-
Name
2,6-Dichloro-4-nitrophenol
- EINECS 210-563-6
- CAS No. 618-80-4
- Article Data40
- CAS DataBase
- Density 1.682 g/cm3
- Solubility
- Melting Point 123-126 °C (dec.)
- Formula C6H3Cl2NO3
- Boiling Point 285.2 °C at 760 mmHg
- Molecular Weight 208.001
- Flash Point 126.3 °C
- Transport Information UN 2811 6.1/PG 1
- Appearance yellow crystalline powder
- Safety 26-36-36/37/39
- Risk Codes 20/21/22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 4-Nitro-2,6-dichlorophenol;NSC 4123;phenol, 2,6-dichloro-4-nitro-;
- PSA 66.05000
- LogP 3.13040
Synthetic route

| Conditions | Yield |
|---|---|
| With Zn(NO3)2*2N2O4 In dichloromethane at 20℃; for 0.0833333h; | 98% |
| With tetra(n-butyl)ammonium dichromate(VI); sodium nitrite In dichloromethane for 23h; Reflux; chemoselective reaction; | 98% |
| With Tetraethylene glycol; silica gel; dinitrogen tetraoxide In dichloromethane at 20℃; for 0.0833333h; | 97% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; chlorine In water at 10℃; for 3.5h; Solvent; Temperature; | 93% |
| With sodium periodate; sulfuric acid; sodium dodecyl-sulfate; acetic acid; sodium chloride In water at 40℃; for 2h; | 93% |
| With trichloroisocyanuric acid; brilliant green carbocation In acetonitrile at 20℃; for 0.25h; Irradiation; regioselective reaction; | 58% |
-
-
1620783-12-1
2-(2,6-dichloro-4-nitrophenoxy)pyridine

-
-
618-80-4
2,6-dichloro-4-nitrophenol

| Conditions | Yield |
|---|---|
| Stage #1: 2-(2,6-dichloro-4-nitrophenoxy)pyridine With methyl trifluoromethanesulfonate In toluene at 100℃; for 2h; Inert atmosphere; Schlenk technique; Stage #2: With sodium In methanol at 80℃; for 0.5h; Inert atmosphere; Schlenk technique; | 77% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With acetic acid; sodium nitrite |

| Conditions | Yield |
|---|---|
| With water; chlorine |
-
-
25319-98-6
3,5-dichloro-4-hydroxybenzene sulfonic acid

-
-
618-80-4
2,6-dichloro-4-nitrophenol

| Conditions | Yield |
|---|---|
| beim Nitrieren; |
-
-
381-73-7
Difluoroacetic acid

-
-
46063-82-5
2,6-dichloro-4-nitro-phenolate

-
A
-
618-80-4
2,6-dichloro-4-nitrophenol

-
B
-
83193-04-8
2,2-difluoroacetate

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 20℃; Equilibrium constant; Rate constant; |

-
-
46063-82-5
2,6-dichloro-4-nitro-phenolate

-
-
18716-27-3
tribenzylamine; protonated form

-
A
-
618-80-4
2,6-dichloro-4-nitrophenol

-
B
-
620-40-6
tribenzylamine

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 20℃; Equilibrium constant; Rate constant; |


| Conditions | Yield |
|---|---|
| With water; copper(II) ion at 25℃; Rate constant; MES buffer - pH 6.3; |
-
-
58107-26-9
3,5-dibromo-4-fluorophenol

-
A
-
618-80-4
2,6-dichloro-4-nitrophenol

| Conditions | Yield |
|---|---|
| In chloroform at 20℃; Equilibrium constant; |

-
-
100-02-7
4-nitro-phenol

-
-
7782-50-5
chlorine

-
A
-
618-80-4
2,6-dichloro-4-nitrophenol

-
B
-
619-08-9
2-chloro-4-nitrophenol

-
-
7647-01-0
hydrogenchloride

-
-
100-02-7
4-nitro-phenol

-
A
-
618-80-4
2,6-dichloro-4-nitrophenol

-
B
-
619-08-9
2-chloro-4-nitrophenol

-
C
-
118-75-2
chloranil

-
-
616-59-1
2-hydroxyl-5-nitrobenzenesulfonic acid

-
-
7732-18-5
water

-
-
7782-50-5
chlorine

-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
3217-15-0
4-bromo-2,6-dichloro-phenol

