-
Name
2,5-Di(tert-amyl)hydroquinone
- EINECS 201-222-2
- CAS No. 79-74-3
- Article Data37
- CAS DataBase
- Density 0.99 g/cm3
- Solubility 200ng/L at 20℃
- Melting Point 179 °C
- Formula C16H26 O2
- Boiling Point 364.8 °C at 760 mmHg
- Molecular Weight 250.381
- Flash Point 162.7 °C
- Transport Information
- Appearance White powder
- Safety Poison by intraperitoneal route. Low toxicity by ingestion. When heated to decomposition it emits acrid smoke and irritating fumes.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Hydroquinone,2,5-di-tert-pentyl- (6CI,7CI,8CI); 2,5-Bis(1,1-dimethylpropyl)-1,4-benzenediol;2,5-Bis(1,1-dimethylpropyl)hydroquinone;2,5-Di(tert-amyl)-1,4-benzohydroquinone; 2,5-Di(tert-amyl)hydroquinone;2,5-Di-t-amylhydroquinone; 2,5-Di-tert-amylhydroquinone;2,5-Di-tert-pentylhydroquinone; AH 25; Antage DAH; DAH; DAHQ; DTAHQ; Lowinox AH25; NSC 455; NSC 6267; Santovar A; Santovar TAHQ
- PSA 40.46000
- LogP 4.47300
Synthetic route

| Conditions | Yield |
|---|---|
| methanesulfonic acid In toluene at 55 - 70℃; for 7h; Product distribution / selectivity; | 90% |
| toluene-4-sulfonic acid In toluene at 85 - 110℃; for 4.5 - 7.5h; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetic acid at 20℃; for 7h; | 78% |
| With sulfuric acid | |
| methanesulfonic acid In toluene at 88 - 110℃; for 8.5h; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With acetic acid man gibt dann Amylen zu; |
-
-
513-35-9
2-methyl-but-2-ene

-
-
7664-93-9
sulfuric acid

-
-
64-19-7
acetic acid

-
-
123-31-9
hydroquinone

-
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone


| Conditions | Yield |
|---|---|
| With ethanol; zinc(II) chloride |
-
-
4584-63-8
2,5-bis-(1,1-dimethylpropyl)-1,4-benzoquinone

-
-
64-19-7
acetic acid

-
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone

-
-
64-17-5
ethanol

-
-
90-05-1
2-methoxy-phenol

-
A
-
1020-31-1
3,5-Di-tert-butylcatechol

-
B
-
128-39-2
2,6-di-tert-butylphenol

-
C
-
2934-05-6
2,4-diisopropylphenol

-
D
-
2078-54-8
2,6-diisopropylphenol

-
E
-
2934-07-8
2,4,6-triisopropylphenol

-
F
-
4130-42-1
2,6-di-tert-butyl-4-ethylphenol

-
G
-
2444-28-2
2,6-di-tert-butyl-4-hydroxyphenol

-
H
-
87-97-8
2,6-di-tert-butyl-4-methoxymethylene-phenol

-
I
-
1138-52-9
3,5-Di-tert-butylphenol

-
J
-
1879-09-0
2,4-dimethyl-6-tert-butylphenol

-
K
-
52417-48-8
2-(2',5'-dimethoxyphenyl)propionaldehyde

-
L
-
17540-75-9
2,6-di-tert-butyl-4-sec-butylphenol

-
M
-
2050-46-6
1,2-diethoxybenzene

-
N
-
21112-37-8
1,4-dimethoxy-2-tert-butylbenzene

-
O
-
5076-72-2
1-ethoxy-4-methoxybenzene

-
P
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone

-
Q
-
120-95-6
2,4-di-tert-amylphenol

-
R
-
876-20-0
2,5-diethyl phenol

-
S
-
33963-27-8
4-ethoxy-3-methoxytoluene

-
T
-
59056-76-7
1,2-dimethoxy-4-butylbenzene

-
U
-
131358-04-8
1,2-diethoxy-4-ethyl-benzene

-
V
-
1620-98-0
3,5-di-t-butyl-4-hydroxybenzaldehyde

| Conditions | Yield |
|---|---|
| With ortho-tungstic acid at 300℃; for 6h; Autoclave; |

