-
Name
Ethylene glycol diglycidyl ether
- EINECS 218-746-2
- CAS No. 2224-15-9
- Article Data12
- CAS DataBase
- Density 1.178 g/cm3
- Solubility
- Melting Point
- Formula C8H14O4
- Boiling Point 266.774 °C at 760 mmHg
- Molecular Weight 174.197
- Flash Point 123.633 °C
- Transport Information UN 2810
- Appearance Clear yellow liquid
- Safety 26-36/37/39
- Risk Codes 38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Ethylene glycol diglycidyl ether;1,2-Bis(2,3-epoxypropoxy)ethane;1,2-Bis(glycidyloxy)ethane;2,2'-(1,2-Ethanediylbis(oxymethylene))bisoxirane;Diglycidylethylene glycol;Glycol diglycidyl ether;Oxirane, 2,2'-(1,2-ethanediylbis(oxymethylene))bis-;2,2'-(Ethylenebis(oxymethylene))bisoxirane;Ethylene glycol bis(2,3-epoxypropyl) ether;
- PSA 43.52000
- LogP -0.18280
Synthetic route

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride; water; sodium hydroxide at 40℃; for 0.75h; | 89% |
| With fluoroboric acid; trifluorormethanesulfonic acid; boron trifluoride at 40 - 65℃; for 5h; Temperature; Reagent/catalyst; Sonication; Inert atmosphere; Molecular sieve; Large scale; | 55.7% |
| With potassium hydroxide; benzyltrioctylammonium chloride at 40℃; for 0.75h; | 54% |
-
-
13078-45-0
1,10-Dichloro-4,7-dioxa-2,9-decanediol

-
-
2224-15-9
ethylene glycol diglycidyl ether

| Conditions | Yield |
|---|---|
| With sodium hydroxide Dehydrochlorination; | 40 g |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate at 80℃; for 3h; | 99.3% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
134918-67-5
Ethyl 1H,1H-perfluorooctanoxyacetate

-
-
57-14-7
1,1-dimethylhydrazine

| Conditions | Yield |
|---|---|
| In methanol Heating; | 99% |

-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
26131-32-8
methyl 2,4,4,5,7,7,8,8,9,9,9-undecafluoro-2,5-bis(trifluoromethyl)-3,6-dioxanonanoate

-
-
57-14-7
1,1-dimethylhydrazine

| Conditions | Yield |
|---|---|
| In methanol Heating; | 96% |

-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
133609-46-8
Methyl perfluoro-2,5,8,11-tetramethyl-3,6,9,12-tetraoxapentadecanoate

-
-
57-14-7
1,1-dimethylhydrazine

| Conditions | Yield |
|---|---|
| In methanol Heating; | 96% |


| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide at 110℃; under 7500.75 Torr; for 3h; | 94% |
| With C42H44N8O6Zn(2+)*2Cl(1-) In neat (no solvent) at 100℃; under 750.075 Torr; for 24h; Kinetics; Pressure; Temperature; Autoclave; | 94.5% |
| With C44H20AlCl9N4; bis(triphenylphosphine)iminium chloride In neat (no solvent) at 120℃; under 22502.3 Torr; for 1h; Autoclave; |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
39187-47-8
Methyl perfluoro-2,5,8-trimethyl-3,6,9-trioxadodecanoate

-
-
57-14-7
1,1-dimethylhydrazine

| Conditions | Yield |
|---|---|
| In methanol Heating; | 94% |


| Conditions | Yield |
|---|---|
| With Amberlyst-15 resin at 70℃; for 5h; | 93.2% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
13140-34-6
methyl undecafluoro-2-methyl-3-oxahexanoate

-
-
57-14-7
1,1-dimethylhydrazine

| Conditions | Yield |
|---|---|
| In methanol Heating; | 92% |

-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
57260-71-6
1-t-Butoxycarbonylpiperazine

-
-
158328-41-7
ethylene glycol di<2-hydroxy-3-(N'-tert-butoxycarbonylpiperazinyl)>propyl ether

| Conditions | Yield |
|---|---|
| In chloroform for 24h; Heating; | 91% |

| Conditions | Yield |
|---|---|
| With N-benzyl-trimethylammonium hydroxide at 0℃; for 0.5h; Neat (no solvent); regiospecific reaction; | 90% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
-
1072-71-5
2,5-Dimercapto-1,3,4-thiadiazole

