hangzhou verychem science and technology co.ltd
Hangzhou Verychem Science And Technology Co. Ltd. was set up in year 2004, it’s a young but fast growing company. In the twelve years history, it invested two pharmaceutical raw material production plants, one researching company, and several
Cas:109-99-9
Min.Order:1 Metric Ton
FOB Price: $20.0
Type:Lab/Research institutions
inquiryVD BIOTECH LIMITED
Appearance:Transparent Liquid Storage:Cool and Dry Place Package:Packed in Drum Application:Industry Use, Solvent. Port:Qingdao
Cas:109-99-9
Min.Order:1 Metric Ton
FOB Price: $6250.0
Type:Trading Company
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% ----------------------------------------------------------------
Cas:109-99-9
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryLIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc.
Cas:109-99-9
Min.Order:1 Kilogram
FOB Price: $0.9 / 1.0
Type:Lab/Research institutions
inquiryWuhan Fortuna Chemical Co.,Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; ...... Appearance:Colorless transparent liquid Storage:2-8°C Package:180kg/drum
Cas:109-99-9
Min.Order:180 Kilogram
FOB Price: $4.0 / 7.0
Type:Trading Company
inquiryChemwill Asia Co., Ltd.
Highly quality and large production Factory Chemwill Asia co.,Ltd is one of the leading manufacturer in CHINA. Our main production base is located in Xuzhou industry park. We produce a wide range of organics includin
Cas:109-99-9
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryAlity Chemical Corporation
Tetrahydrofuran Basic information Product Name: Tetrahydrofuran Synonyms: TETRAMETHYLENE ETHER GLYCOL 2000 POLYMER;Tetrahydrofuran ,99.8% [Tetrahydrofuran,ACS/HPLC Certified];Tetrahydrofuran, 99.6%, stabilized with BHT, for analysis ACS;Tetrahy
Henan Tianfu Chemical Co., Ltd.
Tianfu Chemical, built in 2009, is a professional supplier for API materials in China. With 10 years development, we have bulit our own factory and lab to produce a certain of products. And we have established stable business relationship
Cas:109-99-9
Min.Order:1 Kilogram
FOB Price: $200.0
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:109-99-9
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Name Tetrahydrofuran Synonyms 1,4-Epoxybutane; Butylene oxide; Cyclotetramethylene oxide; Furanidine; THF Molecular Formula C
Hubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best s
Cas:109-99-9
Min.Order:10 Gram
Negotiable
Type:Other
inquiryChangchun Artel lmport and Export trade company
Superiority We can customize and synthesize products that other suppliers may not be able to provide. we offer low price custom synthesis and contract manufacturing Advantage cof, mof ligand manufacturer Product Detail
Cas:109-99-9
Min.Order:100 Kilogram
Negotiable
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional chemical raw materials and chemical reagents research and development production enterprises. We have several production line,So we can control the lowest price. We also have sever
Cas:109-99-9
Min.Order:220 Liter
FOB Price: $2.8 / 2.9
Type:Trading Company
inquiryWuhan Zenuo Biological Medicine Technology Co Ltd
Product Name: Tetrahydrofuran Synonyms: 1,4-epoxy-butan;agrisynththf;Butane alpha,delta-oxide;Butane, 1,4-epoxy-;butane,alpha,delta-oxide;butanealpha,delta-oxide;Dynasolve 150;Hydrofuran CAS: 109-99-9 MF: C4H8O MW: 72.11 EINECS: 203-726-
Cas:109-99-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei yanxi chemical co.,LTD.
who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and development production of reagent tens
Cas:109-99-9
Min.Order:1 Kilogram
FOB Price: $200.0
Type:Trading Company
inquirySiwei Development Group Ltd.
Product name: Tetrahydrofuran CAS No.: 109-99-9 Molecule Formula:C4H8O Molecule Weight:184.23 Purity: 99.0% Package: 180kg/drum Description:Colorless transparent liquid Manufacture Standards:Enterprise Standard TES
Cas:109-99-9
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Henan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...)
Cas:109-99-9
Min.Order:1 Kilogram
FOB Price: $5.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Win-Win chemical Co.Ltd
Stock products, own laboratoryAppearance:Colorless to light yellow clear liquid Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:109-99-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXi'an yuanfar international trade company
with strong MFG,with best service,with best payment term Methods of tetrahydrofuran Industrial production of the earliest sugar aldehyde as raw material, the mixture of sugar aldehyde and steam through the reactor filled with zinc - chromium - mang
Hunan chemfish Pharmaceutical co.,Ltd
It is used as chromatographic reagent, organic solvent and nylon intermediate. Application:It is used as chromatographic reagent, organic solvent and nylon intermediate.
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Guangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Afine Chemicals Limited
excellent quality and reliable supplierAppearance:COA mentioned Storage:COA mentioned Package:Standard or custom package Application:pharmaceutical raw materails Transportation:sea/air /courier Port:China main port
Shenzhen Foris Technology Co. LTD
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
LEAP CHEM Co., Ltd.
Best Seller, High Quality, Competitive Price, Fast Delivery, Quick ResponseAppearance:powder, or liquid Storage:Stored in room temperature, ventilated place Package:Bottle, barrel, cargo, container, etc. Application:Pharmaceuticals, intermediates, AP
MIT-IVY INDUSTRY CO.,LTD.
We can supply the products with more competitive cost. Raw material in low price makes your own products more competitive in your market.quotation &MSDS can be sent on request. 1. Direct factory sales 2. High quality prices 3. Fast,
Cas:109-99-9
Min.Order:15 Metric Ton
FOB Price: $5.0 / 10.0
Type:Manufacturers
inquiryAmadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:109-99-9
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryQINGDAO ON-BILLION INDUSTRAIL CO.,LTD
1. Tetrahydrofuran is one of our main products. 2. Colorless transparents liquid, no visible impurity. 3. The mature Technical support and professional logistic support 4. The best quality in your requirement. 5. The competitive price owi
Synthetic route

