-
Name
trans-clomifene citrate
- EINECS
- CAS No. 7599-79-3
- Article Data3
- CAS DataBase
- Density g/cm3
- Solubility
- Melting Point 143-145°C
- Formula C26H28ClNO•C6H8O7
- Boiling Point 509°Cat760mmHg
- Molecular Weight 598.093
- Flash Point 261.6°C
- Transport Information
- Appearance
- Safety An experimental teratogen. Other experimental reproductive effects. When heated to decomposition it emits very toxic fumes of Cl− and NOx.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms
- PSA 144.60000
- LogP 5.31410
Synthetic route
Conditions

| Conditions | Yield |
|---|---|
| Stage #1: ethanamine, 2-[4-[(1E)-2-chloro-1,2-diphenyl ethenyl]phenoxy]-N,N-diethyl-, (±)-1,1‘-binaphthyl-2,2’-diylhydrogenphosphate With sodium hydroxide In water; toluene at 20℃; for 2h; Stage #2: citric acid In acetone at 0 - 50℃; for 2h; | 97.07 g |
| Stage #1: ethanamine, 2-[4-[(1E)-2-chloro-1,2-diphenyl ethenyl]phenoxy]-N,N-diethyl-, (±)-1,1‘-binaphthyl-2,2’-diylhydrogenphosphate With ammonia In ethyl acetate Stage #2: citric acid In ethanol; ethyl acetate at 20℃; for 1h; | |
| Stage #1: ethanamine, 2-[4-[(1E)-2-chloro-1,2-diphenyl ethenyl]phenoxy]-N,N-diethyl-, (±)-1,1‘-binaphthyl-2,2’-diylhydrogenphosphate With ammonium hydroxide In tert-butyl methyl ether; water; isopropyl alcohol for 1h; Stage #2: citric acid In ethanol; water at 60 - 65℃; | 269 g |

-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: hydrogenchloride / toluene / Dean-Stark; Reflux 2.1: acetic acid; 1,3-dichloro-5,5-dimethylhydantoin / toluene / 2 h / 60 °C 2.2: 4.5 h / 0 °C / Heating 3.1: methanol / 1 h / 20 - 45 °C 4.1: sodium hydroxide / toluene; water / 2 h / 20 °C 4.2: 2 h / 0 - 50 °C View Scheme |
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: methanol / 1 h / 20 - 45 °C 2.1: sodium hydroxide / toluene; water / 2 h / 20 °C 2.2: 2 h / 0 - 50 °C View Scheme |
-
-
19957-52-9
2-{4-(1,2-diphenylethenyl)phenoxy}-N,N-diethylethanamine hydrochloride

-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: acetic acid; 1,3-dichloro-5,5-dimethylhydantoin / toluene / 2 h / 60 °C 1.2: 4.5 h / 0 °C / Heating 2.1: methanol / 1 h / 20 - 45 °C 3.1: sodium hydroxide / toluene; water / 2 h / 20 °C 3.2: 2 h / 0 - 50 °C View Scheme |
-
-
73404-00-9
1-{4-[2-(diethylamino)ethoxy]phenyl}-1,2-diphenylethanol

-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sulfuric acid / dichloromethane / 1 h / 0 - 20 °C 2.1: N-chloro-succinimide / dichloromethane / 32 h / 20 °C 2.2: 0.5 h / 20 °C / pH 8 - 9 3.1: methanol / 2 h / 20 °C 4.1: ammonia / ethyl acetate 4.2: 1 h / 20 °C View Scheme |
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: methanol / 2 h / 20 °C 2.1: ammonia / ethyl acetate 2.2: 1 h / 20 °C View Scheme |

-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: N-chloro-succinimide / dichloromethane / 32 h / 20 °C 1.2: 0.5 h / 20 °C / pH 8 - 9 2.1: methanol / 2 h / 20 °C 3.1: ammonia / ethyl acetate 3.2: 1 h / 20 °C View Scheme |
Conditions

| Conditions | Yield |
|---|---|
| In ethanol; water at 65℃; Solvent; | 20.2 g |
-
-
7599-79-3
clomiphene citrate

