Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:15243-33-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryWuhan Fortuna Chemical Co.,Ltd
Unique advantages for Ruthenium Carbonyl Cas 15243-33-1 Guaranteed purity High quality & competitive price Quality control Fast feedback Prompt shipment Appearance:Orange powder Storage:N/A Package:100g,1kg/foil bag Application:Cata
Cas:15243-33-1
Min.Order:100 Gram
FOB Price: $10.0 / 15.0
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
high purity Storage:Sealed, dry, microtherm , avoid light and smell. Package:meet customer's demand Application:organic synthesis Transportation:by sea or by air Port:shanghai
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:15243-33-1
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:15243-33-1
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:15243-33-1
Min.Order:1 Kilogram
FOB Price: $1300.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:15243-33-1
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:15243-33-1
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Cas:15243-33-1
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China and Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED intermediat
Cas:15243-33-1
Min.Order:1 Gram
FOB Price: $4.0 / 5.0
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:15243-33-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
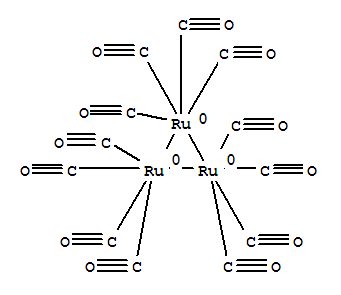
Product Name: RUTHENIUM CARBONYL MF: C12O12Ru3 MW: 639.33 EINECS: 239-287-4 Mol File: 15243-33-1.mol RUTHENIUM CARBONYL Structure RUTHENIUM CARBONYL Chemical Properties Melting point 150 °C storage temp. Inert atmosphere,
Cas:15243-33-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
RUTHENIUM CARBONYL CAS:15243-33-1 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic interm
Cas:15243-33-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:15243-33-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
We provide tailor-made photoinitiator solutions for different performances by custom synthesisi.blends or other technical supports low-odor uv formulations non-yellowing uv formulations benzene-free uv formulations polymeric uv formul
Cas:15243-33-1
Min.Order:100 Kilogram
Negotiable
Type:Trading Company
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:15243-33-1
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:15243-33-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChemlyte Solutions
Chemlyte Solutions believe that customers and suppliers deserve much more than what traditional distributors can offer. To grow in today s fast-paced and increasingly competitive market it is essential to be able to quickly adapt to market forces eff
Cas:15243-33-1
Min.Order:100 Gram
Negotiable
Type:Other
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:15243-33-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquirySHANGHAI SYSTEAM BIOCHEM CO., LTD
We are one of a few suppliers that can offer custom synthesis service of this product We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to prov
Cas:15243-33-1
Min.Order:100 Gram
FOB Price: $100.0 / 2000.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:15243-33-1
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:15243-33-1
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:15243-33-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
Cas:15243-33-1
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:15243-33-1
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Suzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Cas:15243-33-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Cas:15243-33-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
16406-48-7
ruthenium pentacarbonyl

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In further solvent(s) byproducts: CO; exposure to an N2-atmosphere, hydrocarbon soln.;; | 100% |
| In pentane storing of soln. for 1 day under Ar at room temp.; | |
| In neat (no solvent) decompn. of (Ru(CO)5) at room temp.;; |

-
-
201230-82-2
carbon monoxide

-
-
21907-50-6
cesium trifluoroacetate

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
184584-51-8
fac-Cs[Ru(CF3COO)3(CO)3]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran CO-atmosphere; stirring Ru-complex soln. (30°C, 8 h); sepn. of Ru3(CO)12, evapn.; | A 95% B 99% |


-
-
74-85-1
ethene

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With CO In hexane under N2; Ru compd. is dissolved in hexane, transferred to a Hoke cylinder, charged with CO (1 atm) and ethylene (3 atm), allowed to sit at room temp. for 20 h; gases are vented, IR, mixt. is reduced in volume, cooled to -20°C: crystn. of Ru3(CO)12, soln. is filtered after 12 h, reduced in volume to half, IR, soln. is cooled to -20°C, supernatant liq. is similar worked up; elem. anal.; | A n/a B 98% |


