-
Name
2',6'-Dihydroxyacetophenone
- EINECS 211-833-6
- CAS No. 699-83-2
- Article Data27
- CAS DataBase
- Density 1.291 g/cm3
- Solubility sparingly soluble in water
- Melting Point 156-158 °C(lit.)
- Formula C8H8O3
- Boiling Point 262.8 °C at 760 mmHg
- Molecular Weight 152.15
- Flash Point 127 °C
- Transport Information
- Appearance yellowish-beige powder
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Acetophenone,2,6-dihydroxy- (3CI);Acetophenone, 2',6'-dihydroxy- (7CI,8CI);1,3-Benzenediol, 2-acetyl-;1-(2,6-Dihydroxyphenyl)ethanone;2-Acetylbenzene-1,3-diol;2-Acetylresorcinol;Ethanone,1-(2,6-dihydroxyphenyl)-;NSC615;
- PSA 57.53000
- LogP 1.30040
Synthetic route
-
-
144152-28-3
2,6-diacetoxyacetophenone

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| With water; sodium acetate In ethanol for 5h; Reflux; | 92% |

| Conditions | Yield |
|---|---|
| With hydrogen fluoride supported on silica gel In neat (no solvent) at 55℃; for 4h; Green chemistry; | 60% |
| With Fluoro Flash In neat (no solvent) at 80℃; for 4h; Catalytic behavior; Fries Phenol Ester Rearrangement; Green chemistry; |
-
-
30992-74-6
4,5-dihydroxy-2H-benzopyran-2-one

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With aluminium trichloride; chlorobenzene auf Siedetemperatur; | |
| With aluminium trichloride; toluene at 120℃; |
-
-
62970-17-6
3-acetyl-2,4-dihydroxy-benzoic acid methyl ester

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| With sodium hydroxide |
-
-
52956-20-4
2-acetyl-2-chloro-1,3-cyclohexanedione

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In N,N-dimethyl-formamide | |
| (i) HCl, DMF, (ii) aq. HCl; Multistep reaction; | |
| With hydrogenchloride In d7-N,N-dimethylformamide at 120 - 130℃; for 0.333333h; Yield given; |
-
-
108-24-7
acetic anhydride

-
-
141-97-9
ethyl acetoacetate

-
-
108-46-3
recorcinol

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| Multistep reaction; |

-
-
37514-00-4
2-acetyl-1,3-cyclohexanedione enol

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| With mercury(II) diacetate; sodium acetate In acetic acid |
-
-
7446-70-0
aluminium trichloride

-
-
2040-04-2
2,6-dimethoxyacetophenone

-
-
108-90-7
chlorobenzene

-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
7446-70-0
aluminium trichloride

-
-
2040-04-2
2,6-dimethoxyacetophenone

-
-
108-88-3
toluene

-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
16932-49-3
2,6-dimethoxybenzonitrile

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: toluene; diethyl ether / Erhitzen des vom Aether befreiten Reaktionsgemisches auf Siedetemperatur,anshcliessendes Behandeln mit Eis und wss.HCl und Erwaermen der erhaltenen wss.Loesung 2: AlCl3; chlorobenzene / auf Siedetemperatur View Scheme | |
| Multi-step reaction with 2 steps 1: toluene; diethyl ether / Erhitzen des vom Aether befreiten Reaktionsgemisches auf Siedetemperatur,anshcliessendes Behandeln mit Eis und wss.HCl und Erwaermen der erhaltenen wss.Loesung 2: AlCl3; toluene / 120 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aluminium chloride / 215 °C 2: aqueous KOH View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 250 °C 2: aluminium chloride / 215 °C 3: aqueous KOH View Scheme |
-
-
75-36-5
acetyl chloride

-
-
95-50-1
1,2-dichloro-benzene

-
-
108-46-3
recorcinol

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; aluminium trichloride |

-
-
4056-73-9
2-acetyl-1,3-cyclohexanedione

-
-
7783-06-4
hydrogen sulfide

-
-
699-83-2
2,6-dihydroxylacetophenone

| Conditions | Yield |
|---|---|
| In toluene | 0.91 g (59 mole %) |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 0 - 20℃; for 12h; Inert atmosphere; |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
109-70-6
1,3-chlorobromopropane

-
-
105277-74-5
2-(3-chlorophenoxy)-6-hydroxyacetophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 24h; Heating; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 4h; | 46% |
| With potassium carbonate In acetone |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
149071-05-6
3-methyl-4-hydroxyindazole

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethylene glycol at 20 - 160℃; for 2.5h; | 100% |
| With hydrazine hydrate In ethylene glycol at 160℃; for 2h; Cyclization; | 68% |
| With hydrazine In ethylene glycol at 20 - 160℃; for 2.33333h; | 68% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
100-44-7
benzyl chloride

-
-
3886-19-9
1-[2',6'-bis(phenylmethoxy)phenyl]ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 65 - 100℃; for 18h; | 99% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
609-65-4
o-chlorobenzoyl chloride

