-
Name
1,4-Bis(diphenylphosphino)butane-palladium(II) chloride
- EINECS 243-231-4
- CAS No. 29964-62-3
- Article Data16
- CAS DataBase
- Density
- Solubility
- Melting Point 285-305 °C
- Formula C28H28Cl2P2Pd
- Boiling Point 542°C at 760 mmHg
- Molecular Weight 603.804
- Flash Point 299.5°C
- Transport Information
- Appearance yellow solid
- Safety 24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Palladium,[1,4-butanediylbis[diphenylphosphine]-P,P']dichloro-, (SP-4-2)-;Palladium,dichloro[tetramethylenebis[diphenylphosphine]]- (8CI);Palladium,[1,4-butanediylbis[diphenylphosphine-kP]]dichloro-, (SP-4-2)- (9CI);Phosphine,1,4-butanediylbis[diphenyl-, palladium complex;(1,4-Bis(diphenylphosphino)butane)palladium dichloride;1,4-Butylenebis(diphenylphosphine)palladium dichloride;Dichloro[1,4-bis(diphenylphosphino)butane]palladium;Dichloro[1,4-bis(diphenylphosphino)butane]palladium (II);[1,4-Bis(diphenylphosphino)butane]dichloropalladium;[1,4-Bis(diphenylphosphino)butane]palladium(II)dichloride;
- PSA 27.18000
- LogP 7.41140
Synthetic route
-
-
7647-01-0
hydrogenchloride

-
-
7440-05-3
palladium

-
-
7688-25-7
1,4-di(diphenylphosphino)-butane

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| Stage #1: hydrogenchloride; palladium With nitric acid In water Stage #2: 1,4-di(diphenylphosphino)-butane In water; N,N-dimethyl-formamide at 50℃; for 1h; | 98.9% |

-
-
21264-30-2, 90243-59-7, 14592-56-4
dichloro bis(acetonitrile) palladium(II)

-
-
7688-25-7
1,4-di(diphenylphosphino)-butane

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| In benzene at 110℃; for 48h; Schlenk technique; Inert atmosphere; | 97% |
| In 1,2-dichloro-ethane N2-atmosphere; stirring (room temp., 24 h); concg. (reduced pressure), filtering, washing (Et2O), drying (vac.); elem. anal.; | 94% |

-
-
7688-25-7
1,4-di(diphenylphosphino)-butane

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethanol stirring for 2 h; evapn., washing with H2O, drying at 80°C for 12 h; | 88% |
-
-
15617-18-2, 39958-10-6, 14220-64-5
bis(benzonitrile)palladium(II) dichloride

-
-
7688-25-7
1,4-di(diphenylphosphino)-butane

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| elem. anal.; | 83% |
| In not given | |
| In ethanol; dichloromethane | |
| In toluene at 20℃; for 0.5h; Inert atmosphere; | |
| In toluene at 20℃; for 0.5h; Inert atmosphere; |
-
-
570397-18-1
dichloro-(1,2-dimethoxy-4,5-bis(2-pyridylethynyl)benzene)palladium

-
-
7688-25-7
1,4-di(diphenylphosphino)-butane

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide room temp., 30 min; monitored by (1)H NMR; | 75% |
-
-
7688-25-7
1,4-di(diphenylphosphino)-butane

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| In acetonitrile stirring (70°C, 2 h), partial solvent evapn. (1/5); recrystn. (dimethylformamide), drying (vacuum); elem. anal.; | 61.4% |
| In dimethyl sulfoxide prepn. according to: A. Westland, J. Chem. Soc., (1965) 3060; ligand is mixed with PdCl2 in DMSO and heated at 100°C until a clear soln. is obtained; cooling down, filtration, washing with pentane; | >90 |
-
-
12107-56-1
dichloro(cycloocta-1,5-diene)palladium (II)

-
-
7688-25-7
1,4-di(diphenylphosphino)-butane

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| In dichloromethane under N2, addn. of complex in CH2Cl2 to soln. of ligand in CH2Cl2, stirred for 25°C for 2 h, white pptn.; evapd. under reduced press., addn. of toluene, heated under reflux for 4 h, pptn., filtered, dried in vac.; |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
75-09-2
dichloromethane

