 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| In dichloromethane Irradiation; | 100% |
| In acetonitrile for 16h; Inert atmosphere; Reflux; | 100% |
| With water for 5h; Inert atmosphere; UV-irradiation; Sealed tube; chemoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 100% |
| In ethyl acetate at 0 - 5℃; for 3h; Solvent; Cooling with ice; Reflux; | 89% |
| In chloroform at 20℃; for 1h; Cooling with ice; | 85% |

| Conditions | Yield |
|---|---|
| With Phenyltrichlorosilane at 100 - 180℃; for 1h; | 99% |
| With phosphorus pentoxide | |
| With phosphorus pentoxide at 170℃; under 100 Torr; man fraktioniert das ueberdestillierte Phenylisocyanat; |
-
-
18100-80-6
2,3,5-triphenyl-4-thiazolone

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
A
-
20851-13-2
dimethyl 2,5-diphenylthiophene-3,4-dicarboxylate

-
B
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| In xylene Heating; | A 99% B n/a |


| Conditions | Yield |
|---|---|
| In toluene at -5 - 75℃; Solvent; | 98% |
| Stage #1: phosgene; n-butyl isocyanide In chlorobenzene at 81℃; for 2.33333h; Stage #2: aniline In chlorobenzene at 81 - 88℃; for 1h; | 76% |
| With 1-Chloronaphthalene at 230 - 240℃; unter Durchleiten durch ein Porzellanrohr; |
-
-
66702-57-6
2-dimethylamino-5-(4-nitro-phenyl)-4-oxo-3-phenyl-4,5-dihydro-thiazolium betaine

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
A
-
103-71-9
phenyl isocyanate

-
B
-
85013-66-7
2-Dimethylamino-5-(4-nitro-phenyl)-thiophene-3,4-dicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| In xylene Heating; | A n/a B 98% |

-
-
66702-60-1
2-cyano-5-(4-nitro-phenyl)-4-oxo-3-phenyl-4,5-dihydro-thiazolium betaine

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
A
-
103-71-9
phenyl isocyanate

-
B
-
85013-63-4
2-Cyano-5-(4-nitro-phenyl)-thiophene-3,4-dicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| In xylene Heating; | A n/a B 98% |

-
-
59208-07-0
5-(4-nitro-phenyl)-4-oxo-2,3-diphenyl-4,5-dihydro-thiazolium betaine

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
A
-
103-71-9
phenyl isocyanate

-
B
-
59086-14-5
2-(4-Nitro-phenyl)-5-phenyl-thiophene-3,4-dicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| In xylene Heating; | A n/a B 97% |


| Conditions | Yield |
|---|---|
| With air; palladium dichloride In 1,4-dioxane Heating; | 96% |
| With mercury(II) oxide at 170℃; | |
| Multi-step reaction with 3 steps 1: sodium hydride / 5,5-dimethyl-1,3-cyclohexadiene; mineral oil / 2 h / 20 °C / Inert atmosphere 2: chloroform / 1 h / 20 °C 3: acetonitrile / 25 °C View Scheme |

| Conditions | Yield |
|---|---|
| In chlorobenzene at 160 - 240℃; under 11251.1 Torr; for 1h; Pressure; Solvent; Temperature; Inert atmosphere; Green chemistry; | 94.9% |
-
-
66702-58-7
5-(4-nitro-phenyl)-4-oxo-3-phenyl-2-phenylsulfanyl-4,5-dihydro-thiazolium betaine

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
A
-
103-71-9
phenyl isocyanate

-
B
-
85013-62-3
2-(4-Nitro-phenyl)-5-phenylsulfanyl-thiophene-3,4-dicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| In xylene Heating; | A n/a B 94% |

-
-
56409-80-4
5-benzyl-3,4-dihydro-4-oxo-1,2,3-triphenylpyrimidin-1-ium-6-olate

-
-
2983-74-6
4-methoxyphenyl cyanate

-
A
-
103-71-9
phenyl isocyanate

-
B
-
85037-41-8
5-Benzyl-6-(4-methoxy-phenoxy)-2,3-diphenyl-3H-pyrimidin-4-one

-
C
-
25940-64-1
2,4,6-tris(4-methoxyphenoxy)-1,3,5-triazine

| Conditions | Yield |
|---|---|
| In chlorobenzene for 29h; Heating; | A n/a B 93% C n/a |


| Conditions | Yield |
|---|---|
| at 750℃; under 0.002 Torr; | A 93% B n/a |

| Conditions | Yield |
|---|---|
| In chloroform at 60℃; for 18h; | 92% |
-
-
56409-80-4
5-benzyl-3,4-dihydro-4-oxo-1,2,3-triphenylpyrimidin-1-ium-6-olate