-
-
64-17-5
ethanol

-
-
90-05-1
2-methoxy-phenol

-
A
-
94-71-3
2-Ethoxyphenol

-
B
-
1020-31-1
3,5-Di-tert-butylcatechol

-
C
-
2934-05-6
2,4-diisopropylphenol

-
D
-
2078-54-8
2,6-diisopropylphenol

-
E
-
2934-07-8
2,4,6-triisopropylphenol

-
F
-
4130-42-1
2,6-di-tert-butyl-4-ethylphenol

-
G
-
2444-28-2
2,6-di-tert-butyl-4-hydroxyphenol

-
H
-
87-97-8
2,6-di-tert-butyl-4-methoxymethylene-phenol

-
I
-
1138-52-9
3,5-Di-tert-butylphenol

-
J
-
1879-09-0
2,4-dimethyl-6-tert-butylphenol

-
K
-
52417-48-8
2-(2',5'-dimethoxyphenyl)propionaldehyde

-
L
-
17540-75-9
2,6-di-tert-butyl-4-sec-butylphenol

-
M
-
2050-46-6
1,2-diethoxybenzene

-
N
-
21112-37-8
1,4-dimethoxy-2-tert-butylbenzene

-
O
-
5076-72-2
1-ethoxy-4-methoxybenzene

-
P
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone

-
Q
-
120-95-6
2,4-di-tert-amylphenol

-
R
-
876-20-0
2,5-diethyl phenol

-
S
-
33963-27-8
4-ethoxy-3-methoxytoluene

-
T
-
59056-76-7
1,2-dimethoxy-4-butylbenzene

-
U
-
131358-04-8
1,2-diethoxy-4-ethyl-benzene

-
V
-
1620-98-0
3,5-di-t-butyl-4-hydroxybenzaldehyde

| Conditions | Yield |
|---|---|
| With tungsten(VI) oxide at 300℃; for 6h; Reagent/catalyst; Autoclave; |

-
-
64-17-5
ethanol

-
-
120-80-9
benzene-1,2-diol

-
A
-
1020-31-1
3,5-Di-tert-butylcatechol

-
B
-
128-39-2
2,6-di-tert-butylphenol

-
C
-
2934-05-6
2,4-diisopropylphenol

-
D
-
2078-54-8
2,6-diisopropylphenol

-
E
-
2934-07-8
2,4,6-triisopropylphenol

-
F
-
4130-42-1
2,6-di-tert-butyl-4-ethylphenol

-
G
-
2444-28-2
2,6-di-tert-butyl-4-hydroxyphenol

-
H
-
87-97-8
2,6-di-tert-butyl-4-methoxymethylene-phenol

-
I
-
1138-52-9
3,5-Di-tert-butylphenol

-
J
-
1879-09-0
2,4-dimethyl-6-tert-butylphenol

-
K
-
52417-48-8
2-(2',5'-dimethoxyphenyl)propionaldehyde

-
L
-
17540-75-9
2,6-di-tert-butyl-4-sec-butylphenol

-
M
-
2050-46-6
1,2-diethoxybenzene

-
N
-
21112-37-8
1,4-dimethoxy-2-tert-butylbenzene

-
O
-
5076-72-2
1-ethoxy-4-methoxybenzene

-
P
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone

-
Q
-
120-95-6
2,4-di-tert-amylphenol

-
R
-
876-20-0
2,5-diethyl phenol

-
S
-
33963-27-8
4-ethoxy-3-methoxytoluene

-
T
-
59056-76-7
1,2-dimethoxy-4-butylbenzene

-
U
-
131358-04-8
1,2-diethoxy-4-ethyl-benzene

-
V
-
1620-98-0
3,5-di-t-butyl-4-hydroxybenzaldehyde

| Conditions | Yield |
|---|---|
| With ortho-tungstic acid at 300℃; for 6h; Autoclave; |

