-
Name
Methyl 2-methoxy-5-sulfamoylbenzoate
- EINECS 251-358-1
- CAS No. 33045-52-2
- Article Data11
- CAS DataBase
- Density 1.36 g/cm3
- Solubility
- Melting Point 175-177 °C(lit.)
- Formula C9H11NO5S
- Boiling Point 439.8 °C at 760 mmHg
- Molecular Weight 245.256
- Flash Point 219.8 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms o-Anisicacid, 5-sulfamoyl-, methyl ester (8CI);4-Methoxy-3-(methoxycarbonyl)benzenesulfonamide;Methyl2-methoxy-5-sulfamoylbenzoate;Methyl 5-(aminosulfonyl)-2-methoxybenzoate;Methyl 5-sulfamoyl-2-methoxybenzoate;benzoic acid, 5-(aminosulfonyl)-2-methoxy-, methyl ester;Methyl-2-methoxy-5-sulfamoylbenzolcarboxylate;Methyl 2-methoxy-5-aminosulfonylbenzoate;
- PSA 104.07000
- LogP 1.91030
Synthetic route
-
-
33924-48-0
methyl 5-chloro-2-methoxybenzoate

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

| Conditions | Yield |
|---|---|
| With sodium sulfamate; copper(I) bromide In tetrahydrofuran at 50℃; for 10h; Temperature; Reflux; | 96.55% |
| With sodium aminosulfinate; copper(I) bromide In tetrahydrofuran at 40℃; for 18h; Concentration; Temperature; | 96.55% |
| With sodium aminosulfinate; copper(l) chloride In tetrahydrofuran at 40℃; for 8h; Temperature; | 96.55% |
-
-
67-56-1
methanol

-
-
22117-84-6
3-cyano-4-methoxybenzenesulfonamide

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 40℃; for 6h; Time; | 88.6% |
-
-
67-56-1
methanol

-
-
22117-85-7
2-methoxy-5-sulfamoylbenzoic acid

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
22117-85-7
2-methoxy-5-sulfamoylbenzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol | 98.3% |
| Stage #1: methyl 2-methoxy-5-sulfamoyl-benzoate With sodium hydroxide; water In methanol at 20℃; for 1h; Stage #2: With hydrogenchloride In methanol; water | 98.3% |
| Stage #1: methyl 2-methoxy-5-sulfamoyl-benzoate With sodium hydroxide; water In methanol at 20℃; for 1h; Stage #2: With hydrogenchloride In methanol; water | 98.3% |
-
-
64-17-5
ethanol

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
33045-53-3
2-methoxy-5-sulfamoylbenzoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid for 12h; Heating; | 98% |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
22795-99-9
(S)-2-(Aminomethyl)-1-ethylpyrrolidine

-
-
15676-16-1, 23672-06-2, 23672-07-3, 23756-79-8
(S)-(-)-sulpiride

| Conditions | Yield |
|---|---|
| at 90 - 100℃; for 5h; Inert atmosphere; | 93.8% |
| With hydrogenchloride In water; butan-1-ol | 75% |
-
-
26116-12-1
1-ethyl-2-pyrrolidinemethanamine

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
15676-16-1
N-[(1-ethyl-2-pyrrolidinyl)methyl]-2-methoxy-5-sulfamoylbenzamide

| Conditions | Yield |
|---|---|
| In glycerol at 90 - 95℃; for 10h; Inert atmosphere; | 88.4% |
| In ethylene glycol at 90 - 100℃; Solvent; Inert atmosphere; | 85.1% |
| at 100℃; for 3h; | 75% |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
108-24-7
acetic anhydride

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 20℃; | 77% |

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

| Conditions | Yield |
|---|---|
| In methanol at 110℃; for 20h; | 74.4% |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

| Conditions | Yield |
|---|---|
| In methanol at 110℃; for 24h; | 64% |
-
-
251096-84-1
3-(3,4-dichlorobenzylthio)thiophene-2-carboxylic acid

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
1336982-58-1
methyl 5-(N-(3-(3,4-dichlorobenzylthio)thiophene-2-carbonyl)sulfamoyl)-2-methoxybenzoate

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 25℃; for 6h; Inert atmosphere; | 23% |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate


| Conditions | Yield |
|---|---|
| In methanol at 110℃; for 24h; Yield given; |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
113681-63-3
N-benzyl-5-(aminosulfonyl)-2-methoxybenzamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 98 percent / H2SO4 / 12 h / Heating 2: 5 percent / sodium methoxide / 5 h / 100 °C View Scheme |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
113681-64-4
N-(1-methyl-4-piperidinyl)-5-(aminosulfonyl)-2-methoxybenzamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 98 percent / H2SO4 / 12 h / Heating 2: 54 percent / NaH / 1.) 100 deg C, 5 h; 2.) 120-130 deg C, 6 h View Scheme |

