Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:480-47-7
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:480-47-7
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:480-47-7
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:480-47-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:480-47-7
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:480-47-7
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Hangzhou Dingyan Chem is a leading manufacturer and supplier of chemicals in China.We develop,produce and distribute high quality pharmaceuticals, intermediates, special chemicals and other fine chemicals. We could give you: 1.Best quality in your re
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:480-47-7
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Antimex Chemical Limied
hight degree of purity Application:Fine chemical intermediates, used as the main raw material for he synthesis of various pesticides, medicines, surfactants, polymer monomers, Ond Ontifungal agents
Cas:480-47-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Hangzhou Fandachem Co.,Ltd
HydrangenolAppearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transportation:by air, by sea, by express
Shanghai Yuanye Bio-Technology Co., Ltd.
good quality, competitive price, thoughtful after sale serviceAppearance:white powder Storage:Keep it in dry,shady and cool place Package:5mg Application:Pharma;Industry;Agricultural;chemical reaserch Transportation:by express or by sea Port:Any port
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Chengdu Push Bio-technology Co., Ltd
Chengdu Push Bio-technology Co., Ltd. provides more than 4,000 natural monomeric substances, including approximately 6,000 products at different grades, specifications and purities. We have specialized in the R&D of medicinally active ingredients sta
Shaanxi Cuicheng Biomedical Technology Co., Ltd.
Shaanxi Cuicheng Biomedical Technology Co., Ltd. is located in Chuangzhi Industrial Park, Economic and Technological Development Zone, Hanzhong City, Shaanxi Province, China. It is a company specializing in natural medicines and high-throughput scree
Hangzhou Haiqiang Chemical Co.,Ltd.
Hangzhou Haiqiang Chem is a leading manufacturer and supplier of chemicals in China.We develop,produce and distribute high quality pharmaceuticals, intermediates, special chemicals and other fine chemicals. We could give you: 1.Best quali
Wuhan MoonZY Biological Technology Co.,Ltd
instock with good quality and wholesale price Storage:Keep in a cool & dry place Package:Packing material and QTY as your request Application:Pharma;Industry;other application Transportation:Express or as your request Port:Any port of China
Shanghai Run-Biotech Co., Ltd.
1.We have rich experience in the development and production 2.Our factory is in Shanghai of China,We have convenient traffic conditions.3.We have advanced testing equipment and shipping line. Package:5g, 10g, 50g, 100g, 1kg Application:Pharmaceutical
BioBioPha Co., Ltd
In Stock,Offering NMR, HPLC/TLC and COA Reports Application:For pharmaceutical, cosmetics, agrochemical and food industries research / Activity Screening / Standard / Reference compounds
Synthetic route
-
-
82780-51-6
(+/-)-3-(4-methoxyphenyl)-8-methoxy-3,4-dihydroisocoumarin

-
-
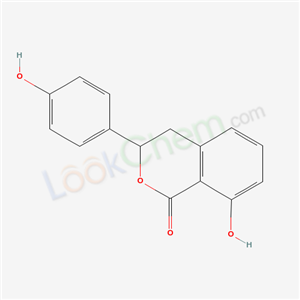
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| With boron tribromide In dichloromethane at -78 - 25℃; | 94% |
| With boron tribromide In dichloromethane 1.) -78 deg C , 1 h , 2.) -78 deg C to RT , 12 h; | 94% |
| With boron tribromide In dichloromethane at -78 - 20℃; | 88% |
-
-
637774-32-4
3-(4'-benzyloxyphenyl)-8-hydroxy-3,4-dihydro-2-benzopyran-1-one

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethyl acetate under 3750.3 Torr; | 84% |

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 4h; Heating; | 81% |
-
-
491-79-2, 113428-99-2
Hydrangeaic acid

-
A
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| With copper dichloride In acetone for 3h; Heating; | A 75% B 11.2% C 5.1% |

-
-
67600-94-6
Hydrangenol-8-O-glucoside

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| With sulfuric acid at 80 - 90℃; for 1h; | 73.5% |
-
-
1093748-41-4
methyl 2-(tert-butyldimethylsilyloxy)-6-(bromomethyl)-benzoate

-
-
120743-99-9
p-[(tert-butyldimethylsilyl)oxy]benzaldehyde

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Stage #1: methyl 2-(tert-butyldimethylsilyloxy)-6-(bromomethyl)-benzoate; p-[(tert-butyldimethylsilyl)oxy]benzaldehyde With titanocene(III) chloride In tetrahydrofuran at 20℃; for 17h; Stage #2: With sulfuric acid; water In tetrahydrofuran | 51% |
-
-
491-79-2, 113428-99-2
Hydrangeaic acid

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| at 180℃; |

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| With sulfuric acid |

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Stage #1: 1-[4-(tert-butyl-dimethyl-silanyloxy)-phenyl]-2-[2-(4,4-dimethyl-4,5-dihydro-oxazol-2-yl)-phenyl]-ethanol With Trimethyl borate; sec.-butyllithium In tetrahydrofuran; hexane; cyclohexane at -78 - 20℃; Stage #2: With dihydrogen peroxide In acetic acid at 20℃; Stage #3: With trifluoroacetic acid In tetrahydrofuran; water Heating; Further stages.; |
-
-
79383-44-1
methyl 2-methyl-6-methoxybenzoate

