Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:615-36-1
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryHangzhou JINLAN Pharm-Drugs Technology Co., Ltd
We can provide GMP validation service that complies with SFDA, FDA, WHO and EU EMPA.Excellent registration team could help us easlily to register our products in different countries.If you and your customer are interested in some products or need C
Cas:615-36-1
Min.Order:1 Gram
Negotiable
Type:Manufacturers
inquiryHangzhou Think Chemical Co. Ltd
2-Bromoaniline CAS No.:615-36-1 Name: 2-Bromoaniline Synonyms: o-Bromoaniline; 2-Bromobenzenamine Molecular Structure Molecular Formula: C6H6BrN Molecula
Cas:615-36-1
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Other
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Appearance:Colorless liquid Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine c
Cas:615-36-1
Min.Order:1 Kilogram
FOB Price: $14.0 / 15.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei yanxi chemical co.,LTD.
Hebei Yanxi Chemical Co., Ltd. is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low car
Cas:615-36-1
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
TIANFUCHEM--615-36-1--2-Bromoaniline in stock Our company was built in 2009 with an ISO certificate.In the past 10 years, we have grown up as a famous fine chemicals supplier in China And we had established stable business relationships
Cas:615-36-1
Min.Order:1 Metric Ton
FOB Price: $2000.0
Type:Lab/Research institutions
inquiryJinan Finer Chemical Co., Ltd
Product Description Name 2-Bromoaniline CAS 615-36-1 Assay 99% Appearance C
LIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc.
Cas:615-36-1
Min.Order:1 Kilogram
FOB Price: $0.9 / 1.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Identification Product name: 2-bromoaniline Product name: 2-bromoaniline Molecular formula: C6H6BrN Molecular weight: 172.03 Structural formula:
Hubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best s
Cas:615-36-1
Min.Order:10 Gram
Negotiable
Type:Other
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:615-36-1
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Afine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:615-36-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:615-36-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Fortuna Chemical Co.,Ltd
CAS:615-36-1 EINECS:172.02 MW:172.02 MF:C6H6BrN Melting point:29 °C Boiling point:229 °C(lit.) Appearance:White crystalline powder Package:25kg/drum Transportation:by sea/air/courier Port:Shanghai
Hangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Nanjing Fred Technology Co.,Ltd.
Taizhou FredChem Co. Ltd. is a high-tech enterprise located in China Medical City specializing in R&D, manufacturing, sales of new APIS, pharmaceutical intermediates and fine chemicals. All of our company is committed to provide high quality ch
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Beijing Greenchem Technology Co.,Ltd. ( Panjin Greenchem Technology Co.Ltd .)
Capability on chemical synthesis1. Beijing High-Tech Enterprises2. Strong R&D Team3. 8 years of experiences in R & D of high-tech Catalyst;4. 5000 production techniques, 69 items of national patents, and 360 kinds of products on sales;5. The producti
Greenutra Resource Inc
2-BromoanilineAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen In tetrahydrofuran; water at 120℃; under 22502.3 Torr; for 4h; Autoclave; | 99% |
| With hydrazine In ethanol at 80℃; for 0.333333h; Catalytic behavior; chemoselective reaction; | 99.6% |
| With hydrogen In water at 120℃; under 15001.5 Torr; for 5h; Green chemistry; chemoselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With 2-((dicyclohexylphosphino)methyl)-1,3-bis(2,6-diisopropylphenyl)-4,5-dimethyl-1H-imidazol-3-ium iodide; ammonia; palladium diacetate; sodium t-butanolate In 1,4-dioxane at 120℃; under 7500.75 Torr; for 24h; Autoclave; Inert atmosphere; | 99% |
| With 2-(di-tert-butylphosphino)-1-mesityl-4,5-diphenyl-1H-imidazole; ammonia; palladium diacetate; sodium t-butanolate In 1,4-dioxane at 120℃; under 7500.75 Torr; for 24h; Autoclave; Inert atmosphere; | > 99 %Chromat. |

