-
Name
N-Phenylmaleimide
- EINECS 213-382-0
- CAS No. 941-69-5
- Article Data163
- CAS DataBase
- Density 1.329 g/cm3
- Solubility Soluble in water (slightly ), methanol, ethanol, and benzene.
- Melting Point 85-87 °C(lit.)
- Formula C10H7NO2
- Boiling Point 299.26 °C at 760 mmHg
- Molecular Weight 173.171
- Flash Point 136.835 °C
- Transport Information UN 2811 6.1/PG 3
- Appearance yellow crystalline powder or needles
- Safety 45-24/25-36/37/39-26
- Risk Codes 25-36/37/38-23/24/25
-
Molecular Structure
-
Hazard Symbols
 T,
T, Xi
Xi
- Synonyms Maleimide,N-phenyl- (8CI);1-Phenyl-1H-pyrrole-2,5-dione;1-Phenyl-pyrrole-2,5-dione;Imilex P;Maleanil;Maleimidobenzene;N-Phenylmaleic acid imide;1H-Pyrrole-2,5-dione,1-phenyl-;N-Phenylpyrrole-2,5-dione;NSC8183;Phenylmaleimide;
- PSA 37.38000
- LogP 1.18100
Synthetic route
-
-
555-59-9, 4437-08-5, 37902-58-2
N-phenylmaleamic acid

-
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With sulfuric acid In dimethyl sulfoxide; toluene Cyclization; Heating; | 100% |
| With sodium acetate at 80 - 85℃; for 4h; | 85% |
| With 1-methyl-3-(4-sulfobutyl)-1H-imidazol-3-ium hydrogensulfate In 1-Propyl acetate for 3h; Reflux; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: aniline With 2,4-dimethyl-6-tert-butylphenol; triethylamine; phosphoric acid; zinc diacetate In xylene for 0.0833333h; Stage #2: maleic anhydride In xylene at 140℃; for 6h; Product distribution / selectivity; | 98% |
| With phosphoric acid; 2,4-dimethyl-6-tert-butylphenol; zinc diacetate; silica gel; triethylamine In xylene at 140℃; for 6h; Product distribution / selectivity; Industry scale; | 98% |
| Stage #1: maleic anhydride With 2,5-dimethylbenzenesulfonic acid; toluene-4-sulfonic acid In tetrahydrofuran; benzene at 85℃; Stage #2: aniline In tetrahydrofuran; benzene at 85℃; for 1h; Temperature; Reagent/catalyst; Solvent; | 98.75% |
-
-
21306-38-7, 65391-92-6, 106974-71-4
7-phenyl-1,7-diaza-bicyclo<2.2.1>heptane-exo-2,3-exo-5,6-tetracarboxylic acid bisphenylimide

-
A
-
941-69-5
N-phenyl-maleimide

-
B
-
106958-76-3
2,5-diphenyl pyrrolo<3,4-c>pyrazole-(2H,5H)-4,6-dione

| Conditions | Yield |
|---|---|
| at 310℃; under 0.1 Torr; | A 95% B 70% |
-
-
21306-38-7, 65391-92-6, 106974-71-4
7-phenyl-1,7-diaza-bicyclo<2.2.1>heptane-endo-2,3-exo-5,6-tetracarboxylic acid bisphenylimide

-
A
-
941-69-5
N-phenyl-maleimide

-
B
-
106958-76-3
2,5-diphenyl pyrrolo<3,4-c>pyrazole-(2H,5H)-4,6-dione

| Conditions | Yield |
|---|---|
| at 310℃; under 0.1 Torr; | A 95% B 70% |
-
-
36342-11-7
3-chloro-1-phenylpyrrolidine-2,5-dione

-
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With triethylamine In diethyl ether for 2h; | 85% |
-
-
37902-58-2
3-phenylcarbamoylacrylic acid

