Wuhan Fortuna Chemical Co.,Ltd
Chemical Supplier 3-O-Benzyl-4-C-hydroxyMethyl-1,2-O-isopropylidene-alpha-D-ribofuranose Cas 63593-03-3 Company profile Wuhan Fortuna Chemical Co.,Ltd established in 2006, is a big integrative chemical enterprise being engaged in Pharmaceutical
Cas:63593-03-3
Min.Order:1 Gram
FOB Price: $10.0
Type:Trading Company
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:63593-03-3
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine c
Cas:63593-03-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:63593-03-3
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.
Cas:63593-03-3
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:63593-03-3
Min.Order:1 Gram
FOB Price: $1.0 / 99.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:63593-03-3
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
3-O-BENZYL-4-(HYDROXYMETHYL-1,2-O-ISOPROPYLIDENE)-ALPHA-D-ERYTHROPENTOFURANOSE CAS:63593-03-3 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemica
Cas:63593-03-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:63593-03-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:63593-03-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:63593-03-3
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Cas:63593-03-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryNewCan Biotech Limited
NewCan Biotech Limited was established in 2021 and is primarily engaged in the research, development, production, and sales of sugars, nucleosides, nucleotides, phosphorylated monomers, as well as next-generation antiviral and antitumor drug intermed
Cas:63593-03-3
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:63593-03-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShaanxi Cuicheng Biomedical Technology Co., Ltd.
Why Choose Us: 1. Factory direct sales, so we can provide the competitive price and high quality product base on 8 years of production and R&D experience. 2. It is available in stock for quick shipment.Products could be packaged according to cu
Cas:63593-03-3
Min.Order:1 Gram
Negotiable
Type:Other
inquiryChemlyte Solutions
Chemlyte Solutions believe that customers and suppliers deserve much more than what traditional distributors can offer. To grow in today s fast-paced and increasingly competitive market it is essential to be able to quickly adapt to market forces eff
Cas:63593-03-3
Min.Order:100 Gram
Negotiable
Type:Other
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:63593-03-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryYUNBIO TECH CO.,LTD
Our Strength: -Flexible: We work in small teams with rapid communication channels. Respond quickly and efficiently to customer and market needs. -Innovative: We are focusing on the development of new products and keep close relationships with
Cas:63593-03-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:63593-03-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:63593-03-3
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryShanghai Minstar Chemical Co., Ltd
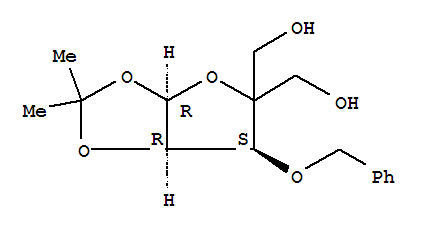
-O-BENZYL-4-(HYDROXYMETHYL-1,2-O-ISOPROPYLIDENE)-ALPHA-D-ERYTHROPENTOFURANOSE Basic information Product Name: 3-O-BENZYL-4-(HYDROXYMETHYL-1,2-O-ISOPROPYLIDENE)-ALPHA-D-ERYTHROPENTOFURANOSE Synonyms: ((3aR,6S,6aR);-2,2-dimethyltetrahydrofuro[2,3
Cas:63593-03-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:63593-03-3
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:63593-03-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:63593-03-3
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquirySuzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Cas:63593-03-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:63593-03-3
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
1, High quality with competitive price:2, Fast and safe delivery3.Excellent pre-sales and after-sales service4. Well-trained and professional technologist and sales with rich experience in the field for 5-10 yearsAppearance:see detailed specification
Cas:63593-03-3
Min.Order:0
Negotiable
Type:Trading Company
inquiryWin-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:63593-03-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Cas:63593-03-3
Min.Order:0
Negotiable
Type:Other
inquiryGIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
Cas:63593-03-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
50-00-0
formaldehyd

