-
Name
IMP
- EINECS 205-045-1
- CAS No. 131-99-7
- Article Data54
- CAS DataBase
- Density 2.319 g/cm3
- Solubility
- Melting Point
- Formula C10H13N4O8P
- Boiling Point 851.369 °C at 760 mmHg
- Molecular Weight 348.209
- Flash Point 468.691 °C
- Transport Information
- Appearance
- Safety 22-24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 5'-IMP;IMP;Inosine 5'-(dihydrogen phosphate);Inosine 5'-monophosphate;Inosine5'-phosphate;Inosine-5'-monophosphoric acid;Inosinic acid;
- PSA 189.83000
- LogP -2.15190
Synthetic route
-
-
78681-84-2
Phosphoric acid (3aR,4R,6R,6aR)-2,2-dimethyl-6-(6-oxo-1,6-dihydro-purin-9-yl)-tetrahydro-furo[3,4-d][1,3]dioxol-4-ylmethyl ester bis-(2,2,2-tribromo-ethyl) ester

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With Nafion 125; lithium perchlorate In N,N-dimethyl-formamide; acetonitrile electrolysis; | 89% |
-
-
61-19-8
5'-adenosine monophosphate

-
A
-
131-99-7
5'-inosine monophosphate

-
B
-
58-64-0
adenosine 5'-diphosphate

-
C
-
56-65-5
ATP

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In methanol at 25℃; Product distribution; biosynthesis; various typ-, and conc. of reagent; | |
| In methanol at 25℃; biosynthesis by Candida boidnii No. 2201; pH = 6.5; | A 14.8 mmol B 36.8 mmol C 24.0 mmol |
| With zinc(II) chloride In methanol at 25℃; biosynthesis by Candida boidnii No. 2201; pH = 6.5; | A 2.0 mmol B n/a C 36.8 mmol |
| In methanol at 25℃; biosynthesis by Candida boidnii No. 2201; pH = 6.5; | A 14.8 mmol B 1.1 mmol C 24.0 mmol |


| Conditions | Yield |
|---|---|
| With triethyl phosphate; trichlorophosphate In water at 0℃; for 2h; Rate constant; Product distribution; var. reaction conditions; | 91 % Chromat. |
| With potassium dihydrogenphosphate; D-glucose; guanosine-inosine kinase from Exiguobacterium acetylicum In various solvent(s) at 32℃; pH=7.2; Enzymatic reaction; | |
| With DL-dithiothreitol; recombinant cytosolic 5'-nucleotidase II; ATP In various solvent(s) at 37℃; pH=7.4; Enzyme kinetics; Further Variations:; Reagents; | |
| With sodium pyrophosphate; sodium acetate buffer; Escherichia blattae wild type phosphotransferase at 30℃; pH=5.0; Enzyme kinetics; Further Variations:; Reagents; | |
| With trimethyl phosphite; trichlorophosphate at 0℃; |
-
-
97-55-2, 7540-64-9, 99945-37-6, 130384-52-0
5-phosphoribosyl-1-pyrophosphate

-
-
68-94-0
hypoxanthine

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With Toxoplasma gondii phosphoribosyltransferase In alkaline aq. solution at 37℃; pH=7.4; Enzyme kinetics; |

| Conditions | Yield |
|---|---|
| With disodium pyrophoshate; Shigella flexneri acid phosphatase In various solvent(s) at 30℃; pH=5.0; Enzyme kinetics; Further Variations:; Reagents; |

| Conditions | Yield |
|---|---|
| With hypoxanthine phosphoribosyltransferase Enzyme kinetics; Further Variations:; Reagents; |

| Conditions | Yield |
|---|---|
| With diothiothreitol; water at 37℃; pH=6.8; Enzyme kinetics; |

- 5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-CATGTACCGATAAGCGTATTATGGAAGGAGGCGTTATGAGGGGTCCA-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/bis(adenosine 5'-monophosphate) complex
-
5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-CATGTACCGATAAGCGTATTATGGAAGGAGGCGTTATGAGGGGTCCA-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/bis(adenosine 5'-monophosphate) complex

