-
Name
Dithizone
- EINECS 200-454-1
- CAS No. 60-10-6
- Article Data21
- CAS DataBase
- Density 1.2 g/cm3
- Solubility Soluble in ethanol, chloroform, dimethylsulfoxide, and pyridine. Insoluble in water.
- Melting Point 168°C (dec.)
- Formula C13H12N4S
- Boiling Point 376.1 °C at 760 mmHg
- Molecular Weight 256.331
- Flash Point 181.2 °C
- Transport Information
- Appearance dark brown or black crystals
- Safety 36-26
- Risk Codes 22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Diazenecarbothioicacid, phenyl-, 2-phenylhydrazide (9CI);Formic acid, (phenylazo)thio-,2-phenylhydrazide (8CI);(Phenylazo)thioformic acid 2-phenylhydrazide;DIZO;Diazenecarbohydrazonothioic acid, N,2-diphenyl-;Diphenylthiocarbazone;Dithizon;Dithizone;NSC 215189;NSC 4275;NSC 97262;NSC 97264;NSC 97353;NSC99124;
- PSA 80.87000
- LogP 4.13580
Dithizone Chemical Properties
Chemical Name: Dithizone
IUPAC NAME: 1-Anilino-3-phenyliminothiourea
CAS No.: 60-10-6
EINECS: 200-454-1
RTECS: LQ9450000
Molecular Formula: C13H12N4S
Molecular Weight: 256.33 g/mol
Melting Point: 168°C (dec.)
Density: 1.2 g/cm3
Flash Point: 181.2 °C
Boiling Point: 376.1 °C at 760 mmHg
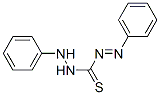
Following is the structure of Diphenylthiocarbazone (60-10-6):
Product Categories about Diphenylthiocarbazone (60-10-6) is Heterocycles
The chemical synonymous of Diphenylthiocarbazone (60-10-6) are Labotest-Bb Lt00452751 ; Diphenylthiocarbazone ; Dithizone ; Dithizone standard solution ; Dithizone, stock solution I ; 1,5-Diphenylthiocarbazone ; Timtec-Bb Sbb008744
Dithizone Uses
Diphenylthiocarbazone (60-10-6) is used as a complex indicator, and for the determination of cobalt, copper, mercury, zinc and silver.
Dithizone Toxicity Data With Reference
| 1. | ipr-mus LD50:200 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD277-689 . | ||
| 2. | ipr-mus LD50:200 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD277-689 . | ||
| 3. | ivn-mus LD50:56 mg/kg | CSLNX* U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. (Aberdeen Proving Ground, MD 21010) NX#07955 . |
Dithizone Consensus Reports
Reported in EPA TSCA Inventory.
Dithizone Safety Profile
Poison by intravenous and intraperitoneal routes. Can cause eye injury and glycosuria. When heated to decomposition it emits highly toxic fumes of NOx and SOx.
Hazard Codes:
Xi: Irritant ![]()
Risk Statements about Diphenylthiocarbazone (60-10-6):
R22 Harmful if swallowed.
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements about Diphenylthiocarbazone (60-10-6):
S26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36 Wear suitable protective clothing.
Dithizone Specification
Diphenylthiocarbazone (60-10-6) is a sulfur-containing organic compound. It is a good ligand, and forms complexes with many metals such as lead and mercury.It may be prepared by reacting phenylhydrazine with carbon disulfide, followed by reaction with potassium hydroxide.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View