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol diglycidyl ether; 2,5-Dimercapto-1,3,4-thiadiazole In methanol at 60℃; for 18h; Stage #2: With sodium methylate In methanol at 20℃; for 0.5h; Stage #3: (3-chloropropyl)triethoxysilane In methanol at 70℃; for 12h; | 90% |


| Conditions | Yield |
|---|---|
| at 120℃; for 4h; | 82% |

| Conditions | Yield |
|---|---|
| With sodium hydride In water Cycloaddition; Heating; | 82% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
10239-34-6
N,N'-dibenzyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| With sodium hydride In water Cycloaddition; Heating; | 80% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

| Conditions | Yield |
|---|---|
| With sodium azide; ammonium chloride In methanol; water at 80℃; for 24h; | 80% |
| With sodium azide In methanol; water at 80℃; |

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol diglycidyl ether With Tributylphosphine oxide; lithium bromide In toluene for 2h; Heating; Stage #2: phenyl isocyanate In toluene for 12h; Heating; Further stages.; | 78% |

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol diglycidyl ether With lithium bromide In dichloromethane for 2h; Heating; Stage #2: carbon disulfide In dichloromethane for 10h; Heating; Further stages.; | 76% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
23978-55-4
1,4,10,13-tetraoxa-7,16-diazacyclooctadecane

-
A
-
119017-33-3
3,10,23,29-tetrahydroxy-5,8,15,18,25,28,35,38,43,46,51,54-dodecaoxa-1,12,21,32-tetraazatricyclo<30.30.8.812,21>hexapentacontane

-
B
-
117536-17-1
3,10-dihydroxy-5,8,15,18,23,26-hexaoxa-1,12-diazabicyclo<10.8.8>octacosane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethanol for 10h; Heating; | A 1% B 69% |

-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
23978-55-4
1,4,10,13-tetraoxa-7,16-diazacyclooctadecane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethanol Heating; | 69% |

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol diglycidyl ether With lithium bromide In tetrahydrofuran for 0.5h; Inert atmosphere; Reflux; Stage #2: phenyl isocyanate In tetrahydrofuran Inert atmosphere; Reflux; Stage #3: carbon disulfide In tetrahydrofuran at 20℃; for 7h; Inert atmosphere; | 67% |

-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
75-04-7
ethylamine

-
-
92237-85-9
1-Ethylamino-3-[2-(3-ethylamino-2-hydroxy-propoxy)-ethoxy]-propan-2-ol

| Conditions | Yield |
|---|---|
| In water for 50h; Ambient temperature; | 64% |

| Conditions | Yield |
|---|---|
| With ammonia for 50h; Ambient temperature; | 63% |

| Conditions | Yield |
|---|---|
| With lithium bromide In dichloromethane for 5h; Inert atmosphere; Reflux; regioselective reaction; | 58% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
13078-45-0
1,10-Dichloro-4,7-dioxa-2,9-decanediol

| Conditions | Yield |
|---|---|
| With hydrogenchloride | 56% |

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol diglycidyl ether With lithium bromide In tetrahydrofuran for 0.5h; Inert atmosphere; Reflux; Stage #2: phenyl isocyanate In tetrahydrofuran for 6h; Inert atmosphere; Reflux; | 54% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
175854-39-4
1,4,7-tris(tert-butyloxycarbonyl)-1,4,7,10-tetraazacyclododecane

| Conditions | Yield |
|---|---|
| In ethanol for 120h; Heating; | 53.2% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
31249-95-3
Kryptofix 21

-
-
129940-62-1
3,10-dihydroxy-5,8,13,18,23-pentaoxa-1,12-diazabicyclo<10.8.5>pentacosane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethanol for 10h; Heating; | 51% |
-
-
3511-19-1
2,3-dichlorotetrahydrofuran

-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
137675-79-7
1,12-di(3-chloro-2-tetrahydrofuryl)-3,10-dichloro-1,5,8,12-tetraoxadodecane

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In tetrachloromethane at 40℃; for 1h; | 50.7% |
-
-
2224-15-9
ethylene glycol diglycidyl ether

-
-
294-92-8
diaza[12]crown-4

-
A
-
113769-01-0
3,10,20,27-tetrahydroxy-5,8,15,22,25,32,37,42-octaoxa-1,12,18,29-tetraazatricyclo<27.27.5.51,29>tetratetracontane