-
A
5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-CATGTACCGATAAGCGTATTATGGAAGGAGGCGTTATGAGGGGTCCA-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/5'-CTCCTTCCATACGGTGCACCTTCCTC-3' -
5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-CATGTACCGATAAGCGTATTATGGAAGGAGGCGTTATGAGGGGTCCA-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/5'-CTCCTTCCATACGGTGCACCTTCCTC-3'

-
B
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid sodium salt; sodium chloride; adenosine deaminase In water at 25 - 70℃; for 1.33333h; pH=7.4; Enzymatic reaction; |

- 5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-AGGTGCACGTGGTCCAGTGCTGCAGTG-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/adenosine 5'-monophosphate complex
-
5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-AGGTGCACGTGGTCCAGTGCTGCAGTG-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/adenosine 5'-monophosphate complex

-
A
5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-AGGTGCACGTGGTCCAGTGCTGCAGTG-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/5'-CTCCTTCCATACGGTGCACCTTCCTC-3' -
5'-Cy5-ATACGCTTATCGGTACATGAGCACTGCAGCACTGGACCAC-Iowa black RQ-3'/5'-AGGTGCACGTGGTCCAGTGCTGCAGTG-3'/5'-ACCTGGGGGAGTATTGCGGAGGAAGGTGCGTGGTCCAGTGCTGCAGTG-3'/5'-CTCCTTCCATACGGTGCACCTTCCTC-3'

-
B
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With adenosine deaminase aq. buffer; Enzymatic reaction; |

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| Stage #1: C24H24N6O9PPol With ammonium hydroxide In water at 55℃; for 15h; solid phase reaction; Stage #2: With trifluoroacetic acid In dichloromethane at 20℃; for 0.333333h; solid phase reaction; |

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; for 0.333333h; solid phase reaction; |

| Conditions | Yield |
|---|---|
| With Leishmania donovani adenine phosphoribosyltransferase; sodium fluoride; magnesium chloride for 1h; pH=7.4; Kinetics; aq. buffer; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With Dr0824 from Deinococcus radiodurans R1; water at 30℃; for 16h; pH=7.5; Reagent/catalyst; Enzymatic reaction; |
-
-
68-94-0
1,7-dihydro-6H-purin-6-one

-
-
97-55-2, 7540-64-9, 99945-37-6, 130384-52-0
5-phosphoribosyl-1-pyrophosphate

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With mycobacterium tuberculosis hypoxanthine–guanine phosphoribosyltransferase In aq. buffer at 25℃; Kinetics; Enzymatic reaction; | |
| With recombinant Thermus thermophilus HB8 xanthine phosphoribosyltransferase; magnesium chloride In aq. buffer at 70℃; for 0.166667h; pH=6; Catalytic behavior; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With recombinant Thermus thermophilus HB27 hypoxanthine phosphoribosyltransferase; magnesium chloride In aq. buffer at 70℃; for 0.0333333h; pH=8; Catalytic behavior; Kinetics; Concentration; Reagent/catalyst; Temperature; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole Condensation; | 74% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; for 4h; pH=8; | 71% |
-
-
131-99-7
5'-inosine monophosphate

-
-
120138-93-4
C10H14N4O10P2

| Conditions | Yield |
|---|---|
| With sodium hypophosphate at 60℃; pH 6; | 66% |

| Conditions | Yield |
|---|---|
| With 2,2'-dipyridyldisulphide; triphenylphosphine; sodium iodide In dimethyl sulfoxide for 0.666667h; Ambient temperature; |

| Conditions | Yield |
|---|---|
| With 2,2'-dipyridyldisulphide; triphenylphosphine |

| Conditions | Yield |
|---|---|
| With 2,2'-dipyridyldisulphide; triphenylphosphine; sodium iodide In dimethyl sulfoxide for 0.666667h; Ambient temperature; |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide Ambient temperature; |

