-
Name
L(-)-Borneol
- EINECS 207-353-1
- CAS No. 464-45-9
- Article Data149
- CAS DataBase
- Density 0.992g/cm3
- Solubility insoluble in water
- Melting Point 204-208 ºC
- Formula C10H18 O
- Boiling Point 210 ºC
- Molecular Weight 154.252
- Flash Point 65 ºC
- Transport Information 1312
- Appearance White powder or chunks
- Safety Mildly toxic by ingestion. A skin irritant. When heated to decomposition it emits acrid smoke and irritating fumes. See also ALCOHOL, DENATURED; ALCOHOLS, C6-12; ALCOHOLS, C9-11; ALCOHOLS, C12-13, ETHOXYLATED; ALCOHOLS, C12-15, ETHOXYLATED; ALCOHOLS, C12-16, ETHOXYLATED; ALCOHOLS, C14-15, ETHOXYLATED; ALCOHOLS, C16-18, ETHOXYLATED; ALCOHOLS, C8-10, ETHOXYLATED PROPOXYLATED; ALCOHOLS, C12-15, ETHOXYLATED PROPOXYLATED; ALCOHOLS, N.O.S..
- Risk Codes R11
-
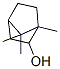
Molecular Structure
-
Hazard Symbols

- Synonyms Bicyclo[2.2.1]heptan-2-ol,1,7,7-trimethyl-, (1S-endo)-; Borneol, (1S,2R,4S)-(-)- (8CI); (-)-(1S,4S)-Borneol;(-)-Borneol; (-)-[(1S)-endo]-Borneol; (-)-endo-Borneol; (1S)-(-)-Borneol;(1S)-endo-(-)-Borneol; (1S)-endo-Borneol; (1S,2R,4S)-Borneol;(1S-endo)1,7,7-Trimethyl-bicyclo[2.2.1]heptan-2-ol; (2R)-(-)-Borneol;L-Borneol; Linderol; Ngai camphor; endo-(-)-Borneol; endo-(1S)-(-)-Borneol;l-Borneol
- PSA 20.23000
- LogP 2.19350
Synthetic route
-
-
88390-69-6
bornyl trimethylsilyl ether

-
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| With methanol; 1,3-disulfonic acid imidazolium hydrogen sulfate at 20℃; for 0.0833333h; Green chemistry; | 96% |

| Conditions | Yield |
|---|---|
| With trans-bis(triphenylphosphine)palladium dichloride; dibutyltin In methanol at 25℃; for 3h; Inert atmosphere; | 96% |

-
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran at 20℃; for 3h; deprotection; | 94% |
-
-
20053-48-9
L-bornyl p-toluenesulfonate

-
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| With magnesium In methanol for 6h; Ambient temperature; | 90% |

-
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| With methanol; toluene-4-sulfonic acid In tetrahydrofuran at 70℃; for 24h; deprotection of alcoholic OH; | 88% |
-
-
54384-77-9
(1S,2R,4S)-2-(4-Methoxy-benzyloxy)-1,7,7-trimethyl-bicyclo[2.2.1]heptane

-
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; sodium cyanoborohydride In tetrahydrofuran for 12h; Heating; | 85% |
-
-
100703-54-6
<2(R)-(1-bornyloxy)-5,6-dihydro-2H-pyran-5(R)-yl>acetic acid

-
A
-
464-45-9
endo-borneol

-
B
-
87614-57-1
3a(S),7a(S)-dihydro-4H-furo<3,2-c>pyran-2(3H)-one

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In dichloromethane at -78℃; | A n/a B 84% |
| With tin(IV) chloride; potassium carbonate; triethylamine 1.) CH2Cl2, -78 deg C, 30 min, 2.) RT, 10 min; Yield given. Multistep reaction. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonate)oxovanadium; methyl 3,5-bis((1H-1,2,4-triazol-1-yl)methyl)benzoate; oxygen; sodium acetate at 120℃; for 48h; | A 70% B 80% |

| Conditions | Yield |
|---|---|
| With bis(tri-n-butyltin)oxide In acetonitrile at 90℃; for 96h; | 12.7% |
| With the cultured cells of Nicotiana tabacum at 25℃; for 240h; biotransformation of various acetates in the dark; |
-
-
56-23-5
tetrachloromethane