| Conditions | Yield |
|---|---|
| With 3% Pd/C; hydrogen In isopropyl alcohol at 219.84℃; under 25858.1 Torr; for 5h; Inert atmosphere; | 100% |
| With hydrogen; acetic acid In water at 39.84℃; for 2h; Inert atmosphere; | 98% |
| With ruthenium; hydrogen; 1-butyl-3-methylimidazolium Tetrafluoroborate at 25℃; under 22502.3 Torr; for 36h; Autoclave; chemoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With phosphoric acid; 5%-palladium/activated carbon; hydrogen at 100℃; under 3750.38 - 13501.4 Torr; for 7h; Reagent/catalyst; Pressure; Temperature; Autoclave; | 100% |

| Conditions | Yield |
|---|---|
| Trichlorbutylstannan at 80 - 84℃; for 9h; | 99% |
| Trichlorbutylstannan at 80 - 84℃; for 19h; Mechanism; different molar ratios, different times; | 99% |
| zirconium(IV) sulfate at 200℃; under 760.051 Torr; Product distribution / selectivity; Gas phase; | 99.5% |

| Conditions | Yield |
|---|---|
| Stage #1: n-butane With oxygen at 403℃; under 2175.22 Torr; Stage #2: With hydrogen In Phthalic acid dibutyl ester Product distribution / selectivity; | 99.5% |
-
-
110-16-7
maleic acid

-
A
-
109-99-9
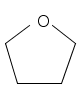
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
110-15-6
succinic acid

-
E
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| With hydrogen; 0.5 percent Pd on Rutile TiO2 at 110℃; Product distribution / selectivity; | A 0.37% B 0.28% C 0.37% D 98.89% E 0.08% |

-
-
110-16-7
maleic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
67-56-1
methanol

-
D
-
110-63-4
Butane-1,4-diol

-
E
-
617-48-1
malic acid

-
F
-
110-15-6
succinic acid

-
G
-
64-19-7
acetic acid

-
H
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen; 0.5percent Pd on Rutile TiO2 at 110℃; Product distribution / selectivity; | A 0.45% B 0.06% C 0% D 0.21% E 0.36% F 98.73% G 0.04% H 0.08% |

-
-
110-16-7
maleic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
591-81-1
4-hydroxybutanoic acid