| Conditions | Yield |
|---|---|
| Stage #1: o-bromo-N-hydroxybenzamide With acetic anhydride; potassium carbonate In dimethyl sulfoxide at 50℃; for 2h; Lossen Rearrangement; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide at 0℃; | 99% |
| With sodium hydroxide In dimethyl sulfoxide at 80℃; for 1.5h; Lossen rearrangement; | |
| Stage #1: o-bromo-N-hydroxybenzamide With potassium carbonate In dimethyl sulfoxide at 90℃; for 3h; Lossen Rearrangement; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide at 20℃; for 0.0833333h; |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In toluene at 20℃; for 3h; Inert atmosphere; | A n/a B 97% |
| With hydrogenchloride; zinc(II) chloride In benzene Ambient temperature; electrolysis; Yield given. Yields of byproduct given. Title compound not separated from byproducts; | |
| With water; zinc at 250℃; for 0.3h; Reduction; Title compound not separated from byproducts; | |
| With 5%-palladium/activated carbon; hydrazine hydrate In methanol at 80℃; for 0.0833333h; Overall yield = 95 %; | |
| With hydrogen In toluene at 110℃; under 7500.75 Torr; for 0.75h; Autoclave; |
-
-
10113-38-9
N-(2-bromophenyl)formamide

-
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With potassium phosphate; C29H55FeNOP2; hydrogen In tetrahydrofuran at 110℃; under 15001.5 Torr; for 3h; Catalytic behavior; | 97% |
| With sodium hydroxide In ethanol; water at 40℃; Kinetics; |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 90℃; for 1.25h; | 96% |
| With D-glucose; potassium hydroxide In water at 85℃; for 0.166667h; Green chemistry; chemoselective reaction; | 85% |
| With tributylgermanium hydride; thiophenol In toluene for 0.5h; Heating; | 5% |
-
-
15426-16-1
bis-(2-bromo-phenyl)-diazene

-
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With aminomethyl polystyrene resin formic acid salt; zinc In methanol at 20℃; for 0.25h; | 94% |
| With formic acid; zinc In methanol at 20℃; for 0.116667h; | 93% |
| With ammonium formate; nickel In methanol at 20℃; for 0.116667h; | 93% |
-
-
15426-16-1
(E)-1,2-bis(2-bromophenyl)diazene

-
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With nickel; hydrazinium monoformate In methanol for 0.116667h; Heating; | 93% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; potassium bromide In water; acetonitrile | A 87% B 12% |
| With pyridinium hydrobromide perbromide In tetrahydrofuran for 0.25h; Product distribution; other aromatic amines; | A 84% B n/a |
| With N-Bromosuccinimide; silver hexafluoroantimonate; 1-methylthiotriptycene In 1,2-dichloro-ethane at 20℃; for 1h; Inert atmosphere; Schlenk technique; Overall yield = 95 percentSpectr.; | A 76% B n/a |
-
-
244205-40-1
(2-bromophenyl)boronic acid

-
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide; air In methanol at 20℃; for 16h; | 87% |
| With copper(I) oxide; ammonium hydroxide; oxygen; sodium hydroxide In water at 25℃; for 15h; | 78% |
| With sodium hydroxide; hydroxylamine-O-sulfonic acid In acetonitrile at 20℃; for 16h; | 70% |
-
-
132431-13-1
N-(2-bromophenyl)-O-benzyl carbamate

-
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; ethanethiol for 36h; Ambient temperature; | 85% |

| Conditions | Yield |
|---|---|
| With water Inert atmosphere; | A 51% B 73% |

| Conditions | Yield |
|---|---|
| With [bis(2-methylallyl)cycloocta-1,5-diene]ruthenium(II); formic acid; bis(trifluoromethanesulfonyl)amide; triethylamine; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In dibutyl ether at 130℃; for 24h; | A n/a B 73% |
-
-
1164112-16-6
2-(2-bromophenyl)-1,3,2-dioxaborolane

-
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide; air In methanol at 20℃; for 15h; | 71% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetra(n-butyl)ammonium hydroxide; ammonia In water at 45℃; for 48h; Inert atmosphere; Sealed tube; chemoselective reaction; | 71% |
| Stage #1: 1-Bromo-2-iodobenzene With TurboGrignard In tetrahydrofuran at -45℃; for 2h; Inert atmosphere; Stage #2: With C10H17NO In tetrahydrofuran; toluene at -45℃; for 2h; Inert atmosphere; | 29% |