-
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With ammonium peroxodisulfate In 1,4-dioxane; dimethyl sulfoxide at 100℃; for 7h; | 84% |
| With sodium acetate; acetic anhydride at 70℃; for 1h; | 72% |
| With sodium acetate; acetic anhydride at 100℃; for 0.5h; | 43% |

| Conditions | Yield |
|---|---|
| In toluene at 110℃; under 760.051 Torr; for 8h; | A 83% B 55 mg C 6 mg |


| Conditions | Yield |
|---|---|
| With acetic acid at 100℃; for 1.16667h; | 78% |

| Conditions | Yield |
|---|---|
| With acetic acid at 100℃; for 1.33333h; | 75% |

| Conditions | Yield |
|---|---|
| With oxygen; Rose Bengal lactone In water; N,N-dimethyl-formamide for 18h; Inert atmosphere; Irradiation; | 74% |
-
-
136163-24-1
N-phenylmaleisoimidium perchlorate

-
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With piperidine; triethylamine In diethyl ether for 10h; | 65% |
| With morpholine; triethylamine In diethyl ether for 10h; | 65% |
-
-
617-48-1
malic acid

-
-
62-53-3
aniline

-
A
-
20400-73-1
D,L-2-Hydroxy-N-phenylsuccinimide

-
B
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| for 0.25h; Condensation; Heating; microwave irradiation; | A 62% B 19% |


| Conditions | Yield |
|---|---|
| Stage #1: 3-phenylcarbamoylacrylic acid; acetic anhydride With sodium acetate at 50℃; for 2h; Inert atmosphere; Stage #2: With water at 70℃; for 2h; Inert atmosphere; | 44% |

| Conditions | Yield |
|---|---|
| With copper diacetate; oxygen; triethylamine; 4 A molecular sieve In dichloromethane at 45℃; for 20h; | 43% |

| Conditions | Yield |
|---|---|
| With triethylamine; 1,2-bis-(diphenylphosphino)ethane; zinc; dibromo[1,2-bis(diphenylphosphino)ethane]nickel(II) In acetonitrile at 80℃; for 36h; | 37% |
-
-
81280-21-9
C21H19NO3

-
A
-
941-69-5
N-phenyl-maleimide

-
B
-
75950-76-4
1-methoxy-1-methyl-1H-indene

-
-
81280-24-2, 81339-68-6
(5aS,5bR,8aR,8bS,8cR)-8b-Methoxy-8c-methyl-7-phenyl-5b,8a,8b,8c-tetrahydro-5aH-7-aza-dicyclopenta[a,cd]indene-6,8-dione

-
-
81280-24-2, 81339-68-6
(5aS,5bS,8aS,8bS,8cR)-8b-Methoxy-8c-methyl-7-phenyl-5b,8a,8b,8c-tetrahydro-5aH-7-aza-dicyclopenta[a,cd]indene-6,8-dione

| Conditions | Yield |
|---|---|
| at 80 - 100℃; under 0.08 Torr; | A 31% B 3% C 33% D 21% |

-
-
56041-56-6, 77519-61-0
N-Phenyl-endo-6-methyl-8-ethyl-8-azabicyclo<2.2.2>oct-4-en-7-one-1,2-dicarboximide

-
A
-
77475-04-8
1-Ethyl-3-methyl-2-pyridone

-
B
-
941-69-5
N-phenyl-maleimide

-
-
56041-56-6, 77519-61-0
N-Phenyl-exo-6-methyl-8-ethyl-8-azabicyclo<2.2.2>oct-4-en-7-one-1,2-dicarboximide

| Conditions | Yield |
|---|---|
| In xylene for 42h; Heating; | A n/a B n/a C 25% |
| In xylene for 42h; Product distribution; Heating; convertion of endo into exo isomer; | A n/a B n/a C 25% |


| Conditions | Yield |
|---|---|
| In toluene at 110℃; under 760.051 Torr; for 8h; | A 24% B 14 mg |
-
-
90278-98-1
N-phenyl-maleamic acid methyl ester

-
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; phosphorus pentoxide |
-
-
67463-35-8
endo-4-phenyl-4-aza-10-oxatricyclo<5.2.1.0>dec-8-ene-3,5-dione

-
A
-
110-00-9
furan

-
B
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| In chloroform at 43℃; under 750060 Torr; Kinetics; Thermodynamic data; var.temp. and solv: dioxane; Ea and ΔS(excit.); |
-
-
99237-86-2
exo-10-acetyl-4-phenyl-4,10-diazatricyclo<5.2.1.02,6>dec-8-ene-3,5-dione