-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
55-21-0
benzamide

| Conditions | Yield |
|---|---|
| In water; ethylene glycol; acetic acid |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

-
-
900165-91-5
3-(Hydroxymethyl)-4-methoxybenzenesulfonamide

| Conditions | Yield |
|---|---|
| With lithium borohydride In tetrahydrofuran; methanol at 20℃; for 8h; |
-
-
33045-52-2
methyl 2-methoxy-5-sulfamoyl-benzoate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: zinc(II) chloride / 20 °C 2: silver(I) acetate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer / 1,2-dichloro-ethane / 5 h / 60 °C / Sealed tube View Scheme |
Methyl 2-methoxy-5-sulfamoylbenzoate Specification
The Methyl 2-methoxy-5-sulfamoylbenzoate, with the CAS registry number 33045-52-2 and EINECS registry number 251-358-1, is also known as Methyl 5-(Aminosulfonyl)-2-methoxybenzoate. It belongs to the following product categories: Fine chemical & intermediates; Organic Building Blocks; Sulfonamides/Sulfinamides; Sulfur Compounds. And the molecular formula of this chemical is C9H11NO5S. What's more, it is usually used as intermediate of sulpiride.
The physical properties of Methyl 2-methoxy-5-sulfamoylbenzoate are as following: (1)ACD/LogP: 0.78; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.78; (4)ACD/LogD (pH 7.4): 0.78; (5)ACD/BCF (pH 5.5): 2.3; (6)ACD/BCF (pH 7.4): 2.29; (7)ACD/KOC (pH 5.5): 63.1; (8)ACD/KOC (pH 7.4): 62.92; (9)#H bond acceptors: 6; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 81.29 ?2; (13)Index of Refraction: 1.541; (14)Molar Refractivity: 56.66 cm3; (15)Molar Volume: 180.2 cm3; (16)Polarizability: 22.46 10-24cm3; (17)Surface Tension: 48.3 dyne/cm; (18)Density: 1.36 g/cm3; (19)Flash Point: 219.8 °C; (20)Enthalpy of Vaporization: 69.68 kJ/mol; (21)Boiling Point: 439.8 °C at 760 mmHg; (22)Vapour Pressure: 6.2E-08 mmHg at 25°C.
Uses of Methyl 2-methoxy-5-sulfamoylbenzoate: It can react with ethanol to produce ethyl 5-sulfamoyl-o-anisate. This reaction will need reagent H2SO4. The reaction time is 12 hours with heating, and the yield is about 98%.
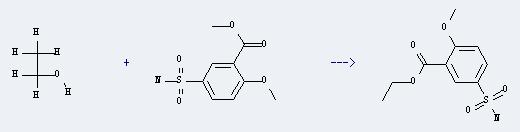
You can still convert the following datas into molecular structure:
(1)SMILES: O=S(=O)(c1ccc(OC)c(C(=O)OC)c1)N
(2)InChI: InChI=1/C9H11NO5S/c1-14-8-4-3-6(16(10,12)13)5-7(8)9(11)15-2/h3-5H,1-2H3,(H2,10,12,13)
(3)InChIKey: MKDYDRQLKPGNNU-UHFFFAOYAY
Related Products
- Methyl 1-Benzyl-5-oxopyrrolidine-3-carboxylate
- Methyl (((methoxymethylphosphinothioyl)thio)acetyl)methylcarbamate
- Methyl (+)-(3R)-7-[4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methanesulfonylamino)pyrimidin-5-yl]-3-hydroxy-5-oxo-(6E)-heptenoate
- Methyl (2-amino-5-methyl-1,3-thiazol-4-yl)acetate
- Methyl (2-chloromethyl)oxazole-4-carboxylate
- Methyl (2E)-3-(4-methylphenyl)propenoate
- Methyl (2E)-3-cyclohexylprop-2-enoate
- Methyl (2R)-2-[(tert-butoxycarbonyl)amino]-3-iodopropanoate
- Methyl (2R)-2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate
- Methyl (2R)-2-amino-2-cyclohexylethanoate hydrochloride
- 33045-53-3
- 330457-42-6
- 330457-44-8
- 330459-34-2
- 3304-59-4
- 33046-81-0
- 3304-70-9
- 330-48-3
- 33048-55-4
- 330-54-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View