Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:7727-15-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:7727-15-3
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:7727-15-3
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Competitive Price High Quality Fast Delivery custom-made Welcome to Henan Tianfu Chemical Co., Ltd. website. Our company engages in Sodium Tripolyphosphate (STPP) and Sodium Hexametabphosphate (SHMP) production; development of noble me
Cas:7727-15-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Product name: Aluminum Bromide CAS: 7727-15-3 MF: AlBr3 MW: 266.69 EINECS No: 231-779-7 Puiry: 99.0%min Appearance:White powder Storage:RT. Package:drum Application:Pharmaceutical Intermediates Transportation:By sea or by air Port:Qingd
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7727-15-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,or Intermediates,fine chemicals Transportation:air,sea,courier
Cas:7727-15-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high qualityAppearance:white crystalline powder Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Cas:7727-15-3
Min.Order:0
Negotiable
Type:Manufacturers
inquiryZhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:7727-15-3
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hangzhou Ocean Chemical Co., Ltd.
Ocean inorganic department is a professional supplier and exporter engaging in inorganic chemical materials and metal organic compounds. Over the past years, our company is committed to improving the product quality and developing new products, in or
Hubei Langyou International Trading Co., Ltd
Aluminium bromide CAS NO.7727-15-3 Application:Aluminium bromide CAS NO.7727-15-3
Hunan Russell Chemicals Technology Co.,Ltd
low price and high purityAppearance:solid or liquid Storage:in sealed air resistant place Package:As customer require Application:Pharma;Industry;Agricultural Transportation:by sea or by airplane Port:any port in China
Cas:7727-15-3
Min.Order:0
Negotiable
Type:Trading Company
inquiryHunan Longxianng Runhui Trading Co.,Ltd
Aluminium bromide CAS NO.7727-15-3Appearance:off-white fine crystalline powder Storage:Dry and ventilated Package:Standard or as customer's require Application:intermediates Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small a
Cas:7727-15-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBeantown Chemical
in stock Application:7727-15-3
Cas:7727-15-3
Min.Order:0
Negotiable
Type:Trading Company
inquiryHENAN SUNLAKE ENTERPRISE CORPORATION
Aluminium bromide Basic information Product Name: Aluminium bromide
Cas:7727-15-3
Min.Order:100 Gram
Negotiable
Type:Trading Company
inquiryChangzhou Extraordinary Pharmatech co.,LTD
Changzhou Extraordinary Pharmatech co., LTD. As a leading chemical manufacturer and supplier in China.DAS authentication is passed.We can provide the popular precursor chemicals, we have our own strong R & D team, have our own laboratories and fa
Hebei Ruishun Trade Co.,Ltd
Profile HeBei Ruishun Trade Co.LTD, registered capital one million,have a production of pharmaceutical raw materials, pharmaceutical raw materials factory reagent r&d center,Seek development by credit
Xiamen BaiFuchem Co.,Ltd
BaiFuChem is a Professional chemical raw material supplier in China, our main products include Biochemical , Pharma Intermediate and Organic chemical etc. BaiFuChem have wealth of products,experience , expertise and state-of-the-art
Hangzhou Share Chemical Co., Ltd
At Share Chemical Company, we scrupulously abide by our policy of “Excellent Quality at a Reasonable Price”. We strive to satisfy all of our customers by providing the finest quality products supported by the finest in customer servi
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Strem Chemicals, Inc.
1.High Quality Specialty Chemicals2.Expertise and Experience in Manufacturing High Purity, High Quality Chemicals3.Long Term Focus in Inorganics, Organometallics and Metals4.ISO 9001 CertifiedAppearance:white to off-white lumps
Synthetic route
-
-
3530-39-0
phenylaluminium dichloride