-
A
-
609-41-6
N-acetylpyrrole

-
B
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 55.8℃; under 750060 Torr; Kinetics; Thermodynamic data; var.temp. and solv: dioxane; Ea and ΔS(excit.); |

-
A
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With aluminium trichloride In benzene at 25℃; Equilibrium constant; distribution constant AlCl3 between the imide and its adduct with anthracene was measured; |

-
-
99295-58-6
endo-10-benzoyl-4-phenyl-4,10-diazatricyclo<5.2.1.02,6>dec-8-ene-3,5-dione

-
A
-
5145-65-3
N-benzoylpyrrole

-
B
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| In chloroform at 30.7℃; under 750060 Torr; Kinetics; Thermodynamic data; var.temp., pressure and solv: CH2Cl2, AcOEt, MeCN, Me2SO, THF, dioxane; Ea and ΔS(excit.); |
-
-
99237-79-3
exo-10-benzoyl-4-phenyl-4,10-diazatricyclo<5.2.1.02,6>dec-8-ene-3,5-dione

-
A
-
5145-65-3
N-benzoylpyrrole

-
B
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 71.7℃; under 750060 Torr; Kinetics; Thermodynamic data; var.temp. and solv: dioxane; Ea and ΔS(excit.); |
-
-
84319-82-4
N-phenyl-c-3,c-4-bis(2-hydroxyphenyl)-r-1,c-2-cyclobutanedicarboximide

-
A
-
941-69-5
N-phenyl-maleimide

-
B
-
18221-50-6
dihydroxy-stilbene

| Conditions | Yield |
|---|---|
| In methanol for 0.5h; Product distribution; Ambient temperature; Irradiation; correlation between photochemical fission and chemical structure; |

| Conditions | Yield |
|---|---|
| Kinetics; Thermodynamic data; Ambient temperature; decomposition, also 80 deg C, ΔG(excit.); |
-
-
21939-12-8
2,3,5-Triphenyl-tetrahydro-pyrrolo[3,4-d]isoxazole-4,6-dione

-
A
-
941-69-5
N-phenyl-maleimide

-
B
-
201024-81-9
C,N-diphenylnitrone

| Conditions | Yield |
|---|---|
| With lithium perchlorate In diethyl ether at 25℃; Equilibrium constant; various concentrations of LiClO4 (also in the absence of LiClO4); |
-
-
27742-33-2
3,6-epoxy-N-phenyl-1,2,3,6-tetrahydrophthalimide

-
A
-
110-00-9
furan

-
B
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With lithium perchlorate In diethyl ether at 25℃; Equilibrium constant; various concentrations of LiClO4 (also in the absence of LiClO4; |
-
-
941-69-5
N-phenyl-maleimide

-
-
5471-63-6
1,3-diphenylisobenzofuran

-
-
36213-67-9
2,4,9-triphenyl-(3ac,9ac)-3a,4,9,9a-tetrahydro-4r,9c-epioxido-benzo[f]isoindole-1,3-dione

| Conditions | Yield |
|---|---|
| With ethyl acetate for 0.0166667h; Diels-Alder Cycloaddition; | 100% |
| In toluene |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
941-69-5
N-phenyl-maleimide

-
-
53114-48-0
7-Phenyl-2,3,7-triazabicyclo<3.3.0>oct-2-ene-6,8-dione

| Conditions | Yield |
|---|---|
| In diethyl ether for 1h; | 100% |
| In diethyl ether for 1h; Ambient temperature; | 80% |
-
-
941-69-5
N-phenyl-maleimide

-
-
153924-62-0
N-benzylidenephenylglycine methyl ester

-
-
82461-27-6, 113430-23-2
methyl 2,c-4,7-triphenyl-6,8-dioxo-3,7-diazabicyclo-<3.3.O>octane-r-2-carboxylate

| Conditions | Yield |
|---|---|
| With triethylamine Product distribution; Ambient temperature; effect of solvents, metal salts as catalysts, and time; | 100% |
| In perdeuteriopyridine at 110℃; for 1.5h; | 100% |
| In toluene for 48h; Heating; | 86% |
-
-
941-69-5
N-phenyl-maleimide