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 92 percent / NBS; AIBN / CCl4 2: 53 percent / Cp2TiCl2 / tetrahydrofuran / 20 °C 3: 88 percent / BBr3 / CH2Cl2 / -78 - 20 °C View Scheme |
-
-
943595-13-9
methyl 2-(bromomethoxy)-6-methoxybenzoate

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 53 percent / Cp2TiCl2 / tetrahydrofuran / 20 °C 2: 88 percent / BBr3 / CH2Cl2 / -78 - 20 °C View Scheme |
-
-
120743-99-9
p-[(tert-butyldimethylsilyl)oxy]benzaldehyde

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sBuLi / tetrahydrofuran; cyclohexane; hexane / -78 °C 2.1: sBuLi; B(OMe)3 / tetrahydrofuran; cyclohexane; hexane / -78 - 20 °C 2.2: H2O2 / acetic acid / 20 °C 2.3: TFA / tetrahydrofuran; H2O / Heating View Scheme |
-
-
71885-44-4
4,5-dihydro-4,4-dimethyl-2-(2-methylphenyl)-2-oxazoline

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sBuLi / tetrahydrofuran; cyclohexane; hexane / -78 °C 2.1: sBuLi; B(OMe)3 / tetrahydrofuran; cyclohexane; hexane / -78 - 20 °C 2.2: H2O2 / acetic acid / 20 °C 2.3: TFA / tetrahydrofuran; H2O / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 89 percent / sodium ethoxide / ethanol / 24 h / Heating 2: 94 percent / KOH / ethanol / 24 h / Heating 3: 87 percent / trifluoroacetic acid / 1,2-dichloro-ethane; H2O / 5 h / Heating 4: 84 percent / H2 / Pd/C / ethyl acetate / 3750.3 Torr View Scheme | |
| Multi-step reaction with 5 steps 1: 68.2 percent / 10percent aq. NaOH / 14 h / Ambient temperature 2: 78 percent / dimethylformamide / 4 h / 80 °C 3: 1.) NaH, 2.) BuLi, 3.) Bf3*OEt2, 4.) KF 4: 90 percent / 10percent KOH / ethanol / 3 h / Heating 5: 81 percent / conc. HCl / methanol / 4 h / Heating View Scheme |
-
-
118842-88-9
(3-acetyloxy-2-ethoxycarbonylbenzyl)-triphenylphosphonium bromide

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 89 percent / sodium ethoxide / ethanol / 24 h / Heating 2: 94 percent / KOH / ethanol / 24 h / Heating 3: 87 percent / trifluoroacetic acid / 1,2-dichloro-ethane; H2O / 5 h / Heating 4: 84 percent / H2 / Pd/C / ethyl acetate / 3750.3 Torr View Scheme |
-
-
148324-47-4
2-(4'-benzyloxyphenyl)-1-(3-hydroxy-2-ethoxycarbonylphenyl)ethene

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 87 percent / trifluoroacetic acid / 1,2-dichloro-ethane; H2O / 5 h / Heating 2: 84 percent / H2 / Pd/C / ethyl acetate / 3750.3 Torr View Scheme |
-
-
148324-45-2
2-(4'-benzyloxyphenyl)-1-(3-hydroxy-2-ethoxycarbonylphenyl)ethene

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 94 percent / KOH / ethanol / 24 h / Heating 2: 87 percent / trifluoroacetic acid / 1,2-dichloro-ethane; H2O / 5 h / Heating 3: 84 percent / H2 / Pd/C / ethyl acetate / 3750.3 Torr View Scheme |
-
-
36640-12-7
3-(4-methoxyphenyl)-7-methoxy-2-benzopyran-1(1H)-one

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 2: 78 percent / BBr3 View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 5: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 5 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 5: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 6 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 90 percent / 95percent phosphoric acid / 0.25 h / Heating 5: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 6: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 5 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 64 percent / sodium borohydride, 10percent NaOH 5: 78 percent / BBr3 View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 5: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 5 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 5: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 6 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 90 percent / 95percent phosphoric acid / 0.25 h / Heating 5: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 6: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 5 steps 1: 78.5 percent / polyphosphoric acid / 1.5 h / 70 - 75 °C 2: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 3: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 4: 64 percent / sodium borohydride, 10percent NaOH 5: 78 percent / BBr3 View Scheme |
-
-
98540-26-2
1′-(3-methoxyphenyl)-2′-(4′′-methoxyphenyl)ethan-2′-one

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 2: 1.) 1.6M butyllithium / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 3: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 4: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 2: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 3: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 4: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 5 steps 1: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 2: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 3: 90 percent / 95percent phosphoric acid / 0.25 h / Heating 4: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 5: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) trimethyl orthoformate, methanol, p-toluenesulphonic acid, 2.) p-toluenesulphonic acid / 1.) heating, 15 min, 2.) benzene, heating 2: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 3: 64 percent / sodium borohydride, 10percent NaOH 4: 78 percent / BBr3 View Scheme |
-
-
117970-24-8
3',4-dimethoxydeoxybenzoin ethylene acetal

