-
Name
GLYCERYL TRIBENZOATE
- EINECS
- CAS No. 614-33-5
- Article Data23
- CAS DataBase
- Density 1.243g/cm3
- Solubility
-
Melting Point
68-72 °C(lit.)
- Formula C24H20 O6
- Boiling Point 548.9°C at 760 mmHg
- Molecular Weight 404.419
- Flash Point 238.1°C
- Transport Information
- Appearance
- Safety Mildly toxic by ingestion. When heated to decomposition it emits acrid smoke and irritating fumes. See also ESTERS.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 1,2,3-Propanetriol,tribenzoate (9CI); Glycerol, tribenzoate (6CI,7CI,8CI); Benzoflex S 404;Glyceryl tribenzoate; NSC 2230; Tribenzoin; Uniplex 260
- PSA 78.90000
- LogP 3.92600
Synthetic route

| Conditions | Yield |
|---|---|
| With calcium hydroxide In 5,5-dimethyl-1,3-cyclohexadiene; decane at 220℃; for 1h; Solvent; Reagent/catalyst; Reflux; Large scale; | 99.2% |
| With H2SO4/TiO2-La2O3-solid acid catalyst at 185 - 195℃; for 12h; | 88.6% |
| at 200℃; anschl. auf 270grad; | |
| at 200℃; dann auf 270grad im Druckrohr erhitzen; | |
| With silicate-catalyst at 200 - 220℃; unter Entfernen des entstehenden Wassers; |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 16h; Cooling with ice; | 70% |
-
-
98-88-4
benzoyl chloride

-
-
100-79-8
(R,S)-2,2-dimethyl-1,3-dioxolane-4-methanol

-
-
614-33-5
glyceryl tribenzoate


| Conditions | Yield |
|---|---|
| at 200℃; |

| Conditions | Yield |
|---|---|
| at 250℃; |


| Conditions | Yield |
|---|---|
| at 200℃; | |
| at 200℃; |

| Conditions | Yield |
|---|---|
| at 200℃; bei der Einwirkung auf Kaliumbenzoat; |

| Conditions | Yield |
|---|---|
| im geschlossenen Rohr; |
-
-
3376-49-6
2-benzoyloxypropane-1,3-diol

-
-
98-88-4
benzoyl chloride

-
A
-
614-33-5
glyceryl tribenzoate

-
B
-
76999-62-7
2,3-bis-benzoyloxy-propan-1-ol

-
C
-
40593-13-3
benzoic acid 3-hexanoyloxy-2-hydroxypropyl ester

| Conditions | Yield |
|---|---|
| With (S)-1-methyl-2-((morpholin-1-yl)methyl)pyrrolidine; (methylcyclopentadienyl)tin(II) chloride 1.) CH2Cl2, 0 deg C, 30 min, 2.) CH2Cl2, 5 min, 3.) CH2Cl2, 0 deg C, 1h,; Yield given. Multistep reaction. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| at 180 - 200℃; | |
| at 180 - 200℃; |

| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 48h; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran / 2 h 2: triethylamine / tetrahydrofuran / 48 h View Scheme |

| Conditions | Yield |
|---|---|
| With Merrifield resin-supported N3=P(MeNCH2CH2)3N at 23 - 25℃; for 0.2h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| In hexane for 2h; Inert atmosphere; Schlenk technique; | 75% |

| Conditions | Yield |
|---|---|
| With scandium tris(trifluoromethanesulfonate) at 150℃; for 24h; Sealed tube; | 75% |
-
-
64-17-5
ethanol

-
-
614-33-5
glyceryl tribenzoate

-
-
141-52-6
sodium ethanolate

-
-
93-89-0
benzoic acid ethyl ester


-
-
64-17-5
ethanol

-
-
614-33-5
glyceryl tribenzoate

-
A
-
76999-62-7
2,3-bis-benzoyloxy-propan-1-ol

-
B
-
119-53-9
2-hydroxy-2-phenylacetophenone

-
-
64-17-5
ethanol

-
-
614-33-5
glyceryl tribenzoate

-
A
-
532-32-1
sodium benzoate

-
B
-
93-89-0
benzoic acid ethyl ester

-
C
-
56-81-5
glycerol

-
-
614-33-5
glyceryl tribenzoate

-
-
18838-10-3
N-(2-hydroxyethyl)-benzamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Merrifield resin-supported N3=P(MeNCH2CH2)3N / 0.2 h / 23 - 25 °C / Inert atmosphere 2: Merrifield resin-supported N3=P(MeNCH2CH2)3N / tetrahydrofuran / 23 - 25 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| With tris(2,4-pentanedionato)ruthenium(III); hydrogen; bis(trifluoromethanesulfonyl)amide; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In tetrahydrofuran at 130℃; under 45004.5 Torr; for 18h; Autoclave; | 87 %Chromat. |
GLYCERYL TRIBENZOATE Chemical Properties
Glycerol tribenzoate(614-33-5) has a melting point of 68-72 °C(lit.).
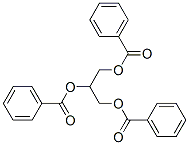
The molecular structure of glycerol tribenzoate(614-33-5):
GLYCERYL TRIBENZOATE Toxicity Data With Reference
| 1. | orl-rat LD50:11,700 mg/kg | NPIRI* Raw Material Data Handbook, Vol.1: Organic Solvents, 1974. 2 (1975),59. |
GLYCERYL TRIBENZOATE Consensus Reports
GLYCERYL TRIBENZOATE Safety Profile
Related Products
- GLYCERYL ACETATE
- Glyceryl behenate
- Glyceryl dioleate
- Glyceryl monocaprate
- Glyceryl monolaurate
- Glyceryl monooleate
- Glyceryl monoricinoleate
- Glyceryl monostearate
- Glyceryl monostearate
- Glyceryl monothioglycolate
- 61436-73-5
- 61436-88-2
- 61437-85-2
- 6143-80-2
- 61438-64-0
- 614-39-1
- 61439-59-6
- 61440-88-8
- 6144-28-1
- 61443-78-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View