-
-
85739-52-2
1,2,3,4,5-Pentamethyl-cyclopenta-2,4-dienylamine

-
-
114564-30-6
(1S,2R,6S,7R,10S)-10-Amino-1,7,8,9,10-pentamethyl-4-phenyl-4-aza-tricyclo[5.2.1.02,6]dec-8-ene-3,5-dione

| Conditions | Yield |
|---|---|
| In dichloromethane at 22℃; for 3.5h; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
114564-20-4
5-(acetylamino)-1,2,3,4,5-pentamethylcyclopentadiene

-
-
114564-29-3, 125277-86-3
N-((1S,2R,6S,7R,10S)-1,7,8,9,10-Pentamethyl-3,5-dioxo-4-phenyl-4-aza-tricyclo[5.2.1.02,6]dec-8-en-10-yl)-acetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 22℃; for 3.5h; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
16883-69-5
N-phenacylpyridinium bromide

-
-
88121-62-4
(3aS,4S,9aR,9bR)-4-Benzoyl-2-phenyl-3a,4,9a,9b-tetrahydro-pyrrolo[3,4-a]indolizine-1,3-dione

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform for 0.166667h; Ambient temperature; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
36377-40-9
pyridinium benzoylmethylide

-
-
88121-62-4
(3aS,4S,9aR,9bR)-4-Benzoyl-2-phenyl-3a,4,9a,9b-tetrahydro-pyrrolo[3,4-a]indolizine-1,3-dione

| Conditions | Yield |
|---|---|
| In chloroform for 0.166667h; Ambient temperature; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
107836-10-2
1-methoxy-1-trimethylsiloxy-1,3,5-hexatriene

-
-
107836-00-0
(1,3-Dioxo-2-phenyl-2,3,3a,4,7,7a-hexahydro-1H-isoindol-4-yl)-acetic acid methyl ester

| Conditions | Yield |
|---|---|
| In benzene for 1h; Ambient temperature; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
82167-48-4
2,3-bis<(trimethyksilyl)methyl>-1,3-butadiene

-
-
82167-60-0
2-phenyl-5,6-di(trimethylsilylmethyl)-3a,4,7,7a-tetrahydro-1H-isoindole-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With hydroquinone In toluene Heating; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
542-92-7
cyclopenta-1,3-diene

-
-
26234-46-8, 29377-36-4, 56552-68-2
N-phenylbicyclo[2.2.1]hept-5-ene-2-endo,3-endo-dicarboximide

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 20℃; for 0.0166667h; Diels-Alder Cycloaddition; | 100% |
| With 1-hexyl-3-methylimidazolium tetrafluoroborate; K-10 montmorillonite at 20℃; for 0.0833333h; Diels-Alder reaction; | 99% |
| With 3-(n-butoxycarbonyl)-1-methylpyridinium bis(trifluoromethanesulfonyl)imide at 20℃; for 0.166667h; Diels-Alder reaction; diastereoselective reaction; | 97% |

| Conditions | Yield |
|---|---|
| In benzene at 80℃; for 30h; | 100% |
-
-
941-69-5
N-phenyl-maleimide


| Conditions | Yield |
|---|---|
| In benzene at 75 - 80℃; for 22h; | 100% |

| Conditions | Yield |
|---|---|
| With acetic anhydride; zinc In toluene at 40 - 86℃; for 48h; Inert atmosphere; chemoselective reaction; | 100% |
| With 1,2,2,3,4,4-hexamethylphosphetane 1-oxide; phenylsilane; water In toluene at 80℃; for 24h; Schlenk technique; Inert atmosphere; | 99% |
| With ammonium formate; acetic acid; palladium on activated charcoal at 110℃; for 0.166667h; Hydrogenation; | 96% |

| Conditions | Yield |
|---|---|
| With polymer supported 1-silyloxy-1,3-butadiene In dichloromethane at 25℃; for 14h; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
252637-04-0
ethyl 3-[(dimethylamino)methyl]-1H-indole-2-acetate

| Conditions | Yield |
|---|---|
| In toluene for 2h; Addition; elimination; Heating; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
153704-91-7
(2Z, 4E)-1,3,6-triacetoxyhexa-2,4-diene

| Conditions | Yield |
|---|---|
| hydroquinone In toluene for 7.5h; Cycloaddition; Heating; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
287920-04-1
tert-butyl-dimethyl-[1-methylene-3-(2-nitro-phenyl)-allyloxy]-silane

| Conditions | Yield |
|---|---|
| In benzene at 20℃; for 72h; Cycloaddition; Diels-Alder reaction; | 100% |
-
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 20h; Diels-Alder reaction; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
325479-30-9
(R,E)-3-(tert-butyldimethylsiloxy)-1-(4-phenyl-2-oxazolidinon-3-yl)-1,3-butadiene