-
-
920006-37-7
2',6'-bis(2-chlorobenzoyloxy)acetophenone

| Conditions | Yield |
|---|---|
| With pyridine; dmap at 20℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-dihydroxylacetophenone With lithium diisopropyl amide In tetrahydrofuran Stage #2: chloro-trimethyl-silane In tetrahydrofuran | 99% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
74-88-4
methyl iodide

-
-
703-23-1
2'-hydroxy-6'-methoxyacetophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 4h; Heating; | 98% |
| With potassium carbonate In acetone for 8h; Reflux; | 97% |
| With potassium carbonate In acetone at 55℃; for 8h; | 95% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
122-04-3
4-nitro-benzoyl chloride

-
-
26505-98-6
2',6'-bis(4-nitrobenzoyloxy)acetophenone

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 12h; | 98% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
107-30-2
chloromethyl methyl ether

-
-
78646-28-3
2′-hydroxy-6′-methoxymethoxyacetophenone

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane for 4.5h; | 97.8% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane for 0.5h; | 95% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; | 92.8% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
77-78-1
dimethyl sulfate

-
-
703-23-1
2'-hydroxy-6'-methoxyacetophenone

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-dihydroxylacetophenone With potassium carbonate In acetone for 0.166667h; Stage #2: dimethyl sulfate In acetone for 6h; Reflux; | 96% |
| With potassium carbonate In acetone for 2h; Inert atmosphere; Reflux; | 96% |
| With potassium carbonate In acetone for 0.333333h; Reflux; | 95% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
466635-70-1
2,6-bis(tert-butyldimethylsilyloxy)acetophenone

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 60℃; for 20h; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: 6-methylheptan-2-one With pyrrolidine; butyric acid In dimethyl sulfoxide for 0.25h; Kabe Chromanone Synthesis; Stage #2: 2,6-dihydroxylacetophenone In dimethyl sulfoxide at 20℃; for 3h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Kabe Chromanone Synthesis; | 95% |
| With pyrrolidine In toluene Dean-Stark; Reflux; |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
100-07-2
4-methoxy-benzoyl chloride

-
-
58124-15-5
2',6'-bis(4-methoxybenzoyloxy)acetophenone

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 12h; | 94% |
| With pyridine | |
| With pyridine for 2h; |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 0 - 23℃; for 3h; Inert atmosphere; | 94% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
572-09-8
2,3,4,6-tetra-O-acetyl-α-D-glucopyranosyl bromide

-
-
23141-00-6
2',6'-dihydroxyacetophenone 2'-O-(2,3,4,6-tetra-O-acetyl-β-D-glucopyranoside)

| Conditions | Yield |
|---|---|
| With benzyltri(n-butyl)ammonium chloride; potassium carbonate In chloroform at 20℃; for 24h; glycosylation; | 93% |
| With benzyltri(n-butyl)ammonium chloride; potassium carbonate In chloroform; water at 20℃; for 27h; | 9% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran 0 deg C to room temp., 0.5 to 1 h; | 92% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
122-01-0
4-chloro-benzoyl chloride

-
-
502916-43-0
2',6'-bis(4-chlorobenzoyloxy)acetophenone

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 12h; | 92% |
| With pyridine for 2h; |

| Conditions | Yield |
|---|---|
| With 4-pyrrolidin-1-ylpyridine; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 0.5h; | 92% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
1193107-75-3
1-(2-((tert-butyldimethylsilyl)oxy)-6-hydroxyphenyl)ethanone

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 0 - 20℃; for 13h; Inert atmosphere; | 92% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
109-94-4
formic acid ethyl ester

-
-
3952-69-0
5-hydroxy-4H-1-benzopyran-4-one

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-dihydroxylacetophenone; formic acid ethyl ester With sodium ethanolate at 0 - 20℃; for 18h; Schlenk technique; Inert atmosphere; Stage #2: With hydrogenchloride In methanol; water at 0 - 20℃; for 24h; | 92% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
87953-93-3
3-chloro-2.6-dihydroxyacetophenone

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride In acetic acid for 2h; Heating; | 90.5% |
| With N-chloro-succinimide; acetic acid at 50℃; for 2h; | 81.5% |
| With thionyl chloride In acetic acid | |
| With thionyl chloride In acetic acid | |
| With N-chloro-succinimide; acetic acid at 50℃; for 2h; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium cyanoborohydride In methanol | 90% |
| With triethylsilane In trifluoroacetic acid at 20℃; for 3h; Inert atmosphere; | 81% |
| With triethylsilane; trifluoroacetic acid for 3h; | 78% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
150942-98-6
t-butyl lithioacetate

| Conditions | Yield |
|---|---|
| In toluene at 100℃; for 2h; | 90% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
100-46-9
benzylamine