-
E
-
110-15-6
succinic acid

-
F
-
64-19-7
acetic acid

-
G
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen; 0.5percent Pd on Rutile TiO2 at 110℃; Product distribution / selectivity; | A 0.77% B 0.38% C 0.24% D 0.05% E 98.28% F 0.02% G 0.26% |


| Conditions | Yield |
|---|---|
| With perchloric acid; sodium perchlorate; water at 25℃; | A n/a B n/a C 98% |

-
-
624-48-6
dimethyl cis-but-2-ene-1,4-dioate

-
A
-
109-99-9
tetrahydrofuran

-
B
-
13436-45-8
2-methoxytetrahydrofuran

-
C
-
96-48-0
4-butanolide

-
D
-
71-23-8
propan-1-ol

-
E
-
64001-06-5
2-(4'-hydroxybutoxy)-tetrahydrofuran

-
F
-
110-63-4
Butane-1,4-diol

-
G
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen; copper catalyst, T 4489, Sud-Chemie AG, Munich at 150 - 280℃; under 187519 Torr; Neat liquid(s) and gas(es)/vapour(s); | A 1% B n/a C 0.4% D n/a E n/a F 98% G 0.5% |


| Conditions | Yield |
|---|---|
| With water In toluene byproducts: hydrogen, dihydronaphthalene; reaction time: 20 h; centrifuged, decanted, pptn. (Yb(OH)3) washed with toluene, soln. contains THF and C10H10 (82%)(detn. by GLC); | A 112 % B 98% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium formate; Aliquat 336 at 105℃; for 0.25h; | 95% |

| Conditions | Yield |
|---|---|
| A n/a B 94.2% | |
| A n/a B 94.2% |


| Conditions | Yield |
|---|---|
| decompn. at 80°C (0.5 h); | A 93% B n/a |
| decompn. at 80°C (0.5 h); | A 93% B n/a |
-
-
110-16-7
maleic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
591-81-1
4-hydroxybutanoic acid

-
E
-
617-48-1
malic acid

-
F
-
110-15-6
succinic acid

-
G
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| With hydrogen; 0.5percent Pd/2.0percent Re on Rutile TiO2 at 110℃; Product distribution / selectivity; | A 1.27% B 4.78% C 1.55% D 1.24% E 0.48% F 90.6% G 0.08% |


-
A
-
109-99-9
tetrahydrofuran

-
B
-
523981-77-3
cyclopentadienylterbiumdibromide

| Conditions | Yield |
|---|---|
| In neat (no solvent) warmed at 40°C under high vac. for 24 h; elem. anal.; | A n/a B 90% |
-
-
110-15-6
succinic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen In 1,4-dioxane at 139.84℃; under 60006 Torr; for 96h; Catalytic behavior; Reagent/catalyst; Time; Temperature; Autoclave; Overall yield = 100 %; | A 0.2% B 3.1% C 89% D 7.6% |
| With hydrogen; 1.0percent Pd/ 3.0percent Re on Rutile TiO2 at 164 - 185℃; for 21 - 237h; Product distribution / selectivity; | A 2.95% B 0% C 81.5% D 3.35% |
| With hydrogen; 0percent Pd/5.0percent Re on Rutile TiO2 at 170 - 185℃; for 90 - 825h; Product distribution / selectivity; | A 3.38% B 0% C 64.14% D 2.86% |

-
-
110-15-6
succinic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
107-92-6
butyric acid

-
E
-
106-97-8
n-butane

-
F
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen In 1,4-dioxane at 139.84℃; under 60006 Torr; for 24h; Catalytic behavior; Reagent/catalyst; Time; Autoclave; Overall yield = > 99 %; | A 0.2% B 3.1% C 89% D n/a E n/a F 7.6% |

-
-
110-16-7
maleic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
617-48-1
malic acid