-
-
325479-39-8
(3aS,4R,7aS)-6-(tert-butyldimethylsilyloxy)-3a,4,7,7a-tetrahydro-2-phenyl-4-[(R)-4-phenyl-2-oxazolidinon-3-yl]-1H-isoindole-1,3-(2H)-dione

| Conditions | Yield |
|---|---|
| In toluene at -45 - 20℃; for 24h; Diels-Alder reaction; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
552290-47-8
2-(1,2,3,4,5-pentamethyl-cyclopenta-2,4-dienyl)-4,5-dihydro-oxazole

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 25℃; Diels-Alder reaction; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 5h; | 100% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 1h; | 100% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 3.5h; | 100% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 5h; | 100% |

-
-
868564-65-2
(-)-(S)-2-(E)-4-(Z)-5-phenyl-4-(p-tolylsulfinyl)penta-2,4-dien-1-ol

-
-
941-69-5
N-phenyl-maleimide

-
-
868564-67-4
(+)-(3aR,4S,7S,7aR,SS)-7-(hydroxymethyl)-2,4-diphenyl-5-(p-tolylsulfinyl)-3a,4,7,7a-tetrahydro-2H-isoindole-1,3-dione

| Conditions | Yield |
|---|---|
| In toluene at 20℃; for 72h; Diels-Alder cycloaddition; | 100% |

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 25℃; Diels-Alder reaction; | 100% |
-
-
941-69-5
N-phenyl-maleimide

| Conditions | Yield |
|---|---|
| With H(1+) In perchloric acid aq. HClO4; to the soln. of Co-compd. in aq. HClO4 was added an org. compd.; after 1-2 h the soln. was dild. with H2O; the react. mixt. was absorbed onto an ion-exchange column; washing with aq. HClO4, elution with NaClO4 (pH 2);; Ba(NO3)2 and K2SO4 were added; KClO4 and BaSO4 were removed by fitration; the soln. was condensed by rotoevapn. at 30°C and was filtered; addn. of HClO4, standing overnight at 8°C; recrystn. from HClO4, cooling for 4 h at the same temp.;; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
146254-96-8
3-[1'-(tert-butyldimethylsilyloxy)ethyl]-4-[1-methyleneprop-2-enyl]azetidin-2-one

-
-
1000700-65-1
5-{(2S,3S)-3-[(R)-1-(tert-butyldimethylsilyloxy)ethyl]-4-oxoazetidin-2-yl}-2-phenyl-3a,4,7,7a-tetrahydroisoindole-1,3-dione

| Conditions | Yield |
|---|---|
| With indium(III) chloride In acetonitrile at 20℃; for 48h; Diels-Alder Reaction; | 100% |
-
-
941-69-5
N-phenyl-maleimide

-
-
872-53-7
cyclopentanealdehyde

-
-
1242461-34-2
(S)-1-(2,5-dioxo-1-phenylpyrrolidin-3-yl)cyclopetanecarboxaldehyde