-
-
201230-82-2
carbon monoxide

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In 2-methoxy-ethanol byproducts: CO2, KCl; heating the Ru complex and CO in 2-methoxyethanol to 85°C for 20 min; filtration, washing (EtOH); | 97% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With carbon monoxide In methanol; dichloromethane; water High Pressure; dissolving Ru2(CH3COO)4Cl in CH3OH-CH2Cl2-H2O (10:10:1), charging into pressure vessel, pressurizing to 1000 psi with CO, stirring at this pressure for 4 h; venting CO atmosphere, partially removing solvent by vac. distn., pptn.filtration; | 96% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With carbon monoxide In methanol CO atmosphere (room temp., 10 min, stirring), pptn.; redn. of vol. (vac.), filtration off, washing (MeOH): 87.7%; evapn. of filtrate to dryness, chromy. (silica, hexane): 7.9%; | 95.7% |
-
-
52621-15-5
[Ru(CO)4(η2-ethylene)]

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In n-heptane N2-atmosphere; refluxing for 2 h; solvent removal (vac.), TLC (CH2Cl2/hexane=1:1); | 95% |

-
-
201230-82-2
carbon monoxide

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With sodium carbonate In ethylene glycol molar ratio Ru:Na2CO3=1:1; bubbling CO for 5 h at 95°C; extn. of ppt. and sublimate with CH2Cl2 (room temp.), evapn., washing (water); | 93% |
-
-
74-85-1
ethene

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With CO In hexane under N2; Ru compd. is dissolved in hexane, transferred to a Hoke cylinder, charged with CO (1 atm) and ethylene (3 atm), allowed to sit at room temp. for 20 h; gases are vented, IR, mixt. is reduced in volume, cooled to -20°C: crystn. of Ru3(CO)12, soln. is filtered after 12 h, reduced in volume to half, IR, soln. is cooled to -20°C, supernatant liq. is similar worked up; elem. anal.; | A n/a B 93% |


| Conditions | Yield |
|---|---|
| With triethylamine In propan-1-ol; water at 85℃; under 2625.26 Torr; Concentration; Reagent/catalyst; Solvent; Temperature; Pressure; Autoclave; | 92.5% |
| In methanol at 125℃; under 48754.9 Torr; for 8h; Autoclave; | |
| With triethylamine In propan-1-ol at 85℃; under 2625.26 Torr; for 17h; Autoclave; Gas phase; |

-
-
27081-10-3
tropylium tetrafluoroborate

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With carbon monoxide In not given dropwise addn. of CO and Ru-cluster soln. (CH2Cl2 or MeCN) to soln. of (C7H7)BF4 (N2 purge, room temp.); IR monitoring; | 92% |

-
-
201230-82-2
carbon monoxide

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With sodium carbonate In ethylene glycol bubbling of CO (1 atm) through a soln. of Ru salt in ethylene glycol for5 min, heating under CO at 110°C for 2 h, cooling to 25°C , addn. of Na2CO3 (Na2CO3:Ru mol. ratio 3:2), heating under CO at 80°C for 7 h; extn. of sublimate and reaction mixt. with dichloromethane at room temp., evapn. to dryness, washing the solid with water and then pentane; | 91% |
| With potassium hydroxide In 2-ethoxy-ethanol under CO (1 atm), 1 h at 80°C, 45 min at 135°C, then addedKOH (2 equiv.) at 75°C under CO bubbling; | 90% |
| With Fe or stainless steel or Fe-Cr-alloy In ethanol byproducts: RuO2, [RuCl2(CO)3]2; High Pressure; by a react. of Ru-contg. compd. in EtOH soln. at high CO pressure (50 bar) in autoclave in the presence of Fe plate or stainless steel (76% Fe, 17% Cr, 7% Ni) or Fe-Cr alloy (72.8% Fe, 22% Cr, 5% Al, 0.1% Y, 0.1 Zr);at 125°C; react. time: > 3 h; cooling in an ice bath; solid was filtered off and the resulting soln. was evapd. to dryness; | 80% |