| Conditions | Yield |
|---|---|
| With sodium acetate buffer; Escherichia blattae wild type 5'-nucleotidase at 30℃; pH=4.0; Enzyme kinetics; Further Variations:; Reagents; | |
| With disodium pyrophoshate; Shigella flexneri acid phosphatase In various solvent(s) pH=6.0; Enzyme kinetics; | |
| With Acinetobacter baumannii recombinant acid phosphatase; nickel dichloride; bovine serum albumin In water pH=6; Cooling with ice; Enzymatic reaction; |
-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With ethylenediaminetetraacetic acid; recombinant TgIMPDHFLAG from Toxoplasma gondii; NAD In various solvent(s) at 37℃; for 1h; pH=8.0; Enzyme kinetics; | |
| With Escherichia coli B3 inosine monophosphate dehydrogenase; nicotinamide adenine dinucleotide Enzyme kinetics; | |
| With mouse inosine monophosphate dehydrogenase type II; NAD; water Kinetics; Enzymatic reaction; | |
| With ethylenediaminetetraacetic acid; human recombinant inosine monophosphate dehydrogenase type II; potassium chloride; nicotinamide adenine dinucleotide; 2-hydroxyethanethiol; diothiothreitol at 30℃; for 0.5h; pH=8; Kinetics; aq. buffer; Enzymatic reaction; | |
| With recombinant N-terminal (His)6-tagged Mycobacterium tuberculosis H37Rv NAD-dependent inosine monophosphate dehydrogenase; NAD; water at 25℃; for 0.0833333h; pH=8; Kinetics; Concentration; Reagent/catalyst; aq. buffer; Enzymatic reaction; |
-
-
131-99-7
5'-inosine monophosphate

-
-
95314-07-1
MopI2'p5'A

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) Ph3P, 2,2-dipyridyl disulfide, 2.) 0.1 M aq. NaI / dimethylsulfoxide / 0.67 h / Ambient temperature 2: aq. Pb(NO3)2, imidazole nitrate buffer / 24 h / 4 °C View Scheme |
-
-
131-99-7
5'-inosine monophosphate

-
-
95314-06-0
MopA2'p5'I

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) Ph3P, 2,2-dipyridyl disulfide, 2.) 0.1 M aq. NaI / dimethylsulfoxide / 0.67 h / Ambient temperature 2: aq. Pb(NO3)2, imidazole nitrate buffer / 24 h / 4 °C View Scheme |
-
-
15020-99-2
cis-dichloro(ethylenediamine)palladium(II)

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| In water Kinetics; byproducts: H2O; 25.1°C, I=0.10 (NaClO4), pH=4.5, varying conc. of Cl(1-) and inosine-5'-phosphate; not isolated, detd. by UV/VIS spectroscopy; |

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| In water Kinetics; byproducts: H2O; pH 4.5, 0.1 M NaClO4, Ru concn. 5E-4 M, nucleotide concn. 0.005-0.025 M;15, 25, or 35°C; |

-
-
16887-00-6
chloride

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| In water Kinetics; 0.01-0.2 M Cl(1-), 20-40°C, 0.1-125 MPa; not isolated, reaction followed by stopped-flow UV/VIS spectroscopy; |

-
-
14267-08-4
N,N,N',N'-tetramethylethylenediamine palladium(II) chloride

-
-
131-99-7
5'-inosine monophosphate


| Conditions | Yield |
|---|---|
| In water Kinetics; byproducts: H2O; 25.1°C, I=0.10 (NaClO4), pH=4.5, varying conc. of Cl(1-) and inosine-5'-phosphate; not isolated, detd. by UV/VIS spectroscopy; ratio of products depending on concn. of ligand, Cl(1-) and reaction time; |

-
-
54831-91-3
(1,10-phenanthroline)(ethylenediamine)platinum(II)

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| 25°C, pH 7-8, I=0.1 or 0.2 M NaCl; calorimetric titrn.; |
-
-
166896-59-9
N,N,N',N'-tetraethylethylenediamine palladium(II) chloride