| Conditions | Yield |
|---|---|
| With (S)-3-amino-3-phenylpropanoic acid; caesium carbonate In dichloromethane at 20℃; for 48h; Michael Addition; enantioselective reaction; | 100% |
| With NH2-Phg-(D-Pro)-Gly-Leu-OH In acetonitrile at 20℃; for 72h; Michael Addition; enantioselective reaction; | 95% |
| Stage #1: cyclopentanealdehyde With dmap; tert-butyl (2S,3R)-2-amino-3-hydroxybutanoate; SULFAMIDE In dichloromethane at 23℃; for 0.0333333h; Michael addition; Stage #2: N-phenyl-maleimide In dichloromethane at 23℃; for 16h; Michael addition; optical yield given as %ee; enantioselective reaction; | 93% |
N-Phenylmaleimide Chemical Properties
Molecular Structure of N-Phenylmaleimide (CAS NO.941-69-5):
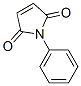
IUPAC Name: 1-Phenylpyrrole-2,5-dione
Molecular Formula: C10H7NO2
Molecular Weight: 173.17 g/mol
Density: 1.329 g/cm3.
Melting Point: 85-87 °C
Boiling Point: 162-163 °C at 12 mm Hg(lit.)
Flash Point: 136.8 °C
Index of Refraction: 1.629
Molar Refractivity: 46.32 cm3
Molar Volume: 130.2 cm3
Surface Tension: 57.4 dyne/cm
XLogP3: 1.1
H-Bond Acceptor: 2
Rotatable Bond Count: 1
Exact Mass: 173.047678
MonoIsotopic Mass: 173.047678
Topological Polar Surface Area: 37.4
Heavy Atom Count: 13
Canonical SMILES: C1=CC=C(C=C1)N2C(=O)C=CC2=O
InChI: InChI=1S/C10H7NO2/c12-9-6-7-10(13)11(9)8-4-2-1-3-5-8/h1-7H
InChIKey: HIDBROSJWZYGSZ-UHFFFAOYSA-N
EINECS: 213-382-0
Product Categories: N-Substituted Maleimides;N-Substituted Maleimides, Succinimides & Phthalimides;Carbonyl Compounds;Cyclic Imides;Organic Building Blocks
N-Phenylmaleimide Uses
N-Phenylmaleimide (941-69-5) is used as sulfide cross-linking agent in caoutchouc and synthetic rubber. It is also used as heat-resistant modifying agent in the resins of ABS, PVC, and PMMA. N-Phenylmaleimide is used as the intermediate of resins and is prepared for heat resistant polymers. N-Phenylmaleimide is widely used in coatings, binder, light-sensitive resins medicines, farm chemicals and so on.
N-Phenylmaleimide Toxicity Data With Reference
| 1. | skn-rbt 500 mg MLD | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) | ||
| 2. | orl-rat LD50:188 mg/kg | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) | ||
| 3. | ipr-rat LDLo:25 mg/kg | NCNSA6 National Academy of Sciences, National Research Council, Chemical-Biological Coordination Center, Review. 5 (1953),22. | ||
| 4. | orl-mus LD50:78 mg/kg | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) | ||
| 5. | orl-rbt LDLo:100 mg/kg | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) |
N-Phenylmaleimide Consensus Reports
Reported in EPA TSCA Inventory.
N-Phenylmaleimide Safety Profile
Hazard Codes:  T,
T,  Xi
Xi
Risk Statements: 25-36/37/38-23/24/25
R25 :Toxic if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
R23/24/25:Toxic by inhalation, in contact with skin and if swallowed.
Safety Statements: 45-24/25-36/37/39-26
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S24/25:Avoid contact with skin and eyes.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
RIDADR: UN 2811 6.1/PG 3
WGK Germany: 3
RTECS: ON5950000
F: 10-21
Hazard Note: Irritant
HazardClass: 6.1
PackingGroup: III
HS Code: 29251995
Poison by ingestion and intraperitoneal routes. When heated to decomposition it emits toxic fumes of NOx.
N-Phenylmaleimide Specification
N-Phenylmaleimide (CAS NO.941-69-5) is also named as 5-21-10-00009 (Beilstein Handbook Reference) ; AI3-01186 ;
BRN 0125098 ; Maleanil ; Maleimidobenzene ; Maleinanil ; N-Fenylimid kyseliny maleinove ; N-Fenylimid kyseliny maleinove [Czech] ; NSC 8183 ; 1H-Pyrrole-2,5-dione, 1-phenyl- ; Maleimide, N-phenyl- . N-Phenylmaleimide (CAS NO.941-69-5) is yellow crystalline powder or needles. It is sparingly water soluble. N-Phenylmaleimide is an imide. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx). Amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Flash point data for N-Phenylmaleimide are not available. It is probably combustible.
Related Products
- N-Phenylmaleimide
- 941695-43-8
- 94169-62-7
- 94171-08-1
- 941712-23-8
- 941716-31-0
- 941716-85-4
- 941716-86-5
- 941717-10-8
- 941867-91-0
- 94187-96-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View