-
E
-
110-15-6
succinic acid

-
F
-
64-19-7
acetic acid

-
G
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen; 0.5percent Pd on Rutile TiO2 at 110℃; for 96 - 238h; Product distribution / selectivity; | A 0.6% B 0.04% C 0.62% D 0.19% E 88.49% F 0.12% G 0.11% |


| Conditions | Yield |
|---|---|
| With hydrogen at 350℃; for 0.6h; Reagent/catalyst; Temperature; Flow reactor; | A n/a B 85.9% |
| With Mg and Yb-containing organic foam into the binaryoxides at 350℃; Reagent/catalyst; Temperature; Inert atmosphere; | A n/a B 71.1% |
| With Er2O3 nanoparticles CM-1000 at 350℃; for 5h; | |
| at 350℃; Reagent/catalyst; Flow reactor; Inert atmosphere; |
-
-
110-16-7
maleic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
591-81-1
4-hydroxybutanoic acid

-
E
-
617-48-1
malic acid

-
F
-
110-15-6
succinic acid

-
G
-
100-21-0
terephthalic acid

-
H
-
64-19-7
acetic acid

-
I
-
802294-64-0
propionic acid

-
J
-
110-17-8
(2E)-but-2-enedioic acid

| Conditions | Yield |
|---|---|
| With hydrogen; 0.5percent Pd/0.2percent Re on Rutile TiO2 at 110℃; for 170 - 1009h; Product distribution / selectivity; | A 0.86% B 4.34% C 0.28% D 1.24% E 0% F 85.51% G 0% H 0.04% I 0% J 0% |

-
-
23055-10-9
1,4-dimethyl but-2-enedioate

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen In water at 130 - 182℃; under 52505.3 Torr; | A 6.34% B 5.33% C 83.96% |

-
-
88-14-2
2-furanoic acid

-
-
64-19-7
acetic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
628-67-1
butane-1,4-diol diacetate

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen; lanthanum(lll) triflate at 180℃; under 15001.5 Torr; for 5h; Autoclave; | A 6% B 83% |

-
-
96-48-0
4-butanolide

-
A
-
109-99-9
tetrahydrofuran

-
B
-
71-23-8
propan-1-ol

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
107-92-6
butyric acid

-
E
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen; 5% platinum on alumina at 250℃; under 152000 Torr; Product distribution; var. catalysts; | A 82.3% B 1% C 4.8% D 1.2% E 1% |

-
-
79372-14-8
decamethylsamarocene(II) bis(tetrahydrofurane)

-
-
75-24-1
trimethylaluminum

-
A
-
109-99-9
tetrahydrofuran

-
B
-
115756-72-4
(C5Me5)2Sm{(μ-Me)AlMe2(μ-Me)}2Sm(C5Me5)2

-
C
-
7429-90-5
aluminium

| Conditions | Yield |
|---|---|
| In toluene byproducts: methane; all manipulations conducted under nitrogen excluding air and water; after 24 h standing of the reaction mixt. the formed metallic-like ppt. was removed by filtration and washed with hot toluene, filtrates combined, solvent removed by rotary evapn.;; recrystn. (hot toluene), elem. anal.;; | A n/a B 80% C n/a |

-
-
624-48-6
dimethyl cis-but-2-ene-1,4-dioate

-
A
-
109-99-9
tetrahydrofuran

-
B
-
13436-45-8
2-methoxytetrahydrofuran

-
C
-
96-48-0
4-butanolide

-
D
-
71-23-8
propan-1-ol

-
F
-
64001-06-5
2-(4'-hydroxybutoxy)-tetrahydrofuran

-
H
-
110-63-4
Butane-1,4-diol

-
I
-
25714-71-0
4-hydroxybutyraldehyde

-
J
-
925-57-5
methyl 4-hydroxybutanoate

-
K
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen at 190℃; under 46504.7 Torr; Gas phase; | A 5.3% B n/a C 10.4% D n/a E n/a F n/a G n/a H 79.1% I n/a J n/a K n/a |

-
-
110-00-9
furan

-
-
67-56-1
methanol

-
A
-
109-99-9
tetrahydrofuran

-
B
-
13179-96-9
1,4-dimethoxybutane

-
C
-
107-31-3
Methyl formate

-
D
-
29006-01-7
Methyl 4-methoxybutyrate

-
E
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen at 170℃; under 52505.3 Torr; for 2h; Autoclave; | A 77.2% B n/a C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen at 130 - 182℃; under 52505.3 Torr; | A 13.09% B 77% |

| Conditions | Yield |
|---|---|
| decompn. at 140°C, <760 Torr; | A 75% B n/a |
-
-
110-63-4
Butane-1,4-diol