-
-
106-95-6
allyl bromide

-
A
-
300-57-2
allylbenzene

-
B
-
92-52-4
biphenyl

-
C
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| bis(η3-allyl-μ-chloropalladium(II)) In diethyl ether Ar atmosphere; stirring (20°C, 5 mole-% catalyst, 8 h); | A 94% B <1 C n/a |
| tetrakis(triphenylphosphine) palladium(0) In diethyl ether Ar atmosphere; stirring (20°C, 5 mole-% catalyst, 20 h); | A <1 B <1 C n/a |

-
-
6591-30-6
diphenylaluminum chloride

-
-
106-95-6
allyl bromide

-
A
-
300-57-2
allylbenzene

-
B
-
92-52-4
biphenyl

-
C
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In diethyl ether Ar atmosphere; stirring (20°C, 5 mole-% catalyst, 20 h); | A 37% B <1 C n/a |



| Conditions | Yield |
|---|---|
| Erhitzen des Reaktionsprodukts im Vakuum auf 200grad; |
-
-
1333-84-2, 1344-28-1
aluminum oxide

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With bromine; pyrographite In not given heating; | |
| With bromine; pyrographite heating; | |
| With sulfur dibromide In solid 350°C, with vaporous sulfur bromide; |

| Conditions | Yield |
|---|---|
| reaction started at 350°C and accelerated by raising of temp.; excess of S2Br2 removed with HBr or nitrogen; | |
| reaction started at 350°C and accelerated by raising of temp.; excess of S2Br2 removed with HBr or nitrogen; |

| Conditions | Yield |
|---|---|
| With aluminum(III) sulfate In water heating on water bath with small amt. of Br2 and H2O; | |
| With bisilicates; carbon heating; | |
| With carbon; silicates heating; |

| Conditions | Yield |
|---|---|
| In not given heating on water bath with small amt. of Br2; | |
| In not given heating on water bath with small amt. of Br2; |

| Conditions | Yield |
|---|---|
| In gas Al and Br2 in vac. were vaporized separately and reacted in gaseous state; distd. in vac.; | |
| With air; water in moist air containing Br2 condensation of droplets of a soln. of AlBr3 occurs on a gray layer that has formed first;; | |
| In neat (no solvent) start of react. by heating;; |

| Conditions | Yield |
|---|---|
| With bromine reacted in the presence of AlBr3; | |
| With bromine Al heated in combustion tube; with H2 or N2 deluted Br2 lead over Al; | |
| With bromine Al sheet heated in combustion tube; vaporous Br2 leaded over Al; AlBr3 flowed out from tube; |

| Conditions | Yield |
|---|---|
| In ethylene dibromide | |
| In ethylene dibromide |


| Conditions | Yield |
|---|---|
| In not given heating of Al with SnBr4 for 30 min gives AlBr3;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction with SnBr2 under formation of AlBr3 above 216°C;; differentialthermoanalytic examinations;; |

| Conditions | Yield |
|---|---|
| at higher temp.;; |


-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| byproducts: Br2, CS2; decompn. at 150°C; | |
| byproducts: Br2, CS2; decompn. at 150°C; |

| Conditions | Yield |
|---|---|
| at 200°C; | |
| at 200°C; |

| Conditions | Yield |
|---|---|
| 120°C; | |
| 120°C; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) mixt. of the starting materials heated in evacuated sealed quartz tubes to 500-900°C; decompn. of educts ocurrs, AlBr3 sublimates off; | A 0% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction at 100°C;; | |
| In neat (no solvent) reaction at 100°C;; |

-
-
10097-32-2
bromine

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With sulfur introducing a vapor of a soln. of S in Br2 over Al oxide; | |
| With S introducing a vapor of a soln. of S in Br2 over Al oxide; |
-
-
119484-32-1
AsBr4(1+)*AlBr4(1-)={AsBr4}{AlBr4}

-
A
-
7726-95-6
bromine

-
B
-
7727-15-3
aluminium bromide

-
C
-
7784-33-0
arsenic(III) bromide

| Conditions | Yield |
|---|---|
| equilibrium at room temp.; |


-
B
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With copper(l) chloride In tetrahydrofuran byproducts: Cu, LiCl; to a soln. of Ti-complex in THF at 0°C is added CuCl; pptn. of Cu, solvent is removed in vac., the residue is dissolved in toluene, pptd. LiCl is centrifuged off, the soln. is vacuum-evaporated, from the residue at 90-110°C is distilled the complex, crystn. at 20°C, elem. anal.; |