-
-
63593-02-2
3-O-benzyl-1,2-O-isopropylidene-α-D-ribo-pentodialdo-1,4-furanose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane Cannizzaro reaction; | 94% |
| With sodium hydroxide In 1,4-dioxane at 28 - 30℃; for 16.83h; | 75% |
| With sodium hydroxide In tetrahydrofuran; water at 20℃; | 47% |
| With sodium hydroxide In tetrahydrofuran; water at 0 - 20℃; | 47% |
| With sodium tetrahydroborate In tetrahydrofuran; water at 20℃; | 52.5 g |
-
-
620630-82-2
1,2 5,6-O-di(isopropylidene)-α-D-allofuranose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose
-
-
57099-04-4
3-O-benzyl-1,2-O-isopropylidene-α-D-allofuranose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaIO4 / methanol 2: 94 percent / aq. NaOH / dioxane View Scheme | |
| Multi-step reaction with 2 steps 1: sodium periodate / water / 1 h / 0 °C 2: sodium hydroxide / water; 1,4-dioxane / 144 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: sodium periodate / water; tetrahydrofuran / 1.5 h / 10 °C / Large scale 2: sodium hydroxide / 1,4-dioxane / 16.83 h / 28 - 30 °C View Scheme |
-
-
22331-21-1
1,2:5,6-di-O-isopropylidene-3-O-benzyl-α-D-allofuranose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aq. HCl / ethanol 2: aq. NaIO4 / methanol 3: 94 percent / aq. NaOH / dioxane View Scheme | |
| Multi-step reaction with 3 steps 1: acetic acid / water / 72 h / 20 °C 2: sodium periodate / water / 1 h / 0 °C 3: sodium hydroxide / water; 1,4-dioxane / 144 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: acetic acid / water / 20 - 25 °C / Large scale 2: sodium periodate / water; tetrahydrofuran / 1.5 h / 10 °C / Large scale 3: sodium hydroxide / 1,4-dioxane / 16.83 h / 28 - 30 °C View Scheme |
-
-
50-00-0
formaldehyd

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane; water at 0 - 20℃; for 144h; | 77 g |
-
-
2595-05-3
Diacetone D-glucose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sodium hydride / mineral oil; acetonitrile / 1 h / 0 °C / Inert atmosphere 1.2: 4.5 h / 0 °C 2.1: acetic acid / water / 72 h / 20 °C 3.1: sodium periodate / water / 1 h / 0 °C 4.1: sodium hydroxide / water; 1,4-dioxane / 144 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1: sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate / toluene / 4 h / 30 - 90 °C / Large scale 2: acetic acid / water / 20 - 25 °C / Large scale 3: sodium periodate / water; tetrahydrofuran / 1.5 h / 10 °C / Large scale 4: sodium hydroxide / 1,4-dioxane / 16.83 h / 28 - 30 °C View Scheme |
-
-
100-39-0
benzyl bromide

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sodium hydride / mineral oil; acetonitrile / 1 h / 0 °C / Inert atmosphere 1.2: 4.5 h / 0 °C 2.1: acetic acid / water / 72 h / 20 °C 3.1: sodium periodate / water / 1 h / 0 °C 4.1: sodium hydroxide / water; 1,4-dioxane / 144 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: sodium hydride / N,N-dimethyl-formamide / 0.5 h / 0 °C 1.2: 0 - 20 °C 2.1: acetic acid; water / 20 °C 3.1: sodium periodate / water; methanol / 1 h / 20 °C 4.1: sodium tetrahydroborate / water; tetrahydrofuran / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: sodium hydride / N,N-dimethyl-formamide / 0.5 h / 0 °C 1.2: 0 - 20 °C 2.1: acetic acid / water / 20 °C 3.1: sodium periodate / water; methanol / 1 h / 20 °C 4.1: sodium hydroxide / water; tetrahydrofuran / 0 - 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: sodium hydride / N,N-dimethyl-formamide / 0.5 h / 0 °C 1.2: 0 - 20 °C 2.1: acetic acid / water / 20 °C 3.1: sodium periodate / methanol; water / 1 h / 20 °C 4.1: sodium hydroxide / tetrahydrofuran; water / 20 °C View Scheme |
-
-
1013943-40-2
3-O-benzyl-1,2:5,6-di-O-isopropylidene-α-D-allofuranose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: acetic acid; water / 20 °C 2: sodium periodate / water; methanol / 1 h / 20 °C 3: sodium tetrahydroborate / water; tetrahydrofuran / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: acetic acid / water / 20 °C 2: sodium periodate / water; methanol / 1 h / 20 °C 3: sodium hydroxide / water; tetrahydrofuran / 0 - 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: acetic acid / water / 20 °C 2: sodium periodate / methanol; water / 1 h / 20 °C 3: sodium hydroxide / tetrahydrofuran; water / 20 °C View Scheme |