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; trimethylsilylazide In tetrachloromethane at 80℃; for 4h; | 70% |

| Conditions | Yield |
|---|---|
| With bromine; hydrogen fluoride; antomony(V) at -40℃; for 0.25h; | A 29% B 62% C 3% |
| With bromine; hydrogen fluoride; antomony(V) at -40℃; for 0.25h; Mechanism; Product distribution; dependence of product distribution on ammount of catalyst; |

-
-
6674-22-2
1,8-diazabicyclo[5.4.0]undec-7-ene

-
-
25216-70-0
2-bromophenyl carbamic acid methyl ester

-
A
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; water at 80℃; under 760.051 Torr; for 2h; | A 61% B 53% |


| Conditions | Yield |
|---|---|
| A 60% B n/a |
-
-
915920-68-2
2-(2-bromophenoxy)propanamide

-
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 140℃; for 4h; Green chemistry; | 60% |
-
-
64-17-5
ethanol

-
-
577-19-5
2-nitrophenyl bromide

-
A
-
90944-48-2
2-bromo-N,N-diethylaniline

-
B
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With Au NCs/TiO2 for 4h; Kinetics; Reagent/catalyst; Inert atmosphere; UV-irradiation; | A 24.2% B 52.8% |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; copper diacetate; potassium bromide In water at 20℃; for 2h; Concentration; Reagent/catalyst; | 50% |
| With D-glucose; Escherichia coli flavin reductase; Bacillus megaterium glucose dehydrogenase; Lechevalieria aerocolonigenes tryptophan-7-halogenase; flavin adenine dinucleotide; NADH In aq. phosphate buffer; isopropyl alcohol pH=7.4; Enzymatic reaction; | |
| With hydrogen bromide; dimethyl sulfoxide In water; ethyl acetate at 60℃; for 12h; | |
| With N-Bromosuccinimide In dichloromethane |
-
-
82127-40-0
E-Ethyl ester of 4-methyl-4,5-epoxy-2-pentenoic acid

-
-
104669-28-5
(2-Bromo-phenyl)-[1-phenyl-meth-(E)-ylidene]-amine

-
C
-
100-52-7
benzaldehyde

-
D
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| scandium tris(trifluoromethanesulfonate) In tetrahydrofuran at 0℃; Title compound not separated from byproducts; | A n/a B 48% C n/a D n/a |


| Conditions | Yield |
|---|---|
| With O-(4-nitrobenzoyl)hydroxylammonium trifluoromethanesulfonate; iron(II) bromide; silver(I) triflimide In 2,2,2-trifluoroethanol; water at 30℃; for 2h; | A 40.2% B 26.8% |
-
-
6165-68-0
thiophene boronic acid

-
-
3302-39-4
1-azido-2-bromobenzene

-
A
-
545442-25-9
2-(2-azidephenyl)thiophene

-
B
-
62532-99-4
2-(thiophen-2-yl)aniline

-
C
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; tetrakis(triphenylphosphine) palladium(0) In 1,2-dimethoxyethane at 100℃; for 6h; Suzuki-Miyaura cross-coupling reaction; | A 40% B 3% C 2% |

-
-
108-86-1
bromobenzene

-
A
-
591-19-5
3-bromoaniline

-
B
-
106-40-1
4-bromo-aniline

-
C
-
615-36-1
2-bromoaniline

| Conditions | Yield |
|---|---|
| With tris-(2-chloro-ethyl)-amine; trifluorormethanesulfonic acid; trifluoroacetic acid In chloroform at 60℃; for 3h; | A 10% B 25% C 30% |
| With hydroxylamine-O-sulfonic acid; iron(II) sulfate In water; acetic acid at 40℃; for 2h; Yield given. Yields of byproduct given; | |
| With hydroxylamine-O-sulfonic acid; iron(II) sulfate In water; acetic acid at 40℃; for 2h; Product distribution; Mechanism; k(C6H5Br)/k(C6H6); |


| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline In acetonitrile at 90℃; for 1h; Temperature; Inert atmosphere; Sealed tube; | 100% |
| In tetrahydrofuran at 20℃; for 24h; | 99% |
| With triethylamine In dichloromethane at 0 - 20℃; for 2h; | 99% |
-
-
103-72-0
phenyl isothiocyanate