-
-
2923-28-6
silver trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| In dichloromethane under N2 atm. to suspn. Pd(dppb)Cl2 in CH2Cl2 AgOTf was added and stirred at room temp. for 24 h with exclusion light; soln. was filtered in air and concd. in vacuo, Et2O was added, ppt. was washed with Et2O and dried in vacuo; elem. anal.; | 96% |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
14239-51-1
bis(N,N-diethyldithiocarbamato)mercury(II)

-
-
75-09-2
dichloromethane

| Conditions | Yield |
|---|---|
| In dichloromethane stirred for 1 h; filtered, soln. concd. (vac.), pptd. (diethyl ether), ppt. filtered, washed (diethyl ether), dried (vac., 3 h), recrystd. (CH2Cl2); elem. anal.; | 94% |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
14239-68-0
cadmium(II) diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In dichloromethane stirred for 1 h; filtered, soln. concd. (vac.), pptd. (diethyl ether), ppt. filtered, washed (diethyl ether), dried (vac., 3 h), recrystd. (CH2Cl2); elem. anal.; | 94% |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
14324-55-1
bis(N,N-diethyldithiocarbamato)zinc(II)

-
-
75-09-2
dichloromethane

| Conditions | Yield |
|---|---|
| In dichloromethane stirred for 1 h; filtered, soln. concd. (vac.), pptd. (diethyl ether), ppt. filtered, washed (diethyl ether), dried (vac., 3 h), recrystd. (CH2Cl2); elem. anal.; | 93% |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
17549-30-3
bis(N,N-diethyldithiocarbamate)lead

-
-
75-09-2
dichloromethane

| Conditions | Yield |
|---|---|
| In dichloromethane stirred for 1 h; filtered, soln. concd. (vac.), pptd. (diethyl ether), ppt. filtered, washed (diethyl ether), dried (vac., 3 h), recrystd. (CH2Cl2); elem. anal.; | 93% |

-
-
14104-20-2
silver tetrafluoroborate

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| In dichloromethane; acetone byproducts: AgCl; N2-atmosphere; stirring (dark, 1 h); filtering, concg. (reduced pressure), pptn. on Et2O addn., filtering, washing (Et2O), drying. (vac.); elem. anal.; | 92% |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
86-93-1
1-Phenyl-1H-tetrazole-5-thiol

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; chloroform for 2h; Reflux; | 78% |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| at 20℃; Schlenk technique; Inert atmosphere; | 77% |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
143848-70-8, 320635-25-4
Pd(1,2-bis(diphenylphosphino)butane)(OSO2CF3)2

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under N2, addn. of 2 equiv. AgSO3CF3 to suspn. of Pd-complex in THF; filtration after 24 h, evapn. (vac.), recrystn. (CH2Cl2/Et2O), elem. anal.; | 58% |
| In not given |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
2123-72-0
1,4-dilithiobutane

| Conditions | Yield |
|---|---|
| In diethyl ether mixt. of the Pd compound and 1,4-dilithiobutane in Et2O stirred for 1 h at -78°C, then at room temp. for 3 h; solid sepd. from the soln., dried in vac. and extd. with toluene; soln. filtered and concd.; addn. of pentane to the filtrate, cooling to -30°C; elem. anal.; | 23% |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
12386-10-6
tetramethylammonium octahydrotriborate

| Conditions | Yield |
|---|---|
| With triethylamine In toluene; acetonitrile byproducts: {(CH3)4N}Cl, (C2H5)3N*B3H7, (C2H5)3N*BH3; under argon, stirring for 3 h, then standing for 1 h; further by-products: Et3NHCl, BH3*PPh2CH2CH2CH2CH2PPh2*BH3;; evapn. of the solvent in vac., chromy. (kieselgel 70-230 mesh, hexane);; | 5% |
-
-
14104-20-2
silver tetrafluoroborate

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| In dichloromethane; acetonitrile byproducts: AgCl; prepn. according to: Pisiano, C., Consiglio, G., Sirioni, A. & Moret, M., J. Chem. Soc., Chem. Commun., (1991) 421. mixing of suspn. of Pd-compd. in CH2Cl2 and AgBF4 in MeCN, stirring for some min; AgCl is filtered off; |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
917-54-4
methyllithium

-
-
113530-47-5
cis-dimethylbis(PPh2C4H8PPh2)palladium(II)

| Conditions | Yield |
|---|---|
| In diethyl ether |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
811-49-4
ethyllithium