-
-
1122-85-6
phenyl cyanate

-
A
-
1919-48-8
2,4,6-triphenoxy-1,3,5-triazine

-
B
-
103-71-9
phenyl isocyanate

-
C
-
85037-39-4
5-Benzyl-6-phenoxy-2,3-diphenyl-3H-pyrimidin-4-one

| Conditions | Yield |
|---|---|
| In chlorobenzene for 18h; Heating; | A n/a B n/a C 92% |

-
-
61522-23-4
2-cyano-4-oxo-3,5-diphenyl-4,5-dihydro-thiazolium betaine

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
A
-
103-71-9
phenyl isocyanate

-
B
-
85013-64-5
2-Cyano-5-phenyl-thiophene-3,4-dicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| In xylene Heating; | A n/a B 92% |


-
A
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| at 220℃; for 0.166667h; | A n/a B 92% |

-
A
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| at 150℃; for 0.166667h; | A n/a B 92% |

| Conditions | Yield |
|---|---|
| With Phenyltrichlorosilane at 85 - 184℃; for 1h; | 90.8% |
| With phosphorus pentachloride at 120 - 140℃; for 3h; | 68% |
| With boron trichloride; triethylamine In benzene for 0.5h; Heating; | 93 % Chromat. |

| Conditions | Yield |
|---|---|
| With pyridine; di(rhodium)tetracarbonyl dichloride; pyridine hydrochloride In chlorobenzene at 205℃; under 50 Torr; for 1h; | 90% |
| With di(rhodium)tetracarbonyl dichloride; pyridine hydrochloride In chlorobenzene at 190℃; under 38000 Torr; for 1h; | 80% |
| Stage #1: nitrobenzene at 120℃; under 75007.5 Torr; for 4h; Inert atmosphere; Autoclave; Stage #2: carbon monoxide Catalytic behavior; Temperature; Reagent/catalyst; Inert atmosphere; Autoclave; | 72.2% |
-
-
109853-15-8
C16H16N4OS2

-
A
-
103-71-9
phenyl isocyanate

-
B
-
13281-49-7
3-Mercapto-1-methyl-5-phenyl-1,2,4-triazol

| Conditions | Yield |
|---|---|
| N-benzyl-N,N,N-triethylammonium chloride In sodium hydroxide; dichloromethane for 12h; Ambient temperature; | A n/a B 89% |
-
-
77219-88-6
bis(trimethylsilyl)benzohydroxamic acid

-
A
-
107-46-0
Hexamethyldisiloxane

-
B
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| at 120℃; | A n/a B 88% |

-
A
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| In benzene for 2h; Heating; | A n/a B 88% |

| Conditions | Yield |
|---|---|
| at 750℃; under 0.002 Torr; Mechanism; regioselectivity, other substituted 1-phenylazetidin-2-ones, var. temp.; | A n/a B 88% |
| at 750℃; under 0.002 Torr; | A n/a B 88% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.0833333h; Ambient temperature; | 87% |

| Conditions | Yield |
|---|---|
| With Phenyltrichlorosilane at 55 - 197℃; for 2h; | 86% |
| With Phenyltrichlorosilane at 55 - 197℃; for 2h; Product distribution; var. silanes, var. time, var. temp.; | 86% |
| With Phenyltrichlorosilane at 100 - 130℃; for 2h; | 81.9% |
-
-
67723-48-2
N-Benzoyl-N,O-bis(trimethylsilyl)hydroxylamine

-
A
-
1821-33-6
N-phenyl-N'-benzoylurea

-
B
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| In decalin at 160℃; for 0.0833333h; | A 5.7% B 86% |
-
-
22513-32-2
acid sodium salt of benzohydroxamic acid

-
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| With Phenyltrichlorosilane at 130 - 140℃; for 2h; | 86% |
| With Phenyltrichlorosilane at 130 - 140℃; for 2h; | 85.8% |