-
-
74-85-1
ethene

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With CO In hexane under N2; Ru compd. is dissolved in hexane, transferred to a Hoke cylinder, charged with CO (1 atm) and ethylene (3 atm), allowed to sit at room temp. for 20 h; gases are vented, IR, mixt. is reduced in volume, cooled to -20°C: crystn. of Ru3(CO)12, soln. is filtered after 12 h, reduced in volume to half, IR, soln. is cooled to -20°C, supernatant liq. is similar worked up; elem. anal.; | A 27% B 91% |

-
-
80800-53-9
HRu3(CO)10(μ-η2-C(O)CH3)

-
-
74-85-1
ethene

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With CO In hexane under N2; Ru compd. is dissolved in hexane, transferred to a Hoke cylinder, charged with CO (1 atm) and ethylene (3 atm), allowed to sit at room temp. for 20 h; gases are vented, IR, mixt. is reduced in volume, cooled to -20°C: crystn. of Ru3(CO)12, soln. is filtered after 12 h, reduced in volume to half, IR, soln. is cooled to -20°C, supernatant liq. is similar worked up; elem. anal.; | A n/a B 91% |

-
-
201230-82-2
carbon monoxide

-
-
10049-08-8
ruthenium(III)chloride

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With caesium carbonate In methanol other Radiation; mixt. of RuCl3, Cs2CO3 and MeOH was sealed in microwave app.; CO (50 psi) was introduced; sealed; heated with stirring to 110°C using power of 300 W; held for 10 min; cooled to 50°C; evapd. (vac.); hexane added; heated; solvent removed; | 90% |
| With Cu or Zn In ethanol other Radiation; γ irradiation of 1E-3 mol/l RuCl3 in ethanol under CO atmosphere at room temp. in presence of Cu or Zn powder; | |
| With zinc In methanol heating RuCl3 with Zn at a CO pressure of 5-10 atm at 65 °C for 30 h;; extraction of thr solid residue of the filtration; pptn. on concentrating;; | |
| With Zn In methanol heating RuCl3 with Zn at a CO pressure of 5-10 atm at 65 °C for 30 h;; extraction of thr solid residue of the filtration; pptn. on concentrating;; |
-
-
52621-15-5
[Ru(CO)4(η2-ethylene)]

-
-
201230-82-2
carbon monoxide

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In n-heptane CO-atmosphere; refluxing for 2 h; TLC (CH2Cl2/hexane=1:1); other unidentified products; | 90% |

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In dichloromethane-d2 thermal decompn. in soln. at room temp. within 15 min; | A 90% B n/a C n/a |


-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
B
-
105121-22-0
ruthenium3(carbonyl)9(μ3-carbonyl)(μ3-sulfido)

| Conditions | Yield |
|---|---|
| With CO In hexane refluxing (30 min, bubbling CO); crystn., solvent removal (vac.), dissoln. in CH2Cl2, thin layer chromy.(SiO2, hexane); elem. anal.; | A n/a B 89% |
-
-
71562-47-5
H3Ru3(μ3-methoxymethylidyne)(carbonyl)9

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
16406-48-7
ruthenium pentacarbonyl

-
C
-
115-10-6
Dimethyl ether

| Conditions | Yield |
|---|---|
| With carbon monoxide; hydrogen In toluene an autoclave containing a soln. of Ru3-cluster in toluene was pressurized to 500 psig with 1:1 CO-H2 and was heated at 130°C for 23 h; cooled, gases were vented through U-trap (liq. N2), condensate was shown to be Me2O and Ru(CO)5 by mass spectrometry, toluene soln. was filtered (ppt. - Ru3(CO)12 identified by IR data), filtrate evapd., residue chromd. on SiO2 to give addnl. Ru3(CO)12; | A 89% B <1 C n/a |

-
-
140111-19-9
Ru3(CO)10H(OCC6H4N(CH3)2)