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium periodate / water; methanol / 1 h / 20 °C 2: sodium tetrahydroborate / water; tetrahydrofuran / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: sodium periodate / water; methanol / 1 h / 20 °C 2: sodium hydroxide / water; tetrahydrofuran / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: sodium periodate / methanol; water / 1 h / 20 °C 2: sodium hydroxide / tetrahydrofuran; water / 20 °C View Scheme |
-
-
610-85-5, 7045-51-4, 19217-07-3, 30412-16-9, 34685-37-5, 36468-79-8, 36468-80-1, 36468-81-2, 36468-82-3, 36468-84-5, 36574-15-9, 36574-18-2, 36574-19-3, 36574-21-7, 36972-23-3, 36978-43-5, 37738-80-0, 40461-75-4, 40461-79-8, 40461-80-1, 40461-83-4, 40550-49-0, 41612-82-2, 41846-85-9, 41846-86-0, 41846-87-1, 41846-88-2, 41846-89-3, 41846-90-6, 41846-91-7, 41846-92-8, 41846-93-9, 41846-94-0, 41846-95-1, 41846-96-2, 41846-97-3, 41847-47-6, 41847-48-7, 41847-49-8, 41847-50-1, 41847-62-5, 41847-63-6, 41847-64-7, 41847-65-8, 41847-66-9, 41847-67-0, 41847-68-1, 51076-04-1, 89673-68-7, 131064-93-2, 131064-94-3, 131064-95-4, 131064-96-5, 131064-97-6, 131065-02-6, 131065-03-7, 131065-04-8, 131065-05-9, 131065-06-0
D-glucofuranose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: zinc(II) chloride; phosphoric acid / 15 - 30 °C / Large scale 2: potassium bromide; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium hypochlorite; sodium hydrogencarbonate / water; ethyl acetate / 0 - 35 °C 3: sodium tetrahydroborate / water / 10 - 15 °C 4: sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate / toluene / 4 h / 30 - 90 °C / Large scale 5: acetic acid / water / 20 - 25 °C / Large scale 6: sodium periodate / water; tetrahydrofuran / 1.5 h / 10 °C / Large scale 7: sodium hydroxide / 1,4-dioxane / 16.83 h / 28 - 30 °C View Scheme |
-
-
582-52-5
1,2:5,6-di-O-isopropylidene-α-D-glucofuranose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: potassium bromide; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium hypochlorite; sodium hydrogencarbonate / water; ethyl acetate / 0 - 35 °C 2: sodium tetrahydroborate / water / 10 - 15 °C 3: sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate / toluene / 4 h / 30 - 90 °C / Large scale 4: acetic acid / water / 20 - 25 °C / Large scale 5: sodium periodate / water; tetrahydrofuran / 1.5 h / 10 °C / Large scale 6: sodium hydroxide / 1,4-dioxane / 16.83 h / 28 - 30 °C View Scheme |
-
-
2847-00-9
1,2:5,6-di-O-isopropylidene-α-D-hexofuran-3-ulose

