-
Name
Tetrabromophthalic anhydride
- EINECS 211-185-4
- CAS No. 632-79-1
- Article Data10
- CAS DataBase
- Density 2.771 g/cm3
- Solubility hydrolysis with water
- Melting Point 269-271 °C(lit.)
- Formula C8Br4O3
- Boiling Point 540.5 °C at 760 mmHg
- Molecular Weight 463.702
- Flash Point 280.7 °C
- Transport Information
- Appearance white to light yellow powder
- Safety 26-37/39-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Phthalicanhydride, tetrabromo- (6CI,7CI,8CI);3,4,5,6-Tetrabromophthalic anhydride;Bromphthal;FG 4000;FireMaster PHT 4;NSC 4874;PHT 4;Saytex RB 49;Tetrabromophthalic acid anhydride;
- PSA 43.37000
- LogP 4.04720
Synthetic route

| Conditions | Yield |
|---|---|
| With sulfuric acid; bromine; tin(IV) chloride at 30 - 80℃; for 13h; Reagent/catalyst; Temperature; | 95.1% |
| With sulfuric acid; bromine; iodine | |
| With sulfuric acid; bromine at 60℃; anschliessend Erhitzen auf 200grad; | |
| With sulfuric acid; bromine at 60℃; anschliessend Erhitzen auf 200grad; |
-
-
13810-83-8
tetrabromobenzene-1,2-dicarboxylic acid

-
-
632-79-1
Tetrabromophthalic anhydride

| Conditions | Yield |
|---|---|
| durch Sublimation; |
-
-
85-44-9
phthalic anhydride

-
-
7664-93-9
sulfuric acid

-
-
7726-95-6
bromine

-
-
632-79-1
Tetrabromophthalic anhydride

| Conditions | Yield |
|---|---|
| at 60℃; anschl. Erhitzen auf 200grad; | |
| at 60℃; anschl. Erhitzen auf 200grad; |


| Conditions | Yield |
|---|---|
| With iron(III) chloride; sulfuric acid; bromine; iodine at 50 - 150℃; for 3h; |
-
-
110-89-4
piperidine

-
-
632-79-1
Tetrabromophthalic anhydride

-
-
80992-18-3
4,5,6,7-tetrabromo-3-hydroxy-3-piperidinophthalide

| Conditions | Yield |
|---|---|
| In ethyl acetate for 1h; Heating; | 99% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
35303-76-5
4-(2-aminoethyl)benzenesulfonamide

-
-
1454891-19-0
4-(2-(4,5,6,7-tetrabromo-1,3-dioxoisoindolin-2-yl)ethyl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With acetic acid at 130℃; for 1.5h; Inert atmosphere; | 99% |
| With acetic acid for 12h; Reflux; |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
56073-96-2
methyl N-[5(6)-(4-aminophenylsulfanyl)benzimidazol-2-yl]carbamate

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid at 40 - 45℃; for 3h; | 98% |
-
-
26782-71-8
D-tert-leucine

-
-
632-79-1
Tetrabromophthalic anhydride

| Conditions | Yield |
|---|---|
| With triethylamine In toluene Reflux; | 97% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
13810-83-8
tetrabromobenzene-1,2-dicarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: Tetrabromophthalic anhydride With potassium hydroxide In water for 0.5h; Inert atmosphere; Reflux; Stage #2: With hydrogenchloride In water at 20℃; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl acetamide at 55℃; for 3.5h; Temperature; Concentration; Reagent/catalyst; Solvent; | 95.6% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
56765-79-8
3,4-dicyanoaniline

| Conditions | Yield |
|---|---|
| In acetic acid for 2h; Reflux; | 95.5% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
24407-32-7
3,4,5,6-tetrabromophthalimide

| Conditions | Yield |
|---|---|
| With iron(III) chloride; guanidine hydrochloride; triethylamine at 60℃; for 0.0833333h; | 95% |
| With formamide In neat (no solvent) for 0.333333h; Reagent/catalyst; Milling; Heating; Green chemistry; | 94% |
| With choline chloride; urea at 140℃; for 1h; Green chemistry; | 92% |
| With potassium cyanate In N,N-dimethyl-formamide for 0.05h; microwave irradiation; | 89% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
35264-29-0
4-(hydrazinecarbonyl)benzene-1-sulfonamide