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
B
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With CO In benzene High Pressure; heating (3 bar CO, 90 min, 80°C); chromy. (hexane); | A 88% B 81% |

-
-
201230-82-2
carbon monoxide

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With KOH In methanol; 2-methoxy-ethanol byproducts: KCl; heating RuCl3*3H2O and CO in 2-methoxyethanol to 125°C for ca. 2 h, after cooling to 20°C addn. of methanolic KOH, heating to 85°C for 20 min; filtration; | 86% |
| In methanol High Pressure; a soln. of RuCl3 in methanol was charged with 2.0 MPa CO and pressurizedto 5.0 MPa, the autoclave was placed in a thermostatic oil bath with st irring for 8 h at 125°C, cooled to room temp.; filtered, recrystd. from toluene; | 54% |
-
-
67-56-1
methanol

-
-
131145-73-8
bis(dimethylgermyl)methane ruthenium tetracarbonyl

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
B
-
131245-48-2
bis(dimethylmethoxygermyl)methane

| Conditions | Yield |
|---|---|
| the Ru complex reacts with an excess of methanol (6 h 80°C); fractional distn.; elem. anal.; | A n/a B 85% |

-
-
128391-81-1
{HRu3(CO)9(μ3-η2:η2:η2-C6H6)}BF4

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
B
-
128363-71-3
Ru3(CO)9(μ3:η(2):η(2):η(2)-C6H6)

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo{5.4.0}undec-7-ene In dichloromethane (N2); added dropwise a soln. of N-compound to soln. of Ru-complex; filtered; evapd. (vac.); chromy.; | A n/a B 85% |

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In toluene byproducts: H2; stirred at 80°C for 1 h; solvent removed under vacuum, chromd. (silica gel, hexane), solvent removed under vacuum from the 2. fraction, crystd. at -30°C; | A n/a B 83% |
-
-
131145-74-9
bis(dimethylgermyl)ethane ruthenium tetracarbonyl

-
-
586-96-9
Nitrosobenzene

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
B
-
120926-63-8
(CH3)2GeCH2CH2Ge(CH3)2O

-
C
-
1227476-15-4
Azobenzene

| Conditions | Yield |
|---|---|
| In benzene byproducts: CO; the Ru complex reacts in benzene with C6H5NO in a closed tube at 140°C (48 h); | A n/a B 82% C n/a |

-
-
98976-61-5
Ru4H3(CO)10C6H5PCH2P(C6H5)2

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| With CO In toluene CO was bubbled through a toluene soln. of complex heated under reflux for 3 h, the soln. was cooled; chromy. on silica gel, eluent heptane-toluene; | A 18.7% B 80.5% |

-
-
201230-82-2
carbon monoxide

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In toluene stainless steel autoclave; pressurizing with 20 atm CO, heating to 160°C for 2 h (stirring); cooling, collection (filtration), washing (cold PhMe); | 80% |
| In further solvent(s) stainless steel autoclave, PhNH2 solvent; pressurizing with 5 atm CO, heating to 160°C for 2 h (stirring); cooling, collection (filtration), washing (cold PhMe); | 70% |

-
-
597-52-4
Triethylsilanol

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In acetonitrile N2 atmosphere; stirring (24 h, room temp.); | 80% |
| In neat (no solvent) N2 atmosphere; 70°C; |
-
-
131145-73-8
bis(dimethylgermyl)methane ruthenium tetracarbonyl

-
-
624-65-7
2-propynyl chloride

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
B
-
98187-50-9
bis(chlorodimethylgermyl)methane

| Conditions | Yield |
|---|---|
| In benzene HCCCH2Cl reacts with the tetracarbonyl ruthenium complex (80°C, 4 h, inert conditions); identification by spectroscopy; | A n/a B 80% |


-
-
5112-95-8
bis(diphenylphosphino)acetylene

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

| Conditions | Yield |
|---|---|
| In dichloromethane react. under dry N2 using Schlenk and vac.-line techniques; addn. of 1.0 equiv. of the acetylene to Ru-complex in CH2Cl2 with stirring (orange soln.), react. for 1 h at room temp.; TLC (Kiesegel, eluent CH2Cl2-hexane 2:3), orange band, slow evapn. of a CH2Cl2-hexane soln.; elem. anal.; | A 20% B 80% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
201230-82-2
carbon monoxide

