
-
1670-82-2
Indole-6-carboxylic acid

-
-
1972-28-7
diethylazodicarboxylate

-
-
1011-65-0
5-methoxycarbonylindole

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran; methanol; dichloromethane | 100% |
-
-
123-56-8
Succinimide

-
-
429666-52-4
3-t-butyldiphenylsilyloxy-1-(4-hydroxymethyl-1,3-thiazol-2-yl)azetidine

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran; toluene | 100% |


| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; Diels-Alder reaction; | 100% |
| In dichloromethane at 20℃; Diels-Alder reaction; | 100% |
| In diethyl ether at 0℃; | 100% |
-
-
51127-13-0, 71963-72-9, 87480-74-8, 13302-43-7
2-phenyl-4-methyl-4H-oxazolin-5-one

-
-
1972-28-7
diethylazodicarboxylate

-
-
1228801-83-9
1,2-bis(ethoxycarbonyl)-3-methyl-5-phenyl-2,3-dihydro-1H-1,2,4-triazole-3-carboxylic acid

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 11h; | 100% |
-
-
34713-70-7
2-Phenylpropanal

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| With (S)-4-(tert-butoxy)-2-methyl-4-oxobutanoic acid In tetrahydrofuran at 0℃; for 24h; enantioselective reaction; | 100% |
| With (S)-N-((1R,2R)-2-(3-(3,5-bis(trifluoromethyl)phenyl)thioureido)cyclohexyl)pyrrolidine-2-carboxamide; salicylic acid In dichloromethane at 0℃; for 23h; optical yield given as %ee; enantioselective reaction; | 96% |
| With trifluoroacetic acid; (1R,2R)-N-1',8'-naphthaloyl-1,2-diaminocyclohexane In 1,2-dichloro-ethane at 25℃; for 5h; enantioselective reaction; | 96% |
-
-
137235-13-3
1-(2,4-dichlorophenyl)-3-(dimethylamino)prop-2-en-1-one

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| In toluene; acetonitrile at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With silver(I) acetate; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at -40℃; for 12h; Schlenk technique; Molecular sieve; Inert atmosphere; stereoselective reaction; | 100% |


| Conditions | Yield |
|---|---|
| With silver(I) acetate; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at -40℃; for 12h; Schlenk technique; Molecular sieve; Inert atmosphere; stereoselective reaction; | 100% |


| Conditions | Yield |
|---|---|
| With silver(I) acetate; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at -40℃; for 12h; Schlenk technique; Molecular sieve; Inert atmosphere; stereoselective reaction; | 100% |


| Conditions | Yield |
|---|---|
| With silver(I) acetate; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at -40℃; for 12h; Schlenk technique; Molecular sieve; Inert atmosphere; stereoselective reaction; | 100% |

-
-
106-48-9
4-chloro-phenol

-
-
145881-74-9
2-<2-N-(benzyloxycarbonyl)aminoethoxy>ethanol

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran; hexane; ethyl acetate | 99.8% |

Diethyl azodicarboxylate Specification
The Diethyl azodicarboxylate with cas registry number of 1972-28-7 belongs to the class of aromatic esters. It is also known as Azodicarboxylic acid diethyl ester; DEAD. Its IUPAC name is called [(E)-acetyloxymethyldiazenyl]methyl acetate. It also has an EINECS registry number which is 217-821-7. Its appearance is clear orange to orange-red liquid. It is sensitive to air. This chemical is stable, but may explode if heated under confinement. It secomposes vigorously if heated above 100 C. It is also incompatible with strong acids, strong bases, strong oxidizing agents, strong reducing agents.
Physical properties about Diethyl azodicarboxylate are:
(1)ACD/LogP: 1.85; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.85; (4)ACD/LogD (pH 7.4): 1.85; (5)ACD/BCF (pH 5.5): 14.91; (6)ACD/BCF (pH 7.4): 14.91; (7)ACD/KOC (pH 5.5): 240.75; (8)ACD/KOC (pH 7.4): 240.75; (9)#H bond acceptors: 6; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 6; (12)Index of Refraction: 1.471; (13)Molar Refractivity: 40.41 cm3; (14)Molar Volume: 144.3 cm3; (15)Surface Tension: 38.7 dyne/cm; (16)Density: 1.2 g/cm3; (17)Flash Point: 106.9 °C; (18)Enthalpy of Vaporization: 46.28 kJ/mol; (19)Boiling Point: 226.3 °C at 760 mmHg; (20)Vapour Pressure: 0.0827 mmHg at 25°C.
Preparation of Diethyl azodicarboxylate:
It can be made by hydrazine-N,N'-dicarboxylic acid diethyl ester using reagent iodobenzene diacetate, solvent CH2Cl2 at ambient temperature. The reaction time is 15 min. The yield is about 88%.

Uses of Diethyl azodicarboxylate:
Diethyl azodicarboxylate can react with pyrrole to give 2,5-bis(N,N'-diethoxycarbonylhydrazinyl)pyrrole using solvent diethyl ether. It also needs heating. The reaction time is 24 hours. The yield is 56%.
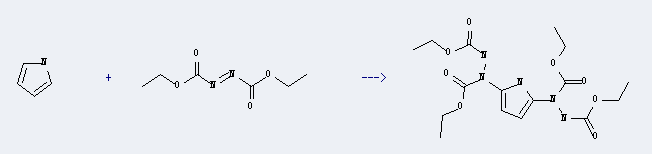
When you are using Diethyl azodicarboxylate, please be cautious about it as the following:
This chemical is highly flammable. It is irritating to eyes, respiratory system and skin and harmful by inhalation, in contact with skin and if swallowed. It may cause lung damage if swallowed. If swallowed, do not induce vomitting; seek medical advice immediately and show this container or label. Wear suitable protective clothing and gloves during using it. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. Keep at temperature of 2-8°C.
You can still convert the following datas into molecular structure:
(1)SMILES:O=C(/N=N\C(=O)OCC)OCC;
(2)Std. InChI:InChI=1S/C6H10N2O4/c1-3-11-5(9)7-8-6(10)12-4-2/h3-4H2,1-2H3/b8-7-;
(3)Std. InChIKey:FAMRKDQNMBBFBR-FPLPWBNLSA-N.
Related Products
- Diethyl (1-phenylpropyl)malonate
- Diethyl (2-(cyclohexylamino)vinyl)phosphonate
- Diethyl (2-(diethoxymethylsilyl)ethyl)phosphonate
- Diethyl (2-(triethoxysilyl)ethyl)phosphonate
- Diethyl (2,4,6-trifluorophenyl)malonate
- Diethyl (2-oxopropyl)phosphonate
- Diethyl (2-thienylmethyl)phosphonate
- Diethyl (4-cyanobenzyl)phosphonate
- Diethyl (4-nitrobenzyl)phosphonate
- Diethyl (beta,gamma-epoxypropyl)phosphonate
- 197234-18-7
- 197239-49-9
- 197244-91-0
- 197252-01-0
- 19727-38-9
- 19727-83-4
- 19728-22-4
- 19728-41-7
- 19728-57-5
- 19728-63-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View