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid for 12h; Reflux; | 95% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
24313-88-0
3,4,5-Trimethoxyaniline

-
-
1293923-48-4
4,5,6,7-tetrabromo-2-(3,4,5-trimethoxyphenyl)isoindoline-1,3-dione

| Conditions | Yield |
|---|---|
| With acetic acid for 12h; Reflux; | 94% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
13755-95-8
2,3-diamino-1,4-naphthoquinone

-
-
1333335-11-7
1,2,3,4-tetrabromonaphtho[2',3':4,5]imidazo[2,1-a]isoindole-6,11,13-trione

| Conditions | Yield |
|---|---|
| With acetic acid In water at 20℃; Reflux; | 94% |

| Conditions | Yield |
|---|---|
| With acetic acid at 130℃; for 1.5h; Inert atmosphere; | 94% |
-
-
860994-58-7
1-((1R,2R)-2-aminocyclohexyl)-3-(3,5-bis(trifluoromethyl)phenyl)thiourea

-
-
632-79-1
Tetrabromophthalic anhydride

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 8h; Inert atmosphere; | 94% |
| With dmap In tetrahydrofuran at 20℃; for 2h; | |
| With triethylamine In tetrahydrofuran Inert atmosphere; Schlenk technique; |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In 1,2,3-trichlorobenzene at 100 - 195℃; for 3h; | 93.9% |


| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid for 12h; Reflux; | 93% |
-
-
110-89-4
piperidine

-
-
632-79-1
Tetrabromophthalic anhydride

-
-
80992-17-2
piperidinium 3,4,5,6-tetrabromo-2-piperidinocarbonylbenzoate

| Conditions | Yield |
|---|---|
| In benzene | 92% |

| Conditions | Yield |
|---|---|
| In toluene for 5h; Heating; | 92% |
| In toluene Condensation; Heating; | |
| In toluene for 3h; Condensation; Heating; Dean-Stark conditions; |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
1073606-09-3
C15H14BrN

-
-
107470-20-2
4,5,6,7-tetrabromo-2-(4-bromo-phenyl)-isoindoline-1,3-dione

| Conditions | Yield |
|---|---|
| In ethanol for 1h; Reflux; | 92% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
98-18-0
m-aminobenzenesulfonamide

-
-
1454891-20-3
3-(4,5,6,7-tetrabromo-1,3-dioxoisoindolin-2-yl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With acetic acid at 130℃; for 2h; Inert atmosphere; | 92% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
1954-94-5
4-amino-2-chlorobenzenesulfonamide

-
-
1454891-23-6
3-chloro-4-(4,5,6,7-tetrabromo-1,3-dioxoisoindolin-2-yl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With acetic acid at 130℃; for 12h; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| In diethyl ether | 91.09% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
63-74-1
sulfanilamide

-
-
1454891-17-8
4-(4,5,6,7-tetrabromo-1,3-dioxoisoindolin-2-yl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With acetic acid for 20h; Reflux; | 91% |
| With acetic acid at 130℃; for 12h; Inert atmosphere; | 75% |
-
-
223723-47-5
3,4-dihydro-6-hydroxy-2,5,7,8-tetramethyl-2H-benzopyran-2-carbohydrazide

-
-
632-79-1
Tetrabromophthalic anhydride

| Conditions | Yield |
|---|---|
| With acetic acid for 2h; Reflux; | 91% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
2368-83-4
4-amino-2-fluorobenzenesulfonamide