-
-
85-44-9
phthalic anhydride

-
-
64-17-5
ethanol

-
-
18172-67-3, 19902-08-0
beta-pinene

-
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| at 135 - 140℃; anschl. verseifen; |


| Conditions | Yield |
|---|---|
| With ethanol; xylene at 135 - 140℃; Hydrolyse des Reaktionsprodukts; | |
| With Pinene; ethanol at 135 - 140℃; Hydrolyse des Reaktionsprodukts; |
-
-
18172-67-3, 19902-08-0
beta-pinene

-
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; acetic acid at 50 - 55℃; | |
| With acetic anhydride; acetic acid; benzenesulfonic acid | |
| With trichloroacetic acid |

| Conditions | Yield |
|---|---|
| With boron trioxide at 90 - 95℃; Hydrolyse des Reaktionsprodukts; |

| Conditions | Yield |
|---|---|
| With 2,2'-iminobis[ethanol]; (-)-diisopinocamphenylborane chloride Product distribution; multistep reaction: 1.) room temperature, 0.5 h, 2.) Et2O, 2 h; stereoselective reduction of α-chiral ketones with (+)- and (-)-B-chlorodiisopinocampheylboranes; diastereomeric excess; selectivity of matched and mismatched pairs; possible mechanism; | |
| With isopropyl alcohol; zirconium(IV) oxide at 150℃; for 0.5h; Yield given. Yields of byproduct given; | |
| With aluminum oxide; sodium; isopropyl alcohol In tetrahydrofuran for 5h; Heating; | A 74.5 % Chromat. B 25.5 % Chromat. |

| Conditions | Yield |
|---|---|
| With isopropyl alcohol; zirconium(IV) oxide at 200℃; for 0.5h; Yield given. Yields of byproduct given; |

-
-
20053-48-9
L-bornyl p-toluenesulfonate

-
A
-
464-45-9
endo-borneol

-
B
-
79-92-5
(+)-camphene

-
C
-
18383-34-1
(+)-bornene

| Conditions | Yield |
|---|---|
| With lithium; ethylamine for 24h; Heating; Title compound not separated from byproducts; | A 90 % Chromat. B 2 % Chromat. C 8 % Chromat. |

-
-
6627-72-1
rac-endo-borneol

-
A
-
464-48-2
(1S)-camphor

-
B
-
464-45-9
endo-borneol

-
C
-
464-49-3
(1R,4R)-1,7,7-trimethylbicyclo[2.2.1]heptan-2-one

| Conditions | Yield |
|---|---|
| at 25℃; Yield given. Yields of byproduct given; |

-
-
117556-75-9
<2(S)-(1-bornyloxy)-5,6-dihydro-2H-pyran-5(S)-yl>acetic acid

-
A
-
464-45-9
endo-borneol

-
B
-
117556-76-0
3a(R),7a(R)-dihydro-4H-furo<3,2-c>pyran-2(3H)-one

| Conditions | Yield |
|---|---|
| With tin(IV) chloride; potassium carbonate; triethylamine 1.) CH2Cl2, -78 deg C, 30 min, 2.) RT, 10 min; Yield given. Multistep reaction; |
-
-
117556-79-3
<2(S)-(1-bornyloxy)-5,6-dihydro-2H-pyran-5(R)-yl>acetic acid