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) 1.6M butyllithium / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 2: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 3: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 2: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 3: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 2: 90 percent / 95percent phosphoric acid / 0.25 h / Heating 3: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 4: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) 1.6M butyllithium, 2.) CO2 / 1.) toluene, hexane, 24 h, 0 deg C, 2.) toluene, hexane, -20 deg C up to r.t. 2: 64 percent / sodium borohydride, 10percent NaOH 3: 78 percent / BBr3 View Scheme |
-
-
117970-26-0
3',4-dimethoxydeoxybenzoin-2'-carboxylic acid

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 90 percent / 95percent phosphoric acid / 0.25 h / Heating 2: 60 percent / H2 / 10percent Pt/C / tetrahydrofuran 3: 78 percent / BBr3 View Scheme | |
| Multi-step reaction with 2 steps 1: 64 percent / sodium borohydride, 10percent NaOH 2: 78 percent / BBr3 View Scheme |
-
-
66947-61-3
5-methoxybicyclo[4.2.0]octa-1,3,5-trien-7-ol

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 96 percent / lithium 2,2,6,6-tetramethylpiperidide / tetrahydrofuran / Ambient temperature 2: 77 percent / PCC / CH2Cl2 / 12 h / Ambient temperature 3: 83 percent / BBr3 / CH2Cl2 / 3 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 96 percent / lithium 2,2,6,6-tetramethylpiperidide / tetrahydrofuran / Ambient temperature 2: 77 percent / PCC / CH2Cl2 / 12 h / Ambient temperature 3: 83 percent / BBr3 / CH2Cl2 / 3 h / Ambient temperature View Scheme |
-
-
157251-01-9
3-(4'-Methoxyphenyl)-1-hydroxy-8-methoxyisochroman

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 77 percent / PCC / CH2Cl2 / 12 h / Ambient temperature 2: 83 percent / BBr3 / CH2Cl2 / 3 h / Ambient temperature View Scheme |
-
-
75676-91-4
1-(4-benzyloxyphenyl)but-1-en-3-one

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 78 percent / dimethylformamide / 4 h / 80 °C 2: 1.) NaH, 2.) BuLi, 3.) Bf3*OEt2, 4.) KF 3: 90 percent / 10percent KOH / ethanol / 3 h / Heating 4: 81 percent / conc. HCl / methanol / 4 h / Heating View Scheme |

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) NaH, 2.) BuLi, 3.) Bf3*OEt2, 4.) KF 2: 90 percent / 10percent KOH / ethanol / 3 h / Heating 3: 81 percent / conc. HCl / methanol / 4 h / Heating View Scheme |

-
-
480-47-7
hydrangenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 90 percent / 10percent KOH / ethanol / 3 h / Heating 2: 81 percent / conc. HCl / methanol / 4 h / Heating View Scheme |
-
-
480-47-7
hydrangenol

-
-
491-79-2, 113428-99-2
Hydrangeaic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol for 1.5h; Heating; | 100% |
| With sodium hydrogencarbonate In methanol |
-
-
480-47-7
hydrangenol

-
-
23255-59-6
lunularic acid

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; palladium dichloride In methanol for 1h; Ambient temperature; | 99.6% |
-
-
480-47-7
hydrangenol

-
-
80394-91-8
dl-3,4-dihydro-8-hydroxy-3-(p-hydroxyphenyl)isocarbostyryl

| Conditions | Yield |
|---|---|
| With ammonia In ethanol at 100℃; for 17h; autoclave; | 64% |

| Conditions | Yield |
|---|---|
| Bei der Kalischmelze; |
-
-
480-47-7
hydrangenol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
183619-44-5
8-(tert-Butyl-dimethyl-silanyloxy)-3-[4-(tert-butyl-dimethyl-silanyloxy)-phenyl]-isochroman-1-one

| Conditions | Yield |
|---|---|
| With 1H-imidazole | |
| With 1H-imidazole In N,N-dimethyl-formamide at 25℃; for 4h; | 240 mg |

| Conditions | Yield |
|---|---|


| Conditions | Yield |
|---|---|
-
-
480-47-7
hydrangenol

-
-
491-79-2, 113428-99-2
Hydrangeaic acid

| Conditions | Yield |
|---|---|
-
-
480-47-7
hydrangenol

-
-
491-79-2, 113428-99-2
Hydrangeaic acid

| Conditions | Yield |
|---|---|
-
-
480-47-7
hydrangenol

-
-
80458-94-2
3'-Deoxythunberginol A

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 240 mg / imidazole / dimethylformamide / 4 h / 25 °C 2: 89.9 percent / DDQ / benzene / 5 h / Heating 3: 100 percent / n-Bu4NF / tetrahydrofuran / 0.08 h / 25 °C View Scheme | |
| Multi-step reaction with 3 steps 1: imidazole 2: 2,3-dichloro-5,6-dicyano-1,4-benzoquinone 3: n-Bu4NF View Scheme |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View