-
-
1454891-22-5
3-fluoro-4-(4,5,6,7-tetrabromo-1,3-dioxoisoindolin-2-yl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With acetic acid at 130℃; for 12h; Inert atmosphere; | 90% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
1094798-12-5
4-amino-2-bromobenzenesulfonamide

-
-
1454891-24-7
3-bromo-4-(4,5,6,7-tetrabromo-1,3-dioxoisoindolin-2-yl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With acetic acid at 130℃; for 12h; Inert atmosphere; | 90% |
-
-
632-79-1
Tetrabromophthalic anhydride


| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 8h; Inert atmosphere; | 90% |
-
-
632-79-1
Tetrabromophthalic anhydride

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 8h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With niobium pentachloride In methanesulfonic acid at 95 - 100℃; for 1.66667h; Friedel-Crafts Acylation; Inert atmosphere; | 90% |
| With Zn0.95*Ti0.05O In neat (no solvent) at 160℃; for 4h; Green chemistry; | 66% |
-
-
632-79-1
Tetrabromophthalic anhydride

-
-
24544-04-5
2,6-diisopropylbenzenamine

| Conditions | Yield |
|---|---|
| With acetic acid at 140℃; for 12h; | 90% |
Tetrabromophthalic anhydride Chemical Properties
The Molecular Weight of 3,4,5,6-TETRABROMOPHTHALIC ANHYDRIDE(632-79-1): 463.7
Molecular Structure :
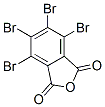
EINECS: 211-185-4
Index of Refraction: 1.725
Molar Refractivity: 66.44 cm3
Molar Volume: 167.3 cm3
Polarizability: 26.33 10-24 cm3
Surface Tension: 72 dyne/cm
Density: 2.771 g/cm3
Boiling Point: 540.5 °C at 760 mmHg
Flash Point: 280.7 °C
Enthalpy of Vaporization: 81.81 kJ/mol
Vapour Pressure: 9.54E-12 mmHg at 25°C
Water Solubility: hydrolysis
IUPAC Name: 4,5,6,7-tetrabromo-2-benzofuran-1,3-dione
Synonyms: 1,3-Isobenzofurandione, 4,5,6,7-tetrabromo-;1,3-Isobenzofurandione,4,5,6,7-tetrabromo-;4,5,6,7-Tetrabromo-2-benzofuran-1,3-dione;4,5,6,7-tetrabromo-3-isobenzofurandione;bromophthal;3,4,5,6-Tetrabromophthalic anhydride;3,4,5,6-tetrabromophthalicanhydride;3-Isobenzofurandione,4,5,6,7-tetrabromo-1;
Tetrabromophthalic anhydride Toxicity Data With Reference
| 1. | eye-rbt 100 mg MLD | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0523271 . | ||
| 2. | orl-rat LD50:>10 g/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0539755 . | ||
| 3. | ihl-rat LC :>10,920 mg/m3/4H | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0522251 . | ||
| 4. | skn-rbt LD50:>10 g/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0522251 . | ||
| 5. | skn-gpg LD50:>1 g/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0521611 . |
Tetrabromophthalic anhydride Safety Profile
The Hazard Codes of 3,4,5,6-TETRABROMOPHTHALIC ANHYDRIDE(632-79-1):
 Xi
Xi HS Code: 29173911
The Risk Statements information of 3,4,5,6-TETRABROMOPHTHALIC ANHYDRIDE(632-79-1):
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements information of 3,4,5,6-TETRABROMOPHTHALIC ANHYDRIDE(632-79-1):
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
24/25: Avoid contact with skin and eyes
37/39: Wear suitable gloves and eye/face protection
WGK Germany: 2
RTECS: TI3350000
Related Products
- Tetrabromophthalic anhydride
- 63279-36-7
- 6328-00-3
- 632-80-4
- 6328-12-7
- 6328-29-6
- 63283-36-3
- 6328-36-5
- 632-83-7
- 6328-37-6
- 63284-71-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View