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: sodium tetrahydroborate / water / 10 - 15 °C 2: sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate / toluene / 4 h / 30 - 90 °C / Large scale 3: acetic acid / water / 20 - 25 °C / Large scale 4: sodium periodate / water; tetrahydrofuran / 1.5 h / 10 °C / Large scale 5: sodium hydroxide / 1,4-dioxane / 16.83 h / 28 - 30 °C View Scheme |
-
-
108-05-4
vinyl acetate

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| With candida antarctica lipase-B In di-isopropyl ether at 45℃; for 1.5h; Reagent/catalyst; Solvent; Green chemistry; Enzymatic reaction; regioselective reaction; | 100% |
-
-
108-24-7
acetic anhydride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane; water at 20℃; | 99% |
-
-
124-63-0
methanesulfonyl chloride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
293751-01-6
3-O-benzyl-4-C-methanesulfonoxymethyl-5-methanesulfonyl-1,2-O-isopropylidene-α-D-erythro-pentofuranose

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 1h; | 98% |
| With pyridine at 0 - 20℃; for 1h; | 98% |
| With pyridine In dichloromethane | 98% |
-
-
123-62-6
propionic acid anhydride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane; water at 20℃; | 98% |
-
-
106-31-0
butanoic acid anhydride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane; water at 20℃; | 98% |
-
-
108-24-7
acetic anhydride

-
-
65-71-4
thymin

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
552856-36-7
1-(2,5-di-O-acetyl-4-C-acetyloxymethyl-3-O-benzyl-β-D-erythro-pentofuranosyl)thymine

| Conditions | Yield |
|---|---|
| Stage #1: acetic anhydride; 1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose With sulfuric acid In water; acetic acid at 0 - 20℃; for 2.41667h; Stage #2: thymin With N,O-bis-(trimethylsilyl)-acetamide; trimethylsilyl trifluoromethanesulfonate In acetonitrile at 0 - 80℃; for 5h; Heating / reflux; | 91% |

-
-
100-39-0
benzyl bromide

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
153186-10-8
((3aR,5R,6S,6aR)-6-(benzyloxy)-5-((benzyloxy)methyl)-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxol-5-yl)methanol

| Conditions | Yield |
|---|---|
| Stage #1: 1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose With sodium hydride In tetrahydrofuran at 0℃; for 0.166667h; Inert atmosphere; Stage #2: benzyl bromide In tetrahydrofuran at 0 - 20℃; for 2h; Inert atmosphere; | 73% |
| With sodium hydride In N,N-dimethyl-formamide; mineral oil at -5 - 20℃; for 3h; | 69% |
| Stage #1: 1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose With sodium hydride In N,N-dimethyl-formamide; mineral oil at -5℃; for 0.5h; Stage #2: benzyl bromide In N,N-dimethyl-formamide; mineral oil at 20℃; for 3h; | 69% |
-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
212970-72-4
{3-[(2,2-dimethyl-1,1-diphenyl-1-silapropoxy)methyl]-(1S,3R,4S,5R)-7,7-dimethyl-2,6,8-trioxa-4-(phenylmethoxy)bicyclo[3.3.0]oct-3-yl}methan-1-ol

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 14h; Etherification; Silylation; | 67% |
| In dichloromethane at 20℃; for 11h; | 67% |
-
-
100-44-7
benzyl chloride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
B
-
153186-11-9
{(1S,3S,4S,5R)-7,7-dimethyl-2,6,8-trioxa-4-(phenylmethoxy)-3-[(phenylmethoxy)methyl]-bicyclo[3.3.0]oct-3-yl}methan-1-ol

-
C
-
153186-10-8
((3aR,5R,6S,6aR)-6-(benzyloxy)-5-((benzyloxy)methyl)-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxol-5-yl)methanol

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at -9℃; for 1h; | A 10% B 11% C 66% |