-
A
-
14741-34-5, 21255-52-7
tricarbonyl bis(triphenylphosphine) iron

-
B
-
51744-69-5, 15020-31-2, 84622-99-1
bis(triphenylphosphine)dicarbonyldibromoiron(II)

-
C
-
7726-95-6
bromine

-
D
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | A <1 B n/a C n/a D n/a |
| In methanol | A <1 B n/a C n/a D n/a |
| In dichloromethane | A <1 B n/a C n/a D n/a |

-
-
361447-05-4
tetraiodophosphonium tetrabromoaluminate

-
A
-
7727-15-3
aluminium bromide

-
B
-
7789-33-5
iodine(I) bromide

-
C
-
13455-01-1
phosphorous triiodide

| Conditions | Yield |
|---|---|
| In carbon disulfide under N2, anaerobic and anhyd. conditions; dissolved in CS2; |


-
A
-
22359-97-3
aluminium(I) bromide

-
B
-
136684-48-5
AlBr2

-
C
-
74-90-8, 6914-07-4
hydrogen isocyanide

-
D
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In neat (no solvent, gas phase); solid matrix byproducts: CH4, H2C=CH2, H2C=CHCH3; N2 atmosphere, high vacuum (1E-6 to 1E-7 mbar) thermolysis at 1000°C, trapping in argon at 15 K; further organic byproducts: H2C=N(CH3)2 and H2CCHCH2; IR-monitoring; |


| Conditions | Yield |
|---|---|
| In solid heating at 400°C; |
-
-
16853-85-3
lithium aluminium tetrahydride

-
-
124-11-8
non-1-ene

-
-
7727-15-3
aluminium bromide

-
-
180334-67-2
1-dibromoaluminiononane

| Conditions | Yield |
|---|---|
| In diethyl ether; benzene Ar, LiAlH4 in ether added to benzene under stirring, most of the solvent removed, benzene and AlBr3 in benzene added, stirred at 20°C for 1 h, org. compound in benzene added dropwise under stirring, warmed to30-40°C for 5-10 min; NMR; | 100% |

-
-
7727-15-3
aluminium bromide

-
-
920-36-5, 90269-51-5
(2-methylpropyl)lithium

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: LiBr; to stirred mixt. of MgBr2, AlBr3 and toluene at room temp., soln. of iso-C4H9Li in hexane added (Ar; ratio MgBr2, AlBr3, RLi: 1:2:8) within 20 min; react. for 3.0 h at 80°C; ppt. filtered off (Ar); (1)H NMR of soln.; | 100% |


| Conditions | Yield |
|---|---|
| In benzene under N2, dropwise addn. of thiol soln. to a suspension of AlBr3 in benzene, dissoln. of AlBr3, stirred for 15 min at room temp.; evapn. of solvent, dried in vac. at room temp. for 3h, elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| With tetraallyltin In benzene byproducts: SnBr4, propene; 2,6-diphenylphenol (12 equiv.) treated with AlBr3 (4 equiv.) in benzene at room temp., tetraallyltin (3 equiv.) added, stirred at room temp. for1 h, solvent evapd. in vacuo for 3 h with concomitant removal of SnBr4 and propene; | 100% |

| Conditions | Yield |
|---|---|
| In benzene under N2, CH3SH introduced in a suspension of AlBr3 for 7 min, dissoln.of AlBr3, stirred for 15 min at room temp.; evapn. of solvent, dried in vac. at room temp. for 3h, elem. anal.; | 100% |
-
-
3908-55-2
Trimethyl(methylthio)silane

-
-
7727-15-3
aluminium bromide

-
-
112908-14-2
aluminium tribromide*trimethylsilyl methyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSMe was added dropwise under N2 to a suspn. of AlBr3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |
-
-
5573-62-6
(ethylthio)trimethylsilane