-
B
-
113769-00-9
meso-3,10-dihydroxy-5,8,15,20-tetraoxa-1,12-diazabicyclo<10.5.5>docosane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethanol for 10h; Heating; | A 14% B 46% |
| In tetrahydrofuran; ethanol for 5h; Heating; | A 37.5% B 22.5% |
| In tetrahydrofuran; ethanol for 10h; Heating; |

Ethylene glycol diglycidyl ether Consensus Reports
Ethylene glycol diglycidyl ether Specification
The Bis(1,2-epoxypropylether)ethanediol, with the CAS registry number 2224-15-9, is also known as Oxirane,2,2'-[1,2-ethanediylbis(oxymethylene)]bis-. It belongs to the product categories of Epoxy Diluents; Oxiranes; Simple 3-Membered Ring Compounds; Epoxide Monomers; Monomers; Polymer Science. Its EINECS number is 218-746-2. This chemical's molecular formula is C8H14O4 and molecular weight is 174.19. What's more, its systematic name is 2,2'-[1,2-Ethanediylbis(oxymethylene)]dioxirane. Its classification codes are: (1)Mutation data; (2)Skin / Eye Irritant; (3)TSCA Flag T [Subject to the Section 4 test rule under TSCA]. This chemical mixed with bisphenol A type epoxy resin can be used as low viscosity composite, impregnation liquid, adhesive and resin modifier.
Physical properties of Bis(1,2-epoxypropylether)ethanediol are: (1)ACD/LogP: -0.304; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.30; (4)ACD/LogD (pH 7.4): -0.30; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 16.29; (8)ACD/KOC (pH 7.4): 16.29; (9)#H bond acceptors: 4; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 7; (12)Polar Surface Area: 43.52 Å2; (13)Index of Refraction: 1.476; (14)Molar Refractivity: 41.752 cm3; (15)Molar Volume: 147.899 cm3; (16)Polarizability: 16.552×10-24cm3; (17)Surface Tension: 45.83 dyne/cm; (18)Density: 1.178 g/cm3; (19)Flash Point: 123.633 °C; (20)Enthalpy of Vaporization: 48.442 kJ/mol; (21)Boiling Point: 266.774 °C at 760 mmHg; (22)Vapour Pressure: 0.014 mmHg at 25°C.
Preparation of Bis(1,2-epoxypropylether)ethanediol: this chemical can be prepared by chloromethyl-oxirane and at the temperature of 40 °C. This reaction will need reagent KOH with the reaction time of 45 min. This reaction will also need catalyst trioctylbenzylammonium chloride. The yield is about 54%.
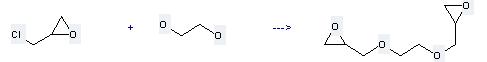
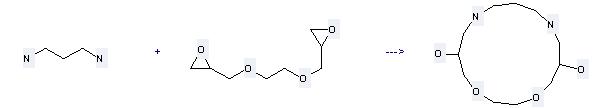
Uses of Bis(1,2-epoxypropylether)ethanediol: it can be used to produce 1,4-dioxa-8,12-diaza-cyclopentadecane-6,14-diol by heating. It will need reagent NaH and solvent H2O. The yield is about 82%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: O(CC1OC1)CCOCC2OC2
(2)Std. InChI: InChI=1S/C8H14O4/c1(9-3-7-5-11-7)2-10-4-8-6-12-8/h7-8H,1-6H2
(3)Std. InChIKey: AOBIOSPNXBMOAT-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 460mg/kg (460mg/kg) | "Prehled Prumyslove Toxikologie; Organicke Latky," Marhold, J., Prague, Czechoslovakia, Avicenum, 1986Vol. -, Pg. 775, 1986. | |
| rat | LD50 | oral | 2500uL/kg (2.5mL/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: ATAXIA GASTROINTESTINAL: CHANGES IN STRUCTURE OR FUNCTION OF SALIVARY GLANDS | National Technical Information Service. Vol. OTS0571709. |
Related Products
- Ethylene
- Ethylene 1-aziridinepropionate
- ETHYLENE ACRYLATE
- Ethylene azelate
- Ethylene bis(chloroformate)
- Ethylene bis(iodoacetate)
- Ethylene brassylate
- Ethylene carbonate
- Ethylene chlorothioarsenate(III)
- Ethylene diisothiouronium dibromide
- 222415-94-3
- 2224-33-1
- 22244-61-7
- 2224-49-9
- 2224-52-4
- 22245-83-6
- 22245-89-2
- 22245-92-7
- 22246-00-0
- 22246-04-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View