-
-
615-36-1
2-bromoaniline

-
-
25688-29-3
1-(2-bromophenyl)-3-phenylthiourea

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 100% |
| In N,N-dimethyl-formamide Heating; | 99% |
| With C64H52CaN6 In neat (no solvent) at 60℃; for 12h; Schlenk technique; Glovebox; Inert atmosphere; | 85% |
-
-
615-36-1
2-bromoaniline

-
-
1548-13-6
p-trifluoromethyl-phenylisocyanate

-
-
23742-77-0
1-(2-bromophenyl)-3-(4-(trifluoromethyl)phenyl)urea

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 3h; Inert atmosphere; | 100% |
| In acetonitrile |
-
-
3282-30-2
pivaloyl chloride

-
-
615-36-1
2-bromoaniline

-
-
65854-92-4
N-(2-bromophenyl)-2,2-dimethylpropanamide

| Conditions | Yield |
|---|---|
| In ethyl acetate at 85℃; | 100% |
| With triethylamine In dichloromethane at 0 - 20℃; for 16h; | 100% |
| With triethylamine In dichloromethane at 0 - 20℃; for 16h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoaniline With n-butyllithium In tetrahydrofuran; hexane at -40℃; Inert atmosphere; Stage #2: benzyl bromide In tetrahydrofuran; hexane at -60 - 20℃; Inert atmosphere; | 100% |
| Stage #1: 2-bromoaniline With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: benzyl bromide In tetrahydrofuran; hexane at -78 - 20℃; Further stages.; | 95% |
| Stage #1: 2-bromoaniline With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.25h; Inert atmosphere; Schlenk technique; Stage #2: benzyl bromide In tetrahydrofuran; hexane at 20℃; for 10h; Inert atmosphere; Schlenk technique; | 90% |

| Conditions | Yield |
|---|---|
| for 0.5h; Heating; | 100% |
| at 70℃; for 20h; | 100% |
| With dmap In chloroform for 0.5h; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| In diethyl ether Ring cleavage; Substitution; | 100% |
| In diethyl ether at 20℃; |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
29976-53-2
N-ethoxycarbonyl-4-piperidone

-
-
615-36-1
2-bromoaniline

-
-
240486-10-6
ethyl 4-(2-bromophenylamino)-4-cyano-1-piperidinecarboxylate

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilyl cyanide; N-ethoxycarbonyl-4-piperidone; 2-bromoaniline With acetic acid for 40h; Stage #2: With ammonia In water | 100% |
| In acetic acid at 20℃; for 112h; Condensation; modified Strecker reaction; | 94% |

-
-
40400-15-5
2-(2-iodophenyl)acetonitrile

-
-
615-36-1
2-bromoaniline

-
-
325141-76-2
2-{2'-amino-[1,1'-biphenyl]-2-yl}acetonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoaniline With Pinacolborane; triethylamine; CyJohnPhos; palladium diacetate In 1,4-dioxane at 80℃; Stage #2: 2-(2-iodophenyl)acetonitrile With barium dihydroxide In 1,4-dioxane at 100℃; Suzuki-Miyaura coupling; Further stages.; | 100% |
| Stage #1: 2-bromoaniline With palladium diacetate; triethylamine; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane; CyJohnPhos In 1,4-dioxane at 25 - 80℃; Inert atmosphere; Stage #2: 2-(2-iodophenyl)acetonitrile With barium hydroxide octahydrate In 1,4-dioxane; water at 100℃; for 1h; Inert atmosphere; | 99% |
| Stage #1: 2-bromoaniline With palladium diacetate; triethylamine; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane; CyJohnPhos In 1,4-dioxane at 25 - 80℃; for 1h; Inert atmosphere; Stage #2: 2-(2-iodophenyl)acetonitrile With barium hydroxide octahydrate In 1,4-dioxane at 100℃; for 1h; | 99% |
| Stage #1: 2-bromoaniline With palladium diacetate; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane; CyJohnPhos In 1,4-dioxane at 80℃; for 1h; Stage #2: 2-(2-iodophenyl)acetonitrile With barium dihydroxide In 1,4-dioxane; water at 100℃; for 1h; Suzuki coupling; Further stages.; | 79% |
| Stage #1: 2-bromoaniline With palladium diacetate; triethylamine; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane; CyJohnPhos In 1,4-dioxane at 80℃; Stage #2: 2-(2-iodophenyl)acetonitrile With barium hydroxide octahydrate In 1,4-dioxane; water at 100℃; Suzuki Coupling; | 77% |
-
-
615-36-1
2-bromoaniline