-
-
911-45-5, 15690-55-8, 15690-57-0
clomiphene
Conditions

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethyl acetate for 0.5h; | 99% |
-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: sodium hydroxide / ethyl acetate / 0.5 h 2.1: tert.-butyl lithium / pentane; tetrahydrofuran / 18.5 h / -40 - 20 °C 3.1: tetrabutylammomium bromide; sodium hydroxide 3.2: 15 h / 20 °C View Scheme |
-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sodium hydroxide / ethyl acetate / 0.5 h 2.1: tert.-butyl lithium / pentane; tetrahydrofuran / 18.5 h / -40 - 20 °C 3.1: tetrabutylammomium bromide; sodium hydroxide 3.2: 15 h / 20 °C 4.1: toluene / 16 h / 100 °C View Scheme |
-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sodium hydroxide / ethyl acetate / 0.5 h 2.1: tert.-butyl lithium / pentane; tetrahydrofuran / 18.5 h / -40 - 20 °C 3.1: tetrabutylammomium bromide; sodium hydroxide 3.2: 15 h / 20 °C 4.1: toluene / 100 °C View Scheme |
-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: sodium hydroxide / ethyl acetate / 0.5 h 2.1: tert.-butyl lithium / pentane; tetrahydrofuran / 18.5 h / -40 - 20 °C 3.1: tetrabutylammomium bromide; sodium hydroxide 3.2: 15 h / 20 °C 4.1: toluene / 100 °C 5.1: acetonitrile View Scheme |
-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: sodium hydroxide / ethyl acetate / 0.5 h 2.1: tert.-butyl lithium / pentane; tetrahydrofuran / 18.5 h / -40 - 20 °C 3.1: tetrabutylammomium bromide; sodium hydroxide 3.2: 15 h / 20 °C 4.1: toluene / 100 °C 5.1: acetonitrile 6.1: hydrogenchloride; triethylsilane / water; methanol / 4 h / 20 °C View Scheme |
-
-
7599-79-3
clomiphene citrate
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: sodium hydroxide / ethyl acetate / 0.5 h 2.1: tert.-butyl lithium / pentane; tetrahydrofuran / 18.5 h / -40 - 20 °C 3.1: tetrabutylammomium bromide; sodium hydroxide 3.2: 15 h / 20 °C 4.1: toluene / 16 h / 100 °C 5.1: hydrogenchloride / water / pH 5 - 7 View Scheme |
-
-
7599-79-3
clomiphene citrate

-
-
133157-86-5
(Z)-1-<4-(2-diethylaminoethoxy)phenyl>-1,2-diphenyl-1-penten-5-ol
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / ethyl acetate / 0.5 h 2: tert.-butyl lithium / pentane; tetrahydrofuran / 18.5 h / -40 - 20 °C View Scheme |
trans-CLOMIPHENE CITRATE Chemical Properties
The molecular formula of trans-CLOMIPHENE CITRATE (7599-79-3) is C32H36ClNO8 and its formula weight is 598.14 g/mol.The boiling point is 509°C at 760mmHg.Enthalpy of vaporization is 77.94 kJ/mol.Its flash point is 261.6°C and the vapor pressenge is 1.77E-10 mmHg at 25°C .The chemical synonyms of trans-CLOMIPHENE CITRATE (7599-79-3) are trans-clomifene citrate .The molecular structure of trans-CLOMIPHENE CITRATE (7599-79-3) is 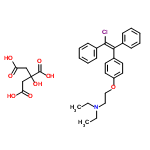 .
.
 .
.trans-CLOMIPHENE CITRATE Safety Profile
An experimental teratogen. Other experimental reproductive effects. When heated to decomposition it emits very toxic fumes of Cl− and NOx.
trans-CLOMIPHENE CITRATE Specification
Its extinguishing agent is sand, dry powder and foam.
Related Products
- trans-CLOMIPHENE CITRATE
- 75998-57-1
- 76003-29-7
- 7600-50-2
- 76005-99-7
- 76006-04-7
- 76006-07-0
- 76006-08-1
- 76006-11-6
- 76006-13-8
- 76006-33-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