-
-
96644-75-6
cis-diethyl-1,4-bis(diphenylphosphino)butane-palladium

| Conditions | Yield |
|---|---|
| In diethyl ether |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
320635-25-4, 143848-70-8
Pd(C6H5)2PC4H8P(C6H5)2(CF3SO3)(1+)*CF3SO3(1-)=Pd((C6H5)2PC4H8P(C6H5)2)(CF3SO3)2

| Conditions | Yield |
|---|---|
| In not given byproducts: AgCl; |
-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
2923-28-6
silver trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| In not given |
-
-
109-99-9
tetrahydrofuran

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: AgCl; |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| In acetonitrile byproducts: AgCl; |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
13862-78-7
ammonium tetrathiotungstate

-
-
7732-18-5
water

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| In dichloromethane; acetonitrile Pd-comp. and W-comp. added to CH2Cl2-CH3CN mixture, 1:1, v/v, stirred for 20 h; filtered, washed with CH2Cl2, distilled H2O, anhydrous EtOH and ether, elem. anal.; |

-
-
29964-62-3
[1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride

-
-
58249-25-5
sale sodico del 1,2-benzisotiazolin-3-one

| Conditions | Yield |
|---|---|
| In ethanol; dichloromethane for 3h; Heating; Overall yield = 78 %; Overall yield = 0.062 g; |

1,4-Bis(diphenylphosphino)butane-palladium(II) chloride Chemical Properties
Empirical Formula: C28H29ClP2Pd
Molecular Weight: 569.3504g/mol
EINECS: 243-231-4
Structure of 1,4-Bis(diphenylphosphino)butane-palladium(II) chloride (CAS NO.29964-62-3):
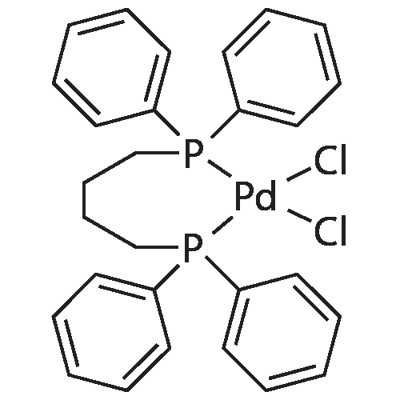
Physical Appearance: yellow solid
Product Categories: Metal Compounds;Catalysts for Organic Synthesis;Classes of Metal Compounds;Homogeneous Catalysts;Metal Complexes;Pd (Palladium) Compounds;Synthetic Organic Chemistry;Transition Metal Compounds
Systematic Name: (diphenylphosphanyl)palladium(1+) chloride - (1-methylpropyl)(diphenyl)phosphane (1:1)
SMILES: [Cl-].[Pd+]P(c1ccccc1)c2ccccc2.CC(CC)P(c1ccccc1)c2ccccc2
InChI: InChI=1/C16H19P.C12H10P.ClH.Pd/c1-3-14(2)17(15-10-6-4-7-11-15)16-12-8-5-9-13-16;1-3-7-11(8-4-1)13-12-9-5-2-6-10-12;;/h4-14H,3H2,1-2H3;1-10H;1H;/q;-1;;+2/p-1/rC16H19P.C12H10PPd.ClH/c1-3-14(2)17(15-10-6-4-7-11-15)16-12-8-5-9-13-16;14-13(11-7-3-1-4-8-11)12-9-5-2-6-10-12;/h4-14H,3H2,1-2H3;1-10H;1H/q;+1;/p-1 InChIKey: WCTRMVZCSUMBIZ-CTPBABKZAY
1,4-Bis(diphenylphosphino)butane-palladium(II) chloride Safety Profile
Hazard Codes:  Xn
Xn
Safety Statements: 24/25
S24/25:Avoid contact with skin and eyes.
WGK Germany: 3
F: 1-10
1,4-Bis(diphenylphosphino)butane-palladium(II) chloride Specification
1,4-Bis(diphenylphosphino)butane-palladium(II) chloride , its cas register number is 29964-62-3. It also can be called (Diphenylphosphino)palladium(1+) chloride - sec-butyl(diphenyl)phosphine (1:1:1) ; Palladium(II) chloride-1,4-bis(diphenylphosphine)butane . You should avoid contact 1,4-Bis(diphenylphosphino)butane-palladium(II) chloride (CAS NO.29964-62-3) with skin and eyes.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 29964-84-9
- 29965-34-2
- 29965-97-7
- 29966-04-9
- 29968-78-3
- 2996-92-1
- 29969-49-1
- 29969-57-1
- 299-70-7
- 29973-13-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View