-
-
13831-31-7
Acetoxyacetyl chloride

-
-
901335-15-7
acetic acid (2-bromo-phenylcarbamoyl)-methyl ester

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane for 2h; | 100% |
| With pyridine In dichloromethane at 20℃; for 2h; |

| Conditions | Yield |
|---|---|
| With caesium carbonate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene; palladium diacetate In 1,4-dioxane at 110℃; for 16h; | 100% |
| With potassium hydroxide In neat (no solvent) at 90℃; for 7.5h; | 60% |
| With tris(dibenzylideneacetone)dipalladium (0); sodium t-butanolate In 1,4-dioxane at 45℃; |
-
-
32161-06-1
1-acetyl-4-piperidinone

-
-
615-36-1
2-bromoaniline

-
-
919118-89-1
1-(4-(2-bromophenylamino)piperidin-1-yl)ethanone

| Conditions | Yield |
|---|---|
| Stage #1: 1-acetyl-4-piperidinone; 2-bromoaniline With sodium tris(acetoxy)borohydride; acetic acid In toluene at 120℃; for 0.25h; Microwave irradiation; Stage #2: With water; sodium hydrogencarbonate In toluene | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-fluoroboronic acid; 2-bromoaniline With potassium carbonate In 1,4-dioxane; water for 0.333333h; Inert atmosphere; Stage #2: With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2 In 1,4-dioxane; water at 100℃; | 100% |
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium carbonate In 1,4-dioxane at 100℃; for 3h; Suzuki Coupling; | 94% |
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium carbonate In 1,4-dioxane at 100℃; for 3h; | 94% |
-
-
615-36-1
2-bromoaniline

-
-
19519-75-6
1-bromo-2-nitrosobenzene

| Conditions | Yield |
|---|---|
| With potassium peroxomonosulphate In dichloromethane; water at 20℃; Inert atmosphere; | 100% |
| With Oxone In dichloromethane; water at 20℃; Inert atmosphere; | 99% |
| With Oxone In dichloromethane; water at 20℃; for 20h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; Inert atmosphere; | 100% |
| With pyridine In dichloromethane at 0 - 20℃; | 98% |
| With pyridine In dichloromethane at 0 - 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene for 24h; Reflux; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene at 100℃; for 24h; Suzuki Coupling; Inert atmosphere; | 85% |

| Conditions | Yield |
|---|---|
| With C11H8Cl4N2O3Pd; tetrabutylammomium bromide; potassium carbonate In methanol; water; N,N-dimethyl-formamide at 100℃; for 0.166667h; Suzuki Coupling; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; tetrabutylammomium bromide; potassium carbonate In chloroform; water at 80 - 100℃; for 0.166667h; Suzuki Coupling; | 99% |
| With 2-[5-(4-methylphenyl)isoxazol-3-yl]-5-(5-phenylisoxazol-3-yl)-1,3,4-oxadiazole·2PdCl2; tetrabutylammomium bromide; potassium carbonate In water; N,N-dimethyl-formamide at 100℃; for 0.0166667h; Suzuki Coupling; | 98% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 120℃; for 48h; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 120℃; for 48h; Inert atmosphere; | 100% |
| Stage #1: 2-bromoaniline With lithium diisopropyl amide In tetrahydrofuran at 0℃; for 0.25h; Stage #2: benzyl bromide In tetrahydrofuran for 0.25h; Further stages; | 83% |
| With N-ethyl-N,N-diisopropylamine In toluene for 24h; Reflux; | |
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 120℃; for 24h; | 82 %Spectr. |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
615-36-1
2-bromoaniline