-
-
16406-48-7
ruthenium pentacarbonyl

| Conditions | Yield |
|---|---|
| In hexane Irradiation (UV/VIS); photolysis (>376nm, filtered 250 W high pressure mercury arc) in n-hexane (ca. 6ml of a 1E-4M soln.) under ca. 600Torr CO for 3h;; monitoring of react. by IR;; | 100% |
| In tetrahydrofuran High Pressure; 80 bar CO, autoclave; 150°C (5 h); | 73% |
| In hexane for 3h; Inert atmosphere; Schlenk technique; UV-irradiation; |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
118734-49-9
4,5-diethyl-2,5-dihydro-3-isopropenyl-2,2-dimethyl-1,2,5-selenasilaborole

-
A
-
118772-38-6
tricarbonyl(η4-4,5-diethyl-2,5-dihydro-3-isopropenyl-2,2-dimethyl-1,2,5-selenasilaborole)ruthenium

-
B
-
201230-82-2
carbon monoxide

| Conditions | Yield |
|---|---|
| In 1,3,5-trimethyl-benzene Ar atmosphere, heating (140°C, 4 h); filtn., solvent removal (0.001 Torr), sublimation (75-80°C, 0.001 Torr); elem. anal.; | A 82% B 100% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
63995-70-0
trisodium tris(3-sulfophenyl)phosphine

| Conditions | Yield |
|---|---|
| In methanol N2-atmosphere, molar ratio = 1 : 1; refluxing (2 h); evapn. (reduced pressure), washing (hexane), drying (vac.); | 100% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
63995-70-0
trisodium tris(3-sulfophenyl)phosphine

| Conditions | Yield |
|---|---|
| In methanol N2-atmosphere, molar ratio = 1 : 2; refluxing (2 h); evapn. (reduced pressure), washing (hexane), drying (vac.); | 100% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
63995-70-0
trisodium tris(3-sulfophenyl)phosphine

| Conditions | Yield |
|---|---|
| In methanol N2-atmosphere, molar ratio = 1 : 3; refluxing (2 h); evapn. (reduced pressure), washing (hexane), drying (vac.); | 100% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
14243-64-2
(triphenylphosphine)gold(I) chloride

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| With (CH3)3NO In dichloromethane; acetonitrile (N2), soln. of Me3NO in CH3CN added dropwise to soln. of Ru complex in CH2Cl2 at -78°C over 15 min, slowly warmed to room temp., reacted for 15 min, treated with Au complex, stirred at room temp. for 1.5 h; evapd.(vac.), elem. anal.; | 100% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
110452-48-7
5,10,15,20-tetrakis(p-tert-butylphenyl)porphyrin

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| Stage #1: dodecacarbonyl-triangulo-triruthenium; 5,10,15,20-tetrakis(p-tert-butylphenyl)porphyrin In 1,2-dichloro-benzene at 160℃; for 12h; Inert atmosphere; Stage #2: acetonitrile In 1,2-dichloro-benzene at 80℃; for 12h; Inert atmosphere; | 100% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
141-82-2
malonic acid

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; benzene byproducts: CO; Ru3(CO)12 and malonic acid were refluxed in benzene/dioxane under a slow flow of N2 for 24 h;; the solid was filtered off from the hot suspension, washed with dioxaneand diethyl ether, and dried under vac.; elem. anal.;; | 99.4% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
16406-48-7
ruthenium pentacarbonyl

| Conditions | Yield |
|---|---|
| With CO In n-heptane 133 atm CO, 100°C, 24 h; | 99% |
| With carbon monoxide In diethylene glycol Irradiation (UV/VIS); 25°C, 1 atm of CO; UV monitoring; not isolated, detected by IR; | |
| With carbon monoxide In octane Irradiation (UV/VIS); 25°C, 1 atm of CO; UV monitoring; not isolated, detected by IR; |
-
-
110-86-1
pyridine