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| In water Kinetics; reaction of Pd(Et4en)Cl2 with inosine 5'-monophosphate in aq. soln. (pH 4.5) at room temp.; monitored by UV-spectroscopy; |
-
-
137793-30-7
(5,6-dimethyl-1,10-phenanthroline)(ethylenediamine)platinum(II)

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| 70°C, pH 8; (13)C-NMR; |
-
-
118170-06-2
Ru{N'-(2-hydroxyethyl)ethylenediamine-N,N,N'-triacetate}(H2O)

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| In water Kinetics; byproducts: H2O; pH 4.5, 0.1 M NaClO4, Ru concn. 5E-4 M, nucleotide concn. 0.005-0.025 M;15, 25, or 35°C; |

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| not isolated, detected by NMR; |

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| With nitric acid In water nucleotide was dissolved in H2O, HNO3 was added to pH=3, Cr(NO3)3 was dissolved in H2O, soln. were mixed and heated to 50°C for 1-2 h; cooled or EtOH was added, filtered, washed with H2O and EtOH, dried in vac. over P2O5; elem. anal.; |

-
-
131-99-7
5'-inosine monophosphate

| Conditions | Yield |
|---|---|
| In water-d2 NMR monitoring; not isolated, detected by NMR; |
INOSINIC ACID Chemical Properties
Product Name: Inosinic acid (131-99-7)
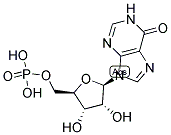
Molecular Formula: C10H13N4O8P
Molecular Weight: 348.206g/mol
Mol File: 131-99-7.mol
Einecs:205-045-1
Storage Temperature: -20°C
Ynonyms of Ethyl Inosinic acid viki (131-99-7) : 5’-inosinicacid ; inosine5’-phosphate ; inosinemonophosphate ; imp ; inosinic acid ; inosine 5'-monophosphate ; inosine-5'-monophosphoric acid ; 1-5'-p .
INOSINIC ACID Uses
Inosinic acid (131-99-7) and its salts such as disodium inosinate are used as flavour enhancers in the food industry,.
INOSINIC ACID Toxicity Data With Reference
| 1. | orl-rat LD50:16 g/kg | ARTODN Archives of Toxicology. 47 (1981),77. | ||
| 2. | ipr-rat LD50:4850 mg/kg | ARTODN Archives of Toxicology. 47 (1981),77. | ||
| 3. | scu-rat LD50:3900 mg/kg | ARTODN Archives of Toxicology. 47 (1981),77. | ||
| 4. | ivn-rat LD50:2730 mg/kg | ARTODN Archives of Toxicology. 47 (1981),77. | ||
| 5. | orl-mus LD50:12 g/kg | ARTODN Archives of Toxicology. 47 (1981),77. | ||
| 6. | ipr-mus LD50:5400 mg/kg | ARTODN Archives of Toxicology. 47 (1981),77. | ||
| 7. | scu-mus LD50:5480 mg/kg | ARTODN Archives of Toxicology. 47 (1981),77. | ||
| 8. | ivn-mus LD50:3300 mg/kg | ARTODN Archives of Toxicology. 47 (1981),77. |
INOSINIC ACID Consensus Reports
Reported in EPA TSCA Inventory.
INOSINIC ACID Safety Profile
Moderately toxic by subcutaneous and intravenous routes. When heated to decomposition it emits toxic fumes of POx and NOx.
INOSINIC ACID Specification
Inosinic acid (131-99-7) is a nucleoside monophosphate. Inosinic acid is important in metabolism. It is the ribonucleotide of hypoxanthine and the first nucleotide formed during the synthesis of purine. It is formed by the deamination of adenosine monophosphate, and is hydrolysed to form inosine.Important derivatives of inosinic acid include purine nucleotides found in nucleic acids and adenosine triphosphate, which is used to store chemical energy in muscle and other tissues.
Related Products
- INOSINIC ACID
- 1320-06-5
- 1320-07-6
- 13200-85-6
- 13200-86-7
- 13201-46-2
- 1320-15-6
- 132019-54-6
- 132036-39-6
- 132036-42-1
- 132036-88-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View