-
-
1592-00-3
2-bromophenyl isocyanate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 100% |
| Stage #1: bis(trichloromethyl) carbonate; 2-bromoaniline In dichloromethane at 20℃; for 0.5h; Stage #2: With triethylamine In dichloromethane at -35 - 20℃; for 2h; | 81% |
| With triethylamine In toluene for 7h; Reflux; Cooling with ice; | 65.6% |
-
-
1711-09-7
3-bromobenzoyl chloride

-
-
615-36-1
2-bromoaniline

-
-
99586-31-9
2‐(3‐bromophenyl)benzo[d]oxazole

| Conditions | Yield |
|---|---|
| With caesium carbonate; copper(l) iodide; 1,10-Phenanthroline In 1,4-dioxane at 120℃; for 24h; Product distribution / selectivity; Inert atmosphere; | 100% |
| With caesium carbonate; copper(l) iodide; 1,10-Phenanthroline In 1,4-dioxane at 120℃; for 8h; | 76% |

| Conditions | Yield |
|---|---|
| With pyridine; copper(I) bromide dimethylsulfide complex; sodium carbonate In acetonitrile at 75℃; for 48h; Inert atmosphere; solid phase reaction; | 100% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 36h; Dean-Stark; Reflux; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 20℃; for 19h; | 100% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; triethylamine; tris-(o-tolyl)phosphine at 125℃; | 99% |
| With palladium diacetate; triethylamine; tris-(o-tolyl)phosphine at 125℃; | 88% |
| With palladium diacetate; triethylamine; tris-(o-tolyl)phosphine at 125℃; | 88% |
-
-
5271-67-0
2-Thiophenecarbonyl chloride

-
-
615-36-1
2-bromoaniline

-
-
136340-96-0
thiophene-2-carboxylic acid (2-bromophenyl)amide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 99% |
| In tetrahydrofuran at 20℃; for 24h; | 96% |
| With triethylamine In dichloromethane at 0 - 20℃; | 84% |
| In toluene | |
| With triethylamine In dichloromethane at 20℃; for 3h; |
-
-
102-92-1
Cinnamoyl chloride

-
-
615-36-1
2-bromoaniline

-
-
71910-51-5
E-N-(2-bromophenyl)-3-phenylpropenamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 99% |
| With potassium carbonate In water; acetone at 0℃; for 2h; | 97% |
| With potassium carbonate In water; acetone at 0℃; for 2h; | 92% |
-
-
615-36-1
2-bromoaniline

-
-
407-25-0
trifluoroacetic anhydride

-
-
2727-71-1
N-(2-bromophenyl)-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| With sodium carbonate In diethyl ether for 24h; Ambient temperature; | 99% |
| With triethylamine In dichloromethane at 0 - 20℃; | 89% |
| With triethylamine In dichloromethane at 0 - 20℃; | 89% |
-
-
10400-19-8
3-pyridinecarbonyl chloride

-
-
615-36-1
2-bromoaniline

-
-
75075-29-5
N-(2-bromophenyl)-3-pyridinecarboxamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 99% |
| With pyridine at 0 - 20℃; Acylation; | |
| With N,N-diethyl-N-isopropylamine In acetonitrile at 60℃; for 24h; Inert atmosphere; Schlenk technique; |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene Heating; | 99% |
| at 120℃; | 70% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



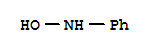




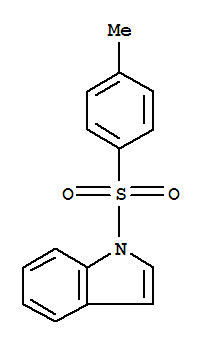







 T,
T,  Xn,
Xn,