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
A
-
212970-72-4
{3-[(2,2-dimethyl-1,1-diphenyl-1-silapropoxy)methyl]-(1S,3R,4S,5R)-7,7-dimethyl-2,6,8-trioxa-4-(phenylmethoxy)bicyclo[3.3.0]oct-3-yl}methan-1-ol

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane for 1h; Heating; | A 38% B 62% |

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
A
-
945628-28-4
C22H36O6Si

-
B
-
314256-39-8
C22H36O6Si

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 16h; | A 22% B 59% |

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
153186-10-8
((3aR,5R,6S,6aR)-6-(benzyloxy)-5-((benzyloxy)methyl)-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxol-5-yl)methanol

| Conditions | Yield |
|---|---|
| Stage #1: 1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.25h; Stage #2: With benzyl bromide In N,N-dimethyl-formamide at 0 - 20℃; | 51% |
-
-
100-39-0
benzyl bromide

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
B
-
153186-11-9
{(1S,3S,4S,5R)-7,7-dimethyl-2,6,8-trioxa-4-(phenylmethoxy)-3-[(phenylmethoxy)methyl]-bicyclo[3.3.0]oct-3-yl}methan-1-ol

-
C
-
153186-10-8
((3aR,5R,6S,6aR)-6-(benzyloxy)-5-((benzyloxy)methyl)-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxol-5-yl)methanol

| Conditions | Yield |
|---|---|
| Stage #1: 1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose With di(n-butyl)tin oxide In methanol for 1h; Substitution; Heating; Stage #2: benzyl bromide With tetrabutylammomium bromide In toluene for 15h; Etherification; Heating; | A 23% B 21% C 19% |

-
-
108-24-7
acetic anhydride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
552856-35-6
Acetic acid (3S,4R)-4,5-diacetoxy-2-acetoxymethyl-3-benzyloxy-tetrahydro-furan-2-ylmethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid at 20℃; for 2h; | 258 mg |
| With sulfuric acid In acetic acid at 0 - 20℃; for 2.41667h; |
-
-
98-88-4
benzoyl chloride

-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
552856-52-7
5-O-benzoyl-4-C-benzoyloxymethyl-3-O-benzyl-1,2-O-isopropylidene-α-D-erythro-pentofuranose

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945382-96-7
1-[4-C-allyl-3,5-di-O-benzyl-2-O-acetyl-β-D-ribofuranosyl]-thymine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: acetic acid; triflic acid / 0.5 h 8.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 8.2: 6 g / TMSOTf / acetonitrile / 0 - 20 °C View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: acetic acid; triflic acid / 0.5 h 8.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 8.2: 6 g / TMSOTf / acetonitrile / 0 - 20 °C 9.1: ammonia / methanol View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945382-98-9
1-[4-C-allyl-3,5-di-O-benzyl-2-O-phenoxythiocarbonyl-β-D-ribofuranosyl]-thymine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: acetic acid; triflic acid / 0.5 h 8.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 8.2: 6 g / TMSOTf / acetonitrile / 0 - 20 °C 9.1: ammonia / methanol 10.1: 4.1 g / DMAP; pyridine View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: acetic acid; triflic acid / 0.5 h 8.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 8.2: 6 g / TMSOTf / acetonitrile / 0 - 20 °C 9.1: ammonia / methanol 10.1: 4.1 g / DMAP; pyridine 11.1: Bu3SnH / toluene / 0.5 h / Heating 11.2: 2,2'-azobis(2-methyl-propionitrile; Bu3SnH / toluene / 2 h / Heating View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: acetic acid; triflic acid / 0.5 h 8.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 8.2: 6 g / TMSOTf / acetonitrile / 0 - 20 °C 9.1: ammonia / methanol 10.1: 4.1 g / DMAP; pyridine 11.1: Bu3SnH / toluene / 0.5 h / Heating 11.2: 2,2'-azobis(2-methyl-propionitrile; Bu3SnH / toluene / 2 h / Heating View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945383-09-5
3,5-di-O-benzyl-4-C-hydroxypropyl-1,2-O-isopropylidene-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 8.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945383-12-0
3,5-di-O-benzyl-4-C-penten-yl-1,2-O-isopropylidene-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 8.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 9.1: BuLi / tetrahydrofuran; hexane / -78 °C 9.2: 3.5 g / tetrahydrofuran; hexane View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 8.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 9.1: BuLi / tetrahydrofuran; hexane / -78 °C 9.2: 3.5 g / tetrahydrofuran; hexane 10.1: acetic acid; triflic acid / 0.5 h / ice bath View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945383-14-2
C30H34N2O7