-
-
7697-37-2
nitric acid

-
-
64-19-7
acetic acid

-
A
-
618-80-4
2,6-dichloro-4-nitrophenol

-
B
-
946-31-6
2-chloro-4,6-dinitro-phenol

-
-
25319-98-6
3,5-dichloro-4-hydroxybenzene sulfonic acid

-
-
7697-37-2
nitric acid

-
-
618-80-4
2,6-dichloro-4-nitrophenol

| Conditions | Yield |
|---|---|
| Reaktion des Kaliumsalzes; entsteht zunaechst; |
-
-
67-56-1
methanol

-
-
104678-53-7
2,6-dichloro-4-methyl-4-nitrocyclohexa-2,5-dienone

-
A
-
618-80-4
2,6-dichloro-4-nitrophenol

-
B
-
624-91-9
methyl nitrite

-
C
-
79817-03-1
3,5-dichloro-4-hydroxybenzyl methyl ether


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: lime/chalk/ 2: beim Nitrieren View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: KOH; chlorine 2: lime/chalk/ 3: beim Nitrieren View Scheme |
-
-
20098-48-0
3,4,5-trichloronitrobenzen

-
-
618-80-4
2,6-dichloro-4-nitrophenol

| Conditions | Yield |
|---|---|
| In diethylene glycol | |
| With potassium phenolate In diethylene glycol at 160℃; for 15h; | |
| With potassium phenolate In diethylene glycol at 160℃; for 15h; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: N-chloro-succinimide; palladium diacetate; toluene-4-sulfonic acid / ethyl acetate / 6 h / Inert atmosphere; Schlenk technique; Heating 2.1: methyl trifluoromethanesulfonate / toluene / 2 h / 100 °C / Inert atmosphere; Schlenk technique 2.2: 0.5 h / 80 °C / Inert atmosphere; Schlenk technique View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: chlorine / methanol / 55 - 60 °C 2.1: sulfuric acid; nitrosylsulfuric acid / toluene / 1 h / 5 - 15 °C 2.2: 7 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-2,6-dichloroaniline With nitrosylsulfuric acid; sulfuric acid In toluene at 5 - 15℃; for 1h; Stage #2: With sulfuric acid; water; copper(II) sulfate In toluene for 7h; Reflux; |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
525584-77-4
trifluoro-methanesulfonic acid 2,6-dichloro-4-nitrophenyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at -40 - -10℃; | 99% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In water at 110℃; Sealed tube; Green chemistry; | 98% |
| With sodium tetrahydroborate In tetrahydrofuran; water at 20℃; for 3h; | 98% |
| With palladium 10% on activated carbon; hydrogen In methanol at 50℃; under 7500.75 Torr; for 2h; Reagent/catalyst; Pressure; Solvent; Autoclave; | 96.21% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
51356-03-7
4,5-dichloro-2-chloromethyl-pyridazin-3(2H)-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol for 20h; Reflux; regioselective reaction; | 95% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
572-09-8
2,3,4,6-tetra-O-acetyl-α-D-glucopyranosyl bromide

-
-
30361-06-9
2',6'-Dichloro-4'-nitrophenyl 2,3,4,6-tetra-O-acetyl-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With silver(l) oxide In acetonitrile at 40℃; | 93.9% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; | 93.5% |

| Conditions | Yield |
|---|---|
| Stage #1: phenylacetic acid With oxalyl dichloride In dichloromethane; N,N-dimethyl-formamide for 1h; Inert atmosphere; Stage #2: 2,6-dichloro-4-nitrophenol With N-ethyl-N,N-diisopropylamine In dichloromethane; N,N-dimethyl-formamide at 20℃; for 12h; Inert atmosphere; | 89% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
3068-32-4
1-bromo-1-deoxy-2,3,4,6-tetra-O-acetyl-a-D-galactopyranoside