-
-
82488-60-6
2,6-Dihydroxyphenylethylidenebenzylamine

| Conditions | Yield |
|---|---|
| In toluene Heating; or ZnCl2, 1) 30 min, 160 deg C, 2) 5 min, 180 deg C; | 90% |
| In toluene Heating; | 90% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
1201-99-6, 20595-45-3, 20595-46-4
trans-2,4-dichlorocinnamic acid

| Conditions | Yield |
|---|---|
| With 4-pyrrolidin-1-ylpyridine; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 0.5h; | 90% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
105-36-2
ethyl bromoacetate

-
-
6769-65-9
ethyl 2-(2-acetyl-3-hydroxyphenoxy)acetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 60℃; for 2h; | 89% |
| With potassium carbonate In N,N-dimethyl-formamide for 3h; Ambient temperature; | 80% |
| With potassium carbonate In acetone for 3h; Reflux; | 61% |
| With potassium carbonate; acetone | |
| With potassium carbonate In acetone at 60℃; for 15h; |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
100-39-0
benzyl bromide

-
-
3886-19-9
1-[2',6'-bis(phenylmethoxy)phenyl]ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 2.5h; | 89% |
| With potassium carbonate; sodium iodide In acetone for 18h; Heating; | 46% |
| With sodium iodide; potassium carbonate In acetone | |
| With potassium carbonate In dimethyl sulfoxide at 20℃; |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
100-44-7
benzyl chloride

-
-
4047-24-9
1-[2-(benzyloxy)-6-hydroxyphenyl]ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 0 - 70℃; Inert atmosphere; | 89% |
| With potassium carbonate In N,N-dimethyl-formamide for 24h; | 78% |
| With potassium hydroxide; potassium carbonate In butanone for 6h; Heating; | 68% |
| With potassium carbonate In N,N-dimethyl-formamide |

| Conditions | Yield |
|---|---|
| With 4-pyrrolidin-1-ylpyridine; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 0.5h; | 89% |
-
-
699-83-2
2,6-dihydroxylacetophenone

-
-
143-15-7
1-dodecylbromide

-
-
1393845-50-5
1-(2-(dodecyloxy)-6-hydroxyphenyl)ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In acetonitrile at 70 - 80℃; | 88% |
2',6'-Dihydroxyacetophenone Chemical Properties
Molecule structure of 2',6'-Dihydroxyacetophenone (CAS NO.699-83-2):
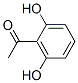
IUPAC Name: 1-(2,6-Dihydroxyphenyl)ethanone
Molecular Weight: 152.14732 g/mol
Molecular Formula: C8H8O3
Density: 1.291 g/cm3
Melting Point: 156-158 °C(lit.)
Boiling Point: 262.8 °C at 760 mmHg
Flash Point: 127 °C
Index of Refraction: 1.595
Molar Refractivity: 40.04 cm3
Molar Volume: 117.8 cm3
Surface Tension: 56.1 dyne/cm
Enthalpy of Vaporization: 52.08 kJ/mol
Vapour Pressure: 0.00656 mmHg at 25 °C
Solubility Dioxane: 50 mg/mL, clear
Water Solubility: sparingly soluble
XLogP3-AA: 1.4
H-Bond Donor: 2
H-Bond Acceptor: 3
Rotatable Bond Count: 1
Tautomer Count: 12
Exact Mass: 152.047344
MonoIsotopic Mass: 152.047344
Topological Polar Surface Area: 57.5
Heavy Atom Count: 11
Canonical SMILES: CC(=O)C1=C(C=CC=C1O)O
InChI: InChI=1S/C8H8O3/c1-5(9)8-6(10)3-2-4-7(8)11/h2-4,10-11H,1H3
InChIKey: YPTJKHVBDCRKNF-UHFFFAOYSA-N
EINECS: 211-833-6
Product Categories: FINE Chemical & INTERMEDIATES; Cromoglyn Sodium; Acetophenone series
2',6'-Dihydroxyacetophenone Uses
2',6'-Dihydroxyacetophenone (CAS NO.699-83-2) is used in organic synthesis. It is also used as pharmaceutical intermediates.
2',6'-Dihydroxyacetophenone Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-24/25
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S24/25:Avoid contact with skin and eyes.
WGK Germany: 3
Hazard Note: Irritant
HS Code: 29145000
2',6'-Dihydroxyacetophenone Specification
2',6'-Dihydroxyacetophenone (CAS NO.699-83-2) is also named as 1,3-Benzenediol, 2-acetyl- ; 2,6-Dihydroxyacetophenone ; 2-Acetylresorcinol ; NSC 615 ; Resorcinol, 2-acetyl- ; gamma-Resacetophenone ; Acetophenone, 2',6'-dihydroxy- (8CI) ; Ethanone, 1-(2,6-dihydroxyphenyl)- (9CI) . 2',6'-Dihydroxyacetophenone (CAS NO.699-83-2) is yellowish-beige powder.
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 69984-73-2
- 6998-83-0
- 699-90-1
- 69991-61-3
- 69991-62-4
- 69991-67-9
- 6999-19-5
- 699-97-8
- 69998-58-9
- 699-98-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View