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 8.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 9.1: BuLi / tetrahydrofuran; hexane / -78 °C 9.2: 3.5 g / tetrahydrofuran; hexane 10.1: acetic acid; triflic acid / 0.5 h / ice bath 11.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 11.2: TMSOTf / acetonitrile / 0 - 20 °C View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945383-16-4
1-[3,5-di-O-benzyl-4-C-penten-yl-2-hydroxy-β-D-ribofuranosyl]-thymine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 12 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 8.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 9.1: BuLi / tetrahydrofuran; hexane / -78 °C 9.2: 3.5 g / tetrahydrofuran; hexane 10.1: acetic acid; triflic acid / 0.5 h / ice bath 11.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 11.2: TMSOTf / acetonitrile / 0 - 20 °C 12.1: 2.8 g / ammonia / methanol View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945383-18-6
1-[3,5-di-O-benzyl-4-C-penten-yl-2-O-phenoxythiocarbonyl-β-D-ribofuranosyl]-thymine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 13 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 8.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 9.1: BuLi / tetrahydrofuran; hexane / -78 °C 9.2: 3.5 g / tetrahydrofuran; hexane 10.1: acetic acid; triflic acid / 0.5 h / ice bath 11.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 11.2: TMSOTf / acetonitrile / 0 - 20 °C 12.1: 2.8 g / ammonia / methanol 13.1: 60 percent / DMAP; pyridine View Scheme |
-
-
63593-03-3
1,2-O-isopropylidene-3-O-benzyl-4-hydroxymethyl-α-D-ribofuranose

-
-
945383-20-0
(1R,2R,5R,7R,8S)-8-benzyloxy-5-benzyloxymethyl-2-methyl-7-(thymin-1-yl)-6-oxa-bicyclo[3.2.1]octane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 14 steps 1.1: 66 percent / NaH / -5 - 20 °C 2.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 3.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 3.2: 17 g / tetrahydrofuran; hexane / -78 °C 4.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 4.2: 95 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 5.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 6.1: BuLi / tetrahydrofuran; hexane / 1 h / 20 °C 6.2: 12 g / tetrahydrofuran; hexane / -78 °C 7.1: 9-borobicyclo(3.3.1)nonane / tetrahydrofuran 7.2: 90 percent / aq. NaOH; aq. H2O2 / tetrahydrofuran / 0.5 h / 20 °C 8.1: oxalyl chloride; DMSO; diisopropylethylamine / CH2Cl2 / -78 - 20 °C 9.1: BuLi / tetrahydrofuran; hexane / -78 °C 9.2: 3.5 g / tetrahydrofuran; hexane 10.1: acetic acid; triflic acid / 0.5 h / ice bath 11.1: N,O-bis(trimethylsilyl)acetamide / acetonitrile / 0.75 h / Heating 11.2: TMSOTf / acetonitrile / 0 - 20 °C 12.1: 2.8 g / ammonia / methanol 13.1: 60 percent / DMAP; pyridine 14.1: Bu3SnH / toluene / 0.25 h / Heating 14.2: 76 percent / 2,2'-azobis(2-methyl-propionitrile; Bu3SnH / toluene / 2 h / Heating View Scheme |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View