-
-
7727-15-3
aluminium bromide

-
-
112908-15-3
aluminium tribromide*trimethylsilyl ethyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSEt was added dropwise under N2 to a suspn. of AlBr3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |
-
-
4551-15-9
phenylthiotrimethylsilane

-
-
7727-15-3
aluminium bromide

-
-
112908-16-4
aluminium tribromide*trimethylsilyl phenyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSPh was added dropwise under N2 to a suspn. of AlBr3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |

-
-
7727-15-3
aluminium bromide

-
-
374569-92-3
supersilylaluminum dibromide

| Conditions | Yield |
|---|---|
| In n-heptane Kinetics; all manipulations with exclusion of air and moisture; compd. prepd. fromAlBr3 and org. compd. reacted in molar ratio of 1:1 at room temp. for 2 4 d (yield depended on react. time); controlled by NMR; volatiles evapd. by oil pump, residue dissolved in heptane, NaBr filtered; | 100% |
-
-
7789-65-3
selenium tetrabromide

-
-
7727-15-3
aluminium bromide

-
-
69951-93-5
tribromoselenium(IV) tetrabromoaluminate

| Conditions | Yield |
|---|---|
| In neat (no solvent) Ar atm. (300 mbar); heating (150-200°C, several days); | 100% |
| In dichloromethane addn. of AlBr3 to SeBr4, stirring (2 h); filtration (under dry N2), drying (vac.); elem. anal.; |

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; ethylene dibromide | 100% |

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; ethylene dibromide | 100% |

-
-
7727-15-3
aluminium bromide

-
-
79560-37-5
bis(η5-cyclopentadienyl)tungsten dihydride * AlBr3

| Conditions | Yield |
|---|---|
| In diethyl ether stoich. amts. (pptn.); | 99% |
-
-
55664-33-0
bis(tricyclohexylphosphine)platinum(0)

-
-
7727-15-3
aluminium bromide

-
-
960294-94-4
[(Cy3P)2Pt(AlBr3)]

| Conditions | Yield |
|---|---|
| In benzene AlBr3 was added to a soln. of Pt-complex in benzene, the soln. was kept at room temp. for 24 h; | 99% |
| In toluene (Ar); Al salt added to a soln. of Pt complex; evapd. (vac.); elem. anal.; | 98% |

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) (Ar); solid state reaction; equimolar mixt. of TlBr, and AlBr3 sealed under vac. in glass ampoules; heated with 10°C/h to 150°C for 48 h; cooled to 25°C with 2.degre.C/h; detn. by XRD; | 99% |
-
-
110-71-4
1,2-dimethoxyethane

-
-
7727-15-3
aluminium bromide

-
-
182561-85-9
aluminium tribromide-1,2-dimethoxyethane

| Conditions | Yield |
|---|---|
| In toluene (N2); stirring (1 h); filtration, washing (toluene); elem. anal.; | 98.7% |

| Conditions | Yield |
|---|---|
| In benzene (glovebox); addn. of a soln. of AlBr3 in C6H6 to a soln. of silicon compd. in C6H6 in vac. with stirring, sealing, heating to 40°C for 30min with shaking; centrifugation, decantation, evapn; elem. anal.; | A 98% B 98% |

-
-
176763-62-5
(1R,2R)-(-)-N,N'-bis(3,5-di-tert-butylsalicydene)-1,2-cyclohexanediaminocobalt(II)

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; water addn. of Co(C6H10(NCHC6H2O(t-Bu)2)2) (1 equiv.) to soln. of AlBr3 (1 equiv.) in CH2Cl2/H2O; stirring at room temp. for 1 h; concn. under reduced pressure, treatment with H2O and CH2Cl2; | 98% |