-
A
-
464-45-9
endo-borneol

-
B
-
87614-57-1
3a(S),7a(S)-dihydro-4H-furo<3,2-c>pyran-2(3H)-one

| Conditions | Yield |
|---|---|
| With tin(IV) chloride; potassium carbonate; triethylamine 1.) CH2Cl2, -78 deg C, 30 min, 2.) RT, 10 min; Yield given. Multistep reaction. Yields of byproduct given; | |
| With tin(IV) chloride In dichloromethane at -78℃; |

| Conditions | Yield |
|---|---|
| With (+)-β-chlorodiisopinocampheylborane; 2,2'-iminobis[ethanol] 1.) room temperature, 2 h, 2.) Et2O, 2 h; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; |


-
A
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| In water; ethylene glycol at 25℃; Equilibrium constant; decomplexation; |

-
A
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| In water; ethylene glycol at 25℃; Equilibrium constant; decomplexation; |

-
A
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| In water; ethylene glycol at 25℃; Equilibrium constant; decomplexation; |

-
A
-
464-45-9
endo-borneol

| Conditions | Yield |
|---|---|
| In water; ethylene glycol at 25℃; Equilibrium constant; decomplexation; |

-
A
-
464-45-9
endo-borneol

-
B
-
202332-62-5
mono<6-(benzylseleno)-6-deoxy>-β-cyclodextrin

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 25℃; Equilibrium constant; |

-
A
-
464-45-9
endo-borneol

-
B
-
202332-61-4
mono<6-(p-tolylseleno)-6-deoxy>-β-cyclodextrin

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 25℃; Equilibrium constant; |

-
A
-
464-45-9
endo-borneol

-
B
-
211802-10-7
mono<6-(m-tolylseleno)-6-deoxy>-β-cyclodextrin

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 25℃; Equilibrium constant; |

-
A
-
464-45-9
endo-borneol

-
B
-
211802-08-3
mono<6-(o-tolylseleno)-6-deoxy>-β-cyclodextrin

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 25℃; Equilibrium constant; |
L(-)-Borneol Chemical Properties
MF: C10H18O
MW: 154.25
EINECS: 207-353-1
mp: 206-209 °C
bp: 210 °C(lit.)
alpha: -36.2 o (c=5, C2H5OH)
vapor density: 5.31 (vs air)
vapor pressure: 33.5 mm Hg ( 25 °C)
FEMA: 2157
refractive index: -36 ° (C=5, EtOH)
Fp: 150 °F
storage temp: Flammables area
Water Solubility: INSOLUBLE
Merck: 14,1338
BRN: 3587558
Synonyms:CAMPHOL-(+)-BORNOL;BORNEO CAMPHOR;BORNEOL FLAKES;BORNYL ALCOHOL;FEMA 2157;L-BORNEO CAMPHOR;L(-)-BORNEOL;L-BORNEOL
The structure of NGAI CAMPHOR is:
L(-)-Borneol Toxicity Data With Reference
2、LD50/LC50: RTECS: CAS# 464-45-9: Draize test, rabbit, skin: 500 mg/24H Mild;Oral, rat: LD50 = 5800 mg/kg;
3、Carcinogenicity: L(-)-Borneol - Not listed as a carcinogen by ACGIH, IARC, NTP, or CA Prop 65.
4、Other: See actual entry in RTECS for complete information. The toxicological properties have not been fully investigated.
| ||||
|
L(-)-Borneol Consensus Reports
L(-)-Borneol Safety Profile
RIDADR: UN 1312 4.1/PG 3
WGK Germany: 2
RTECS: DT5095000
HazardClass: 4.1
PackingGroup: III
HS Code 29061900
Mildly toxic by ingestion. A skin irritant. When heated to decomposition it emits acrid smoke and irritating fumes.
L(-)-Borneol Specification
General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. Will burn if involved in a fire. Flammable solid.
Extinguishing Media: Use water spray, dry chemical, carbon dioxide, or chemical foam.
2、Handling and Storage
Handling: Use spark-proof tools and explosion proof equipment. Avoid breathing dust, vapor, mist, or gas. Avoid contact with eyes, skin, and clothing. Avoid ingestion and inhalation.
Storage: Keep away from sources of ignition. Store in a cool, dry place. Keep container closed when not in use. Store in a tightly closed container. Flammables-area.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View