-
A
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| at 220℃; for 0.166667h; | A n/a B 85% |

| Conditions | Yield |
|---|---|
| at 0 - 20℃; for 48.3333h; | 100% |
| In diethyl ether at 20℃; for 18h; | 99.2% |
| In diethyl ether at 20℃; for 0.75h; Addition; | 97% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; | 100% |
| In 1,4-dioxane at 0 - 20℃; | 87% |
| In diethyl ether for 3h; Heating; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: tryptamine; phenyl isocyanate In dichloromethane at 60℃; Stage #2: With isatoic anhydride-N-(CH2)3-C8F17 In dichloromethane at 60℃; for 2.5h; | 100% |
| In dichloromethane at 60℃; for 5h; Inert atmosphere; Sealed reaction vessel; | 98% |
-
-
34803-66-2
1-(2-pyridyl)piperazine

-
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| Stage #1: 1-(2-pyridyl)piperazine; phenyl isocyanate In dichloromethane at 60℃; Stage #2: With isatoic anhydride-N-(CH2)3-C8F17 In dichloromethane at 60℃; for 2.5h; | 100% |

| Conditions | Yield |
|---|---|
| With cerium(IV) oxide; carbon dioxide at 170℃; under 30003 Torr; for 12h; Inert atmosphere; Autoclave; | 100% |
| With dibutyltin dilaurate at 50℃; for 3h; | 100% |
| at 80℃; for 0.5h; Inert atmosphere; | 98% |
-
-
103-71-9
phenyl isocyanate

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
105902-61-2
3-methylbut-2-en-1-yl phenylcarbamate

| Conditions | Yield |
|---|---|
| In acetonitrile at 70℃; for 23h; Inert atmosphere; | 100% |
| With triethylamine In dichloromethane at 20℃; Inert atmosphere; | 88% |
| With triethylamine In dichloromethane at 20℃; | 88% |
-
-
2216-51-5
(-)-menthol

-
-
103-71-9
phenyl isocyanate

-
-
637336-84-6
phenylcarbamic acid (1R,2S,5R)-2-isopropyl-5-methylcyclohexyl ester

| Conditions | Yield |
|---|---|
| With MoCl2O2(dmf)2 In dichloromethane at 20℃; for 0.333333h; | 100% |
| With MoCl2O2(dmf)2 In dichloromethane at 20℃; for 0.333333h; Reagent/catalyst; Inert atmosphere; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 90h; | 100% |
| With MoCl2O2(dmf)2 In dichloromethane at 20℃; for 0.333333h; | 100% |
| With MoCl2O2(dmf)2 In dichloromethane at 20℃; for 0.333333h; Inert atmosphere; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: phenyl isocyanate; N-butylamine In dichloromethane at 60℃; Stage #2: With isatoic anhydride-N-(CH2)3-C8F17 In dichloromethane at 60℃; for 2.5h; | 100% |
| at 20℃; | 98% |
| In hexane at 25℃; Cooling with ice; | 98% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 100% |
| In acetic acid for 0.0833333h; | 98% |
| In hexane | 95% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 0.166667h; | 100% |
| With diethyl ether |
-
-
103-71-9
phenyl isocyanate

-
-
115-19-5
2-methyl-but-3-yn-2-ol

-
-
6289-19-6
O-α,α-dimethylpropargyl N-phenylcarbamate

| Conditions | Yield |
|---|---|
| With MoCl2O2(dmf)2 In dichloromethane at 20℃; for 0.333333h; | 100% |
| With triethylamine In tetrahydrofuran at 70℃; for 12h; Inert atmosphere; | 94% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 90h; | 88% |
-
-
103-71-9
phenyl isocyanate

-
-
104-84-7
para-methylbenzylamine

-
-
35305-46-5
N-(4-methylbenzyl)-N’-phenylurea

| Conditions | Yield |
|---|---|
| Stage #1: phenyl isocyanate; para-methylbenzylamine In dichloromethane at 60℃; Stage #2: With isatoic anhydride-N-(CH2)3-C8F17 In dichloromethane at 60℃; for 2.5h; | 100% |

| Conditions | Yield |
|---|---|
| chlorodi-(n-butyl)tin acetate In chloroform for 0.833333h; Product distribution; Mechanism; Ambient temperature; catalytic activity, various tin(IV) catalysts; | 100% |
| chlorodi-(n-butyl)tin acetate In chloroform for 0.833333h; Ambient temperature; | 100% |
| With diallyltin(IV)di(2-ethyl hexanoate) In dichloromethane for 0.0833333h; Product distribution; Heating; other alcohols and isocyanates; var. temp. and time; | 93% |