-
-
616-38-6
carbonic acid dimethyl ester

-
A
-
109-99-9
tetrahydrofuran

-
B
-
140947-75-7
butane-1,4-diyl dimethyl dicarbonate

| Conditions | Yield |
|---|---|
| With sodium methylate Reflux; Inert atmosphere; | A 12 %Chromat. B 75% |

-
-
109-99-9
tetrahydrofuran

-
-
33036-62-3
4-Bromo-1-butanol

| Conditions | Yield |
|---|---|
| With dimethylboron bromide; triethylamine In dichloromethane at 0℃; for 2h; | 100% |
| With sulfuric acid; hydrogen bromide | 90% |
| With tetrabutylammomium bromide; hydrogen bromide In water for 0.0833333h; Microwave irradiation; | 81% |

| Conditions | Yield |
|---|---|
| nickel at 100℃; for 1h; | 100% |
| With [(POCOP)Ir(H)(acetone)]+[B(C6F5)4]- In dichloromethane-d2 at 22℃; for 3h; | 100 %Spectr. |

| Conditions | Yield |
|---|---|
| With zinc(II) iodide In dichloromethane for 72h; Product distribution; other cyclic ethers; other Lewis acids; | 100% |
| With zinc(II) iodide In dichloromethane for 72h; | 100% |

| Conditions | Yield |
|---|---|
| With calcium for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With bis(iodozinc)methane at 25℃; for 2h; Substitution; | 100% |
| With bis(iodozinc)methane; lead(II) chloride at 25℃; for 2h; | 99% |
| With sodium iodide In acetonitrile at 0 - 23℃; for 24h; | 99.3% |
-
-
109-99-9
tetrahydrofuran

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
92511-12-1
1-{[dimethyl(1,1-dimethylethyl)silyl]oxy}-4-iodobutane

| Conditions | Yield |
|---|---|
| With sodium iodide In acetonitrile at 55℃; for 16h; Inert atmosphere; | 100% |
| With sodium iodide In tetrahydrofuran; acetonitrile at 55℃; for 18h; Inert atmosphere; | 100% |
| With sodium iodide In acetonitrile at 20℃; Inert atmosphere; Darkness; | 93% |
-
-
109-99-9
tetrahydrofuran

-
-
68602-57-3
trifluoroacetyl triflate

-
-
109244-09-9
Trifluoro-acetic acid 4-trifluoromethanesulfonyloxy-butyl ester

| Conditions | Yield |
|---|---|
| at 0℃; | 100% |

| Conditions | Yield |
|---|---|
| With bis(iodozinc)methane at 25℃; Substitution; | 100% |

| Conditions | Yield |
|---|---|
| With bis(iodozinc)methane at 25℃; Substitution; | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
625-35-4, 3488-22-0, 10487-71-5
trans-chrotonyl chloride

| Conditions | Yield |
|---|---|
| With bis(iodozinc)methane at 25℃; Substitution; | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
102-92-1
Cinnamoyl chloride

-
-
143903-00-8
(E)-3-Phenyl-acrylic acid 4-iodo-butyl ester

| Conditions | Yield |
|---|---|
| With bis(iodozinc)methane at 25℃; Substitution; | 100% |

| Conditions | Yield |
|---|---|
| With bis(iodozinc)methane at 25℃; Substitution; | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
217965-97-4
2,2,4,4,9,9,11,11,13,13,15,15,17,17,21,21-hexadecamethyl-2,4,9,11,13,15,17,21-octasilahexacyclo[10.5.3.15,8.06,18.07,20.014,19]henicosa-1(18),5,7,12(20),14(19)-pentaene

| Conditions | Yield |
|---|---|
| With lithium at 20℃; for 1h; Reduction; | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
87653-00-7
trichloro(4,5-dihydrofuran-3-yl)phosphonium hexachlorophosphate