-
-
94-71-3
2-Ethoxyphenol

-
-
64-17-5
ethanol

-
A
-
1020-31-1
3,5-Di-tert-butylcatechol

-
B
-
128-39-2
2,6-di-tert-butylphenol

-
C
-
2934-05-6
2,4-diisopropylphenol

-
D
-
2078-54-8
2,6-diisopropylphenol

-
E
-
2934-07-8
2,4,6-triisopropylphenol

-
F
-
4130-42-1
2,6-di-tert-butyl-4-ethylphenol

-
G
-
2444-28-2
2,6-di-tert-butyl-4-hydroxyphenol

-
H
-
87-97-8
2,6-di-tert-butyl-4-methoxymethylene-phenol

-
I
-
1138-52-9
3,5-Di-tert-butylphenol

-
J
-
1879-09-0
2,4-dimethyl-6-tert-butylphenol

-
K
-
52417-48-8
2-(2',5'-dimethoxyphenyl)propionaldehyde

-
L
-
17540-75-9
2,6-di-tert-butyl-4-sec-butylphenol

-
M
-
2050-46-6
1,2-diethoxybenzene

-
N
-
21112-37-8
1,4-dimethoxy-2-tert-butylbenzene

-
O
-
5076-72-2
1-ethoxy-4-methoxybenzene

-
P
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone

-
Q
-
120-95-6
2,4-di-tert-amylphenol

-
R
-
876-20-0
2,5-diethyl phenol

-
S
-
33963-27-8
4-ethoxy-3-methoxytoluene

-
T
-
59056-76-7
1,2-dimethoxy-4-butylbenzene

-
U
-
131358-04-8
1,2-diethoxy-4-ethyl-benzene

-
V
-
1620-98-0
3,5-di-t-butyl-4-hydroxybenzaldehyde

| Conditions | Yield |
|---|---|
| With ortho-tungstic acid at 300℃; for 6h; Autoclave; |


| Conditions | Yield |
|---|---|
| With sodium hypochlorite; tetra(n-butyl)ammonium hydrogensulfate In ethyl acetate for 0.0833333h; Ambient temperature; | 78% |
| With ethanol; iron(III) chloride | |
| With thallium(III) trifluoroacetate |
-
-
64-17-5
ethanol

-
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone

-
-
7705-08-0
iron(III) chloride

-
-
4584-63-8
2,5-bis-(1,1-dimethylpropyl)-1,4-benzoquinone

-
-
79-74-3
2,5-di(tert-amyl)-1,4-hydroquinone

| Conditions | Yield |
|---|---|
| With 2,2-diphenyl-1-(2,4,6-trinitrophenyl)hydrazyl In tetrachloromethane; acetonitrile Kinetics; Further Variations:; Solvents; |

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran |
2,5-Di(tert-amyl)hydroquinone Chemical Properties
Molecular Formula: C16H26O2 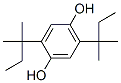
Formula Weight: 250.38
IUPAC Name: 2,5-Bis(2-methylbutan-2-yl)benzene-1,4-diol
Synonyms: 2,5-Bis(1,1-dimethylpropyl)hydroquinone ; 2,5-Di-tert-pentylbenzene-1,4-diol ; 4-Benzenediol,2,5-bis(1,1-dimethylpropyl)-1 ; 2,5-Di-tert-pentylbenzene-1,4-diol
Melting Point of 2,5-Di(tert-amyl)hydroquinone (79-74-3): 179 °C
Flash Point of 2,5-Di(tert-amyl)hydroquinone (79-74-3): 162.7 °C
Boiling Point: 364.8 °C at 760 mmHg
Density of 2,5-Di(tert-amyl)hydroquinone (79-74-3): 0.99 g/cm3
Vapour Pressure: 7.84E-06 mmHg at 25°C
Index of Refraction: 1.513
2,5-Di(tert-amyl)hydroquinone Toxicity Data With Reference
| 1. | orl-rat LD50:2 g/kg | IPSTB3 International Polymer Science and Technology. 3 (1976),93. | ||
| 2. | ipr-mus LD50:200 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD277-689 . | ||
| 3. | orl-rbt LD50:2 g/kg | IPSTB3 International Polymer Science and Technology. 3 (1976),93. |
2,5-Di(tert-amyl)hydroquinone Consensus Reports
Reported in EPA TSCA Inventory.
2,5-Di(tert-amyl)hydroquinone Safety Profile
Poison by intraperitoneal route. Low toxicity by ingestion. When heated to decomposition it emits acrid smoke and irritating fumes.
RTECS : MX6300000
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 79745-37-2
- 79746-17-1
- 79748-87-1
- 79752-03-7
- 79762-54-2
- 797-63-7
- 79-76-5
- 79768-40-4
- 79770-24-4
- 79770-89-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View