| Conditions | Yield |
|---|---|
| at 20℃; for 2h; | 100% |
| With cesium fluoride | 93.1% |
| With triethylamine In acetonitrile for 24.5h; Esterification; | 89% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 70℃; under 6000480 Torr; for 20h; | 100% |
| With sodium nitrite for 0.166667h; Cyclization; microwave irradiation; | 99% |
| With 1,3-bis(2,6-diisopropylphenyl)dihydroimidazol-2-ylidene In tetrahydrofuran at 20℃; for 1h; | 99% |
-
-
1483-24-5
1-methyl-1-benzoylhydrazine

-
-
103-71-9
phenyl isocyanate

-
-
13136-26-0
1-Benzoyl-1-methyl-4-phenylsemicarbazid

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Heating; | 100% |
| In diethyl ether | |
| In ethanol |
-
-
103-71-9
phenyl isocyanate

-
-
78-27-3
1-Ethynyl-1-cyclohexanol

-
-
73623-16-2
1-ethynylcyclohexyl phenylcarbamate

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 90h; | 100% |
| In 1,2-dichloro-ethane at 20℃; for 16h; | 82% |
| With 1-methyl-pyrrolidin-2-one |
-
-
103-71-9
phenyl isocyanate

-
-
30389-18-5
1-Ethynylcyclohexylamine

-
-
42785-83-1
1-(1-ethynylcyclohexyl)-3-phenylurea

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 100% |
| In diethyl ether |
-
-
103-71-9
phenyl isocyanate

-
-
111-42-2
2,2'-iminobis[ethanol]

-
-
20074-78-6
1,1-[Di-(2-hydroxyethyl)]-3-phenylurea

| Conditions | Yield |
|---|---|
| In dichloromethane | 100% |
| In benzene | |
| In dichloromethane; toluene at 20℃; | |
| In dichloromethane at 20℃; for 0.0166667h; Inert atmosphere; | 1.14 g |
-
-
2167-39-7
(+/-)-2-methyloxetane

-
-
103-71-9
phenyl isocyanate

-
-
99855-05-7
6-methyl-3-phenyl-1,3-oxazinan-2-one

| Conditions | Yield |
|---|---|
| With N,N,N,N,N,N-hexamethylphosphoric triamide; diphenyltin diiodide at 80℃; for 3h; sealed tube; | 100% |
| With tetraphenyl stibonium iodide In tetrahydrofuran at 60℃; for 23h; | 27% |

| Conditions | Yield |
|---|---|
| With tetraphenyl stibonium iodide In dichloromethane at 40℃; | 100% |
| tetraphenyl stibonium iodide In dichloromethane at 45℃; for 1h; | 100% |
| tetraphenyl stibonium iodide In dichloromethane at 45℃; for 1h; Heating; | 84% |

| Conditions | Yield |
|---|---|
| In pyridine; ethyl acetate | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane | 100% |
-
-
25355-53-7
1-methyl-6-methylsulfanyl-1,2,3,4-tetrahydro-pyridine

-
-
103-71-9
phenyl isocyanate

-
-
81197-49-1
1-Methyl-2-methylsulfanyl-1,4,5,6-tetrahydro-pyridine-3-carboxylic acid phenylamide

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |
| In diethyl ether for 4h; Ambient temperature; Yield given; |

| Conditions | Yield |
|---|---|
| for 1h; | 100% |
| for 1h; Heating; | 100% |
| at 20℃; | 92% |

| Conditions | Yield |
|---|---|
| 100% |
-
-
2935-90-2
Methyl 3-mercaptopropionate

-
-
103-71-9
phenyl isocyanate

-
-
77585-81-0
3-(N-Phenylcarbamoylmercapto)-propionsaeuremethylester

| Conditions | Yield |
|---|---|
| With N-benzyl-trimethylammonium hydroxide In diethyl ether at 40℃; for 0.5h; | 100% |
-
-
1660-24-8
1-benzoyl-2-methylhydrazine

-
-
103-71-9
phenyl isocyanate

-
-
77919-29-0
1-Benzoyl-2-methyl-4-phenylsemicarbazid

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Heating; | 100% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

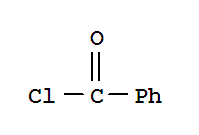














 T+
T+