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
76-05-1
trifluoroacetic acid

-
-
166821-97-2, 167073-80-5
RuH(CF3CO2)(CO)2(C5H5N)2

| Conditions | Yield |
|---|---|
| In pyridine byproducts: CO; 95°C, 10 min; evapn. (reduced pressure), dissoln. in EtOH, Et2O addn., crystn. (0°C); isomer mixt. not sepd.; | 99% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
58201-22-2
[1,2-bis(diphenylphosphino)ethane]tricarbonylruthenium

| Conditions | Yield |
|---|---|
| With carbon monoxide; 1,2-bis-(diphenylphosphino)ethane In benzene equimolar amts., 100 atm CO, 100°C, 4-5 h; crystn. (< 0°C, 4-5 h), washing (C6H6, light petroleum), drying (vac.); elem. anal.; | 99% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
75-05-8
acetonitrile

-
-
103257-53-0
Ru3(CO)10(acetonitrile)2

| Conditions | Yield |
|---|---|
| With trimethylamine-N-oxide In dichloromethane; acetonitrile Ar-atmosphere; dropwise addn. of slight excess Me3NO to Ru-complex soln.(in CH2Cl2/MeCN=10:1) at -78°C, warming to room temp.; evapn. (reduced pressure); elem. anal.; | 99% |
| With trimethylamine-N-oxide In dichloromethane soln. of Me3NO added to a soln. of Ru-compd. and CH3CN at 0°C about 15 min, stirred for 1 h; filtered about silica gel, evapd.; | |
| In dichloromethane filtration (degassed SiO2), the soln. kept at -10°C; | |
| With (CH3)3NO In dichloromethane |

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide (Ar); react. metal carbonyl with Na in HMPA for 1 h at 20°C; | 99% |
| In ammonia NH3 (liquid); NH3 distilled onto Na/Ru mixture (acetone/dry ice bath) under vac., slowly warmed to 235 K, vigorously stirred for ca. 60 min, warmed to 260 K; dried (vac., 260 K, 3.5 h); |

| Conditions | Yield |
|---|---|
| In octane byproducts: CO; High Pressure; n-octane added to Ru complex, degassed (vac.), heated at 120°C (bath) under 5 atm (2)H2 for 9 h with intermediate blowings with N2; allowed to cool to room temp. overnight, filtered, dried (vac.); | 99% |
| In not given according to Knox et al., J. Am. Chem. Soc., 97 (1975) 3942; Bruce et al., Inorg. Synth., 26 (1989) 262; |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
110-15-6
succinic acid

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; toluene byproducts: CO; Ru3(CO)12 and succinic acid were refluxed in toluene/dioxane under a slow flow of N2 for 24 h;; the solid was filtered off from the hot suspension, washed with acetoneand diethyl ether, and dried under vac.; elem. anal.;; | 99% |

| Conditions | Yield |
|---|---|
| With trimethylamine-N-oxide In dichloromethane; acetonitrile Ar-atmosphere; dropwise addn. of excess Me3NO to Ru-complex soln. (in CH2Cl2/MeCN=2:5) at -78°C, warming to room temp.; crystn. on slow removal of solvent (reduced pressure); elem. anal.; | 99% |
-
-
110-65-6
1,4-dihydroxybut-2-yne

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
21050-13-5
bis(triphenylphosphine)iminium chloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Ru3(CO)12 and (PPN)Cl (molar ratio 1:1) dissolved in THF; stirred at room temp. for 3 h under N2 bubbling; monitored by IR spectra; ligand (1 equiv.) added; a few min; evapd.; | 99% |

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
76858-94-1
1,2-bis(di(pentafluorophenyl)phosphino)ethane