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With I2 In dichloromethane a mixt. of B compd., I2, and AlBr3 in CH2Cl2 refluxed for 16.5 h under N2; poured into ice-cold H2O, the org. phase sepd., the aq. layer extd. (Et2O), the combined org. phase washed (Na2S2O3 soln., then H2O), dried (MgSO4), solvent-removed, recrystd. (hexane); elem. anal.; | 98% |
-
-
109-99-9
tetrahydrofuran

-
-
97355-16-3
N,N-bis(2-hydroxy-3,5-di-tert-butylphenyl)amine

-
-
7727-15-3
aluminium bromide

-
-
1384526-06-0
[bis(3,5-di-tert-butyl-2-phenol)amine(2-)]AlBr(THF)

| Conditions | Yield |
|---|---|
| With KH In tetrahydrofuran under inert atm. mixt. ligand and KH in THF were stirred at room temp. for 2 h, soln. was frozen, AlBr3 was added upon thawing, stirred at room temp. overnight; solvent was removed in vacuo, residue was co-evapd. with pentane; elem. anal.; | 98% |


| Conditions | Yield |
|---|---|
| In methyl cyclohexane addn. of 2-butyne to soln. of AlBr3, 0°C, stirred 15h at 25°C, filtered, washed with methylcyclohexane, dried in vac.; elem. anal.; | 97% |

| Conditions | Yield |
|---|---|
| In n-heptane room temp.; | 97% |
| In benzene room temp.; | 97% |
| In benzene room temp.; | 97% |
-
-
7727-15-3
aluminium bromide

-
-
653-38-3
(pentafluoro phenyl) methyl mercury

-
-
4457-90-3
(pentafluoro phenyl) dibromo alane

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating of the components ( molar ratio 1 : 1) in a closed tube at 70°C, 5 days;; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: uranium dioxide; aluminium bromide at 250℃; for 12h; Sealed tube; Stage #2: at 350℃; for 144h; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| In diethyl ether; benzene Ar, LiAlH4 in ether added to benzene under stirring, most of the solvent removed, benzene and AlBr3 in benzene added, stirred at 20°C for 1 h, org. compound in benzene added drowise under stirring, warmed to 30-40°C for 5-10 min; NMR; | 96% |

-
-
26944-84-3
1,3-dimethyl-2-dimethylaminodiazaborolane

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| under N2, at -78°C; excess of Lewis-acid used; elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| In toluene N2-atmosphere; slow addn. of Al-salt to slight excess of alcohol at 25°C, stirring for 4 h; drying (vac.), extn. into CH2Cl2, drying (vac.); elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| In benzene (glovebox); dropwise addn. of a soln. of AlBr3 in C6H6 to a soln. of silicon compd. in C6H6 over 15 min with stirring, refluxing for 7 h; centrifugation, decantation, evapn.; elem. anal.; | A 88% B 96% |

-
-
211381-77-0
(2,2,6,6-tetramethylpiperidino)2AlBr(isoquinoline)

-
-
7727-15-3
aluminium bromide

-
-
211381-88-3
(2,2,6,6-tetramethylpiperidino)2Al(isoquinoline) tetrabromoaluminate

| Conditions | Yield |
|---|---|
| In Bromoform inert atmosphere, addn. of soln. of AlBr3 to soln of isoquinoline complex at 0°C, 30 min; reduction of volume, crystn. (-20°C, overnight); elem. anal.; | 96% |
-
-
16853-85-3
lithium aluminium tetrahydride

-
-
625-65-0
2,4-dimethyl-2-pentene

-
-
7727-15-3
aluminium bromide

-
-
180334-71-8
(CH3)2CHCH(AlBr2)CH(CH3)CH3

| Conditions | Yield |
|---|---|
| In diethyl ether; benzene Ar, LiAlH4 in ether added to benzene under stirring, most of the solvent removed, benzene and AlBr3 in benzene added, stirred at 20°C for 1 h, org. compound in benzene added drowise under stirring, warmed to 30-40°C for 5-10 min; NMR; | 96% |

Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
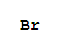















 C,
C, Xi,
Xi, N,
N, F
F