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In dichloromethane at 35℃; for 48h; | 86% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
16420-13-6
N,N-Dimethylthiocarbamoyl chloride

-
-
74875-14-2
O-(2,6-dichloro-4-nitrophenyl) dimethylcarbamothioate

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In N,N-dimethyl-formamide at 25℃; for 18h; | 75% |
| With sodium hydride 1.) DMF, 40 min, 2.) DMF, from 5 to 70 deg C, 40 min; Yield given. Multistep reaction; | |
| Stage #1: 2,6-dichloro-4-nitrophenol With sodium hydride In N,N-dimethyl-formamide at 20℃; for 1h; Stage #2: N,N-Dimethylthiocarbamoyl chloride In N,N-dimethyl-formamide at 20℃; for 16h; | |
| With 1,4-diaza-bicyclo[2.2.2]octane In N,N-dimethyl-formamide at 25℃; for 18h; |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
106-89-8
epichlorohydrin

-
-
95646-29-0
1-<2',6'-dichloro-4'-nitrophenoxy->-2,3-epoxy propane

| Conditions | Yield |
|---|---|
| With pyridine at 90 - 95℃; for 1h; | 75% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
141-78-6
ethyl acetate

-
-
109384-19-2
t-butyl 4-hydroxy piperidine-1-carboxylate

-
-
1972-28-7
diethylazodicarboxylate

-
-
337520-58-8
4-(1-t-Butoxycarbonylpiperidin-4-yloxy)-3,5-dichloronitrobenzene

| Conditions | Yield |
|---|---|
| With triphenylphosphine In hexane; dichloromethane | 72% |


| Conditions | Yield |
|---|---|
| With dmap; diisopropylamine In tetrachloromethane; acetonitrile at -10℃; for 2h; | 71% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
106-95-6
allyl bromide

-
-
95646-26-7
2,6-dichloro-4-nitro-(2-allyl)-oxybenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In N,N-dimethyl-formamide at 80℃; for 3h; | 66% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 48h; Ambient temperature; | 63% |

| Conditions | Yield |
|---|---|
| Stage #1: dysprosium(III) chloride hexahydrate; Triphenylphosphine oxide In ethanol for 0.166667h; Stage #2: 2,6-dichloro-4-nitrophenol With triethylamine In acetonitrile for 0.0833333h; | 61.8% |

-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
16858-01-8
tris[(2-pyridylmethyl)amine]

| Conditions | Yield |
|---|---|
| Stage #1: dysprosium(III) chloride hexahydrate; tris[(2-pyridylmethyl)amine] In ethanol for 0.166667h; Stage #2: 2,6-dichloro-4-nitrophenol With triethylamine In ethanol; acetonitrile Solvent; | 58.4% |


| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 80℃; under 760.051 Torr; for 72h; Autoclave; | 53% |

-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
62921-74-8
2-(2-(2-methoxyethoxy)ethoxy)ethyl p-toluenesulfonate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 60℃; for 144h; | 53% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
100-39-0
benzyl bromide

-
-
848133-03-9
2-(benzyloxy)-1,3-dichloro-5-nitrobenzene

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 20℃; | 52% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 80℃; under 760.051 Torr; for 72h; Autoclave; | 52% |

-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
32527-15-4
1,6-bis(p-toluenesulfonyloxy)-2,4-hexadiyne

-
-
114464-05-0
C18H8Cl4N2O6

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 41h; Ambient temperature; | 50% |
-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
114464-05-0
C18H8Cl4N2O6

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone | 50% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 80℃; under 760.051 Torr; for 72h; Autoclave; | 48% |


| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 80℃; under 760.051 Torr; for 72h; Autoclave; | 48% |