-
-
225779-93-1, 225779-94-2
Ru3(CO)10(1,2-bis(bis-pentafluorophenylphosphino)ethane)

| Conditions | Yield |
|---|---|
| With catalyst:Ph2CO(1-) In tetrahydrofuran catalyst (8 drops) added to THF soln. of Ru compd. (0.078 mmol), ligand (0.086 mmol) added, stirred, catalyst (8-10 drops added), TLC conrol; | 99% |
| With sodium diphenylketyl In tetrahydrofuran under argon, Schlenk setup, 1:2 mixt., heated at 40°C, 5 drops ofsodium diphenylketyl soln. added, stirred (IR control); evapd., dissolved (CH2Cl2), chromd. (silica, C6H12/CH2Cl2), recrystd. (CH2Cl2), elem. anal.; | 77% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
172987-61-0
Ph2PC*C-C*CBu(t)

-
-
163727-43-3
[Ru3(CO)11(P(C6H5)2CCCCC(CH3)3)]

| Conditions | Yield |
|---|---|
| With sodium benzophenone ketyl In tetrahydrofuran | 99% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
7608-31-3
Ph2PC*C-C*CPh

-
-
163727-44-4
[Ru3(CO)11(P(C6H5)2CCCCC6H5)]

| Conditions | Yield |
|---|---|
| With sodium benzophenone ketyl In tetrahydrofuran | 99% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
14857-34-2
dimethyl(ethoxy)silane

| Conditions | Yield |
|---|---|
| In hexane Irradiation (UV/VIS); (N2); irradiation (180 W mercury lamp, 25°C, 2 h); prepd. as soln. (spectr. yield); | 99% |
| In hexane Irradiation (UV/VIS); (N2); reflux (1.5 h); concn., TLC (SiO2; CH2Cl2/hexane); | 30% |

| Conditions | Yield |
|---|---|
| In octane byproducts: CO; High Pressure; n-octane added to Ru complex, degassed (vac.), heated at 120°C (bath) under 5 atm H2 for 9 h with intermediate blowings with N2; allowed to cool to room temp. overnight, filtered, dried (vac.); | 99% |
-
-
37083-37-7
5,10,15,20-tetrakis(2,6-dichlorophenyl)porphyrin

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
127390-19-6
[Ru(2,6-Cl2tpp)CO]

| Conditions | Yield |
|---|---|
| In 1,2,4-trichorobenzene Reflux; Inert atmosphere; | 99% |
-
-
37083-37-7
5,10,15,20-tetrakis(2,6-dichlorophenyl)porphyrin

-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
127390-19-6
carbonyl ruthenium(II) 5,10,15,20-tetra(2,6-dichlorophenyl)porphyrin

| Conditions | Yield |
|---|---|
| In 1,2,4-Trichlorobenzene at 195 - 200℃; Inert atmosphere; | 99% |
| With 1,2,4-Trichlorobenzene at 160 - 165℃; Inert atmosphere; |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
59863-12-6
bis[bis(trimethylsilyl)amino]germanium(II)

-
-
1310738-35-2
[Ru(Ge(HMDS)2)2(CO)3]

| Conditions | Yield |
|---|---|
| In toluene (N2) Ge(HMDS)2 was added to suspn. (Ru3(CO)12) in toluene and heated at 100°C for 1 h; solvent was removed in vacuo; | 99% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
1396690-42-8
2-(2-methylprop-1-en-1-yl)phenyl formate

-
-
4374-68-9
2-oxo-3-isopropyl-2,3-dihydrobenzofuran

| Conditions | Yield |
|---|---|
| With 1-(1-phenylethyl)-1H-imidazole In 1,3,5-trimethyl-benzene at 135℃; for 24h; Inert atmosphere; Sealed tube; regioselective reaction; | 99% |
-
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
-
1396690-44-0
2-(cyclopent-1-en-1-yl)phenyl formate

| Conditions | Yield |
|---|---|
| With 2,4,5-trimethyl-1-phenyl-1H-imidazole In 1,3,5-trimethyl-benzene at 135℃; for 24h; Inert atmosphere; Sealed tube; Overall yield = 75 %; regioselective reaction; | A n/a B 99% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 20h; Inert atmosphere; Reflux; | 99% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 F,
F,  Xn
Xn