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride In benzene at 10 - 20℃; for 2h; Substitution; | 100% |
| With phosphorus pentachloride In benzene at 10 - 20℃; for 2h; Inert atmosphere; | 100% |
-
-
109-99-9
tetrahydrofuran

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: dichloro(mesityl)phosphane; C14H20BrMgP*C4H10O at -78℃; Stage #2: tetrahydrofuran With lithium for 1h; sonication; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: tetrahydrofuran; N,N,N,N,-tetramethylethylenediamine; diphenylphosphane With n-butyllithium In tetrahydrofuran; hexane Stage #2: With tellurium In tetrahydrofuran; hexane at -78 - 20℃; | 100% |


| Conditions | Yield |
|---|---|
| With sodium hydride at 60℃; for 12h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydride at 60℃; for 12h; | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
13368-25-7
1-(bromomethyl)-4-vinylbenzene

- 4-vinylbenzyl-polytetrahydrofuran, with hydroxyl end group, polymerization degree 7.1 by 1H NMR, polydispersity 1.1; monomer(s): 4-vinylbenzyl bromide; tetrahydrofuran
-
4-vinylbenzyl-polytetrahydrofuran, with hydroxyl end group, polymerization degree 7.1 by 1H NMR, polydispersity 1.1; monomer(s): 4-vinylbenzyl bromide; tetrahydrofuran

| Conditions | Yield |
|---|---|
| Stage #1: tetrahydrofuran; 1-(bromomethyl)-4-vinylbenzene With silver(I) hexafluorophosphate at -10℃; for 0.133333h; Stage #2: With sodium hydroxide In water | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
14187-32-7
dibenzo-18-crown-6

| Conditions | Yield |
|---|---|
| With potassium borohydride for 528h; Heating; | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
213380-73-5
bis(α-trifluoromethyl-β,β-difluorovinyl) terephthalate

| Conditions | Yield |
|---|---|
| at 0℃; Irradiation; | 100% |
| at 0℃; Kinetics; Irradiation; |
-
-
109-99-9
tetrahydrofuran

-
-
395099-09-9
(2-amino-5-(benzyloxy)phenyl)(phenyl)methanone

| Conditions | Yield |
|---|---|
| In methanol; water | 100% |
| In methanol; water | 100% |
| In methanol; water | 100% |

-
-
109-99-9
tetrahydrofuran

-
-
40125-53-9
2-bromo-5-oxo-2,5-dihydrofuran

-
-
1030603-64-5
5-(4-bromobutoxy)furan-2(5H)-one

| Conditions | Yield |
|---|---|
| With zinc dibromide In dichloromethane for 4h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran W compd. dissolve in THF, stirred for 15 min, under anaerobic anhydrousconditions; elem. anal.; | 100% |
-
-
109-99-9
tetrahydrofuran

-
-
299410-39-2
Zr(C6H5NCH2CH2CH2NC6H5)2

-
-
10026-11-6
zirconium(IV) chloride

-
-
299410-33-6
Zr(PhN(CH2)3NPh)Cl2(THF)2

| Conditions | Yield |
|---|---|
| In diethyl ether byproducts: LiCl; N2 atm.; THF and diethyl ether were added by vac. transfer at -78°C to the mixt. of complex and ZrCl4, the mixt. was warmed to 0°Cin an ice bath, stirred overnight, allowed to warm to room temp.; volatiles were removed under vac. at 23°C; elem. anal.; | 100% |

-
-
109-99-9
tetrahydrofuran

-
-
354112-52-0
dicarbonyl[tris(pyrazol-1-yl)methanesulfonato]rhodium(I)

-
-
603-35-0
triphenylphosphine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran all manipulations under N2; P compd. added to soln. of complex in THF; after 1 h soln. concd., soln. overlaid with pentane, elem. anal.; | 100% |

-
-
109-99-9
tetrahydrofuran

-
-
168979-95-1
(Sm(OC15H23)2(O(C2H5)2))2(OC13H8)2

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (Ar); quantitative conversion; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (high vac. line); condensing gallium complex in an ampoule with THF, warming to room temp. over a period of 30 min; fractionation, collection in a trap at -20°C; | 100% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 Xi,
Xi, F
F