-
-
618-80-4
2,6-dichloro-4-nitrophenol

-
-
109384-19-2
t-butyl 4-hydroxy piperidine-1-carboxylate

-
-
337520-58-8
4-(1-t-Butoxycarbonylpiperidin-4-yloxy)-3,5-dichloronitrobenzene

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In dichloromethane at 20℃; for 18h; Mitsunobu reaction; | 46% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 0 - 60℃; for 6h; | 39% |
| With potassium carbonate | |
| With potassium carbonate In acetone for 6h; Heating; | |
| With potassium carbonate In N,N-dimethyl-formamide at 0 - 60℃; for 1.5h; |

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-dichloro-4-nitrophenol With triethylamine In acetonitrile for 0.0833333h; Stage #2: dysprosium(III) chloride hexahydrate; tris[(2-pyridylmethyl)amine] In methanol; acetonitrile Stage #3: dichloromethane | 37% |


| Conditions | Yield |
|---|---|
| With triethylamine In ethanol; acetonitrile | 36% |

2,6-Dichloro-4-nitrophenol Specification
The systematic name of this chemical is 2,6-Dichloro-4-nitrophenol. With the CAS registry number 618-80-4 and EINECS registry number 210-563-6, it is also named as 4-Nitro-2,6-dichlorophenol. And the molecular formula of this chemical is C6H3Cl2NO3. It is a kind of yellow crystalline powder, and belongs to the product categories: Organic Building Blocks; Oxygen Compounds; Phenols.
The physical properties of 2,6-Dichloro-4-nitrophenol are as following: (1)ACD/LogP: 2.88; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.2; (4)ACD/LogD (pH 7.4): -0.13; (5)ACD/BCF (pH 5.5): 1.88; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 18.14; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 4; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 55.05 Å2; (13)Index of Refraction: 1.638; (14)Molar Refractivity: 44.47 cm3; (15)Molar Volume: 123.6 cm3; (16)Polarizability: 17.62×10-24cm3; (17)Surface Tension: 63.9 dyne/cm; (18)Density: 1.682 g/cm3; (19)Flash Point: 126.3 °C; (20)Enthalpy of Vaporization: 54.53 kJ/mol; (21)Boiling Point: 285.2 °C at 760 mmHg; (22)Vapour Pressure: 0.00165 mmHg at 25°C.
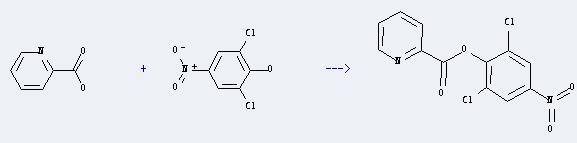
Uses of 2,6-Dichloro-4-nitrophenol: It can react with pyridine-2-carboxylic acid to produce pyridine-2-carboxylic acid 2,6-dichloro-4-nitro-phenyl ester. This reaction will need reagents 1,3-dicyclohexylcarbodiimide and 4-(dimethylamino)pyridine, and the solvent CH2Cl2. The reaction time is 48 hours with ambient temperature, and the yield is about 63%.
You should be cautious while dealing with this chemical. It irritates eyes, respiratory system and skin, and it is also harmful by inhalation, in contact with skin and if swallowed. Therefore, you had better take the following instructions: Wear suitable protective clothing, gloves and eye/face protection, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: Clc1cc(cc(Cl)c1O)[N+]([O-])=O
(2)InChI: InChI=1/C6H3Cl2NO3/c7-4-1-3(9(11)12)2-5(8)6(4)10/h1-2,10H
(3)InChIKey: PXSGFTWBZNPNIC-UHFFFAOYAC
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 61-88-1
- 6188-23-4
- 6188-25-6
- 618-83-7
- 61884-74-0
- 618-84-8
- 618-85-9
- 61886-78-0
- 618-87-1
- 618-88-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View