-
Name
2-Morpholinoethanol
- EINECS 210-734-5
- CAS No. 622-40-2
- Article Data69
- CAS DataBase
- Density 1.06 g/cm3
- Solubility miscible with water
- Melting Point -1 °C
- Formula C6H13NO2
- Boiling Point 224.4 °C at 760 mmHg
- Molecular Weight 131.175
- Flash Point 99.4 °C
- Transport Information
- Appearance clear colourless to yellow liquid
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-(4-Morpholino)ethylalcohol;2-(4-Morpholinyl)-1-ethanol;2-(4-Morpholinyl)ethanol;4-(2-Hydroxyethyl)morpholine;N-(2-Hydroxyethyl)morpholine;N-(b-Hydroxyethyl)morpholine;NSC1946;b-Morpholinoethanol;N-(2-Hydroxyethyl) Morpholine;4-Morpholineethanol;
- PSA 32.70000
- LogP -0.75120
Synthetic route

| Conditions | Yield |
|---|---|
| With triphenylphosphine; ruthenium trichloride at 120℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| at 80℃; for 72h; | 85% |
| With potassium carbonate In acetonitrile for 3h; Reflux; | 83% |
| With potassium carbonate In acetonitrile for 3h; Reflux; | 83% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In acetonitrile for 48h; Heating; | 83% |
| at 90 - 170℃; | |
| With potassium carbonate at 120℃; | |
| With ethanol; copper |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0 - 50℃; for 8h; | 76.6% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 40 - 45℃; | 67% |
| With acetone | |
| With benzene |

| Conditions | Yield |
|---|---|
| With water at 0℃; | |
| With methanol |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 160 - 170℃; | |
| With cation exchanger at 160 - 220℃; | |
| With sulfuric acid at 130 - 150℃; for 15h; |
-
-
637-39-8
triethanolamine hydrochloride

-
A
-
110-91-8
morpholine

-
B
-
622-40-2
2-(morpholin-4-yl)ethanol

-
C
-
1723-94-0
1,2-dimorpholylethane

-
D
-
6425-39-4
2,2'-dimorpholinodiethyl ether

| Conditions | Yield |
|---|---|
| at 190 - 230℃; |


| Conditions | Yield |
|---|---|
| at 200 - 205℃; under 40 Torr; anschliessend Erhitzen mit wss.-aethanol. KOH; | |
| at 200 - 205℃; under 40 Torr; | |
| at 280℃; under 7 Torr; anschliessend Erhitzen mit wss.-aethanol. KOH; |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 65 - 80℃; under 2206.5 - 3677.5 Torr; anschliessend Behandeln mit wss. NaOH; |
-
-
110-91-8
morpholine

-
-
107-21-1
ethylene glycol

-
A
-
622-40-2
2-(morpholin-4-yl)ethanol

-
B
-
1723-94-0
1,2-dimorpholylethane

| Conditions | Yield |
|---|---|
| With triphenylphosphine; ruthenium trichloride at 120℃; for 2h; Product distribution; other catalyst, also with THF, other phosphine, also at 220 degC; | |
| With hydridochlorotris(triphenylphosphine)ruthenium(II) at 100℃; for 5.5h; Yield given. Yields of byproduct given. Title compound not separated from byproducts; | |
| With ruthenium trichloride at 120℃; for 2.5h; Yield given. Yields of byproduct given. Title compound not separated from byproducts; | |
| With Ru(CO)12; triphenylphosphine In tetrahydrofuran at 220℃; for 15h; Yield given; | |
| With tris(triphenylphosphine)ruthenium(II) chloride at 100℃; for 2h; Product distribution; Mechanism; other diol; other secondary amines; other catalysts; var. temp. and reaction times; |

-
-
35855-10-8
methyl 2-morpholin-4-ylacetate

-
-
622-40-2
2-(morpholin-4-yl)ethanol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran Heating; |
-
-
50594-07-5
phenyl-carbamic acid-(2-morpholino-ethyl ester); hydrochloride

-
A
-
622-40-2
2-(morpholin-4-yl)ethanol

-
B
-
124-38-9
carbon dioxide

-
C
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 40℃; Rate constant; Thermodynamic data; var. temp., EA; |


| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In ethanol for 12h; Reflux; |
-
-
102-71-6
triethanolamine

-
-
141-43-5
ethanolamine

-
A
-
622-40-2
2-(morpholin-4-yl)ethanol

-
B
-
111-42-2
2,2'-iminobis[ethanol]

| Conditions | Yield |
|---|---|
| With oxygen; zirconium(IV) oxide In water at 325℃; under 760.051 Torr; Product distribution / selectivity; |

-
-
6342-79-6
1,2-dimorpholinoethane-1,2-dione

-
A
-
622-40-2
2-(morpholin-4-yl)ethanol

-
B
-
51068-78-1
2-hydroxy-1-(morpholin-4-yl)ethan-1-one

-
C
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| With hydrogen; Cu/Al2O3/SiO2 In methanol at 170℃; under 41254.1 Torr; for 16.5h; Autoclave; |

-
-
102-71-6
triethanolamine

-
A
-
622-40-2
2-(morpholin-4-yl)ethanol

-
B
-
6425-39-4
2,2'-dimorpholinodiethyl ether

| Conditions | Yield |
|---|---|
| With sulfuric acid at 130 - 200℃; for 15h; Temperature; Time; |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
89583-07-3
N-(2-bromoethyl)morpholine

| Conditions | Yield |
|---|---|
| With dibromotriphenylphosphorane In dichloromethane at 0 - 20℃; | 100% |
| With phosphorus tribromide In dichloromethane at 0 - 45℃; | 23.7% |
| With hydrogen bromide at 130℃; |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
95925-60-3
4-(2-hydroxyethyl)morpholine 4-oxide

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide | 100% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
448-19-1
4-fluoro-2-methoxy-1-nitrobenzene

-
-
342435-24-9
4-(2-(3-methoxy-4-nitrophenoxy)ethyl)morpholine

| Conditions | Yield |
|---|---|
| With potassium hydroxide; tetrabutylammomium bromide In water; toluene at 18 - 60℃; for 22h; | 100% |
| With tetrabutylammomium bromide; water; potassium hydroxide In toluene at 60℃; for 18h; | 98% |
| With benzyltriethylammonium bromide; potassium hydroxide In water; toluene at 60℃; for 24h; | 42.4% |
| With tetrabutyl-ammonium chloride; potassium hydroxide In toluene at 60℃; for 24h; | 42% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
106-41-2
4-bromo-phenol

-
-
836-59-9
1-bromo-4-(2-morpholinoethoxy)benzene

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 110℃; for 17h; Mitsunobu Displacement; Inert atmosphere; | 100% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; for 2.16667h; |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
851728-86-4
ethyl 1-(2-chlorophenyl)-5-(4-chlorophenyl)-4-hydroxy-1H-pyrazole-3-carboxylate

-
-
935259-86-2
1-(2-chlorophenyl)-5-(4-chlorophenyl)-3-ethoxycarbonyl-4-(2-morpholinoethoxy)-1H-pyrazole

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 20℃; | 100% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
62574-66-7
3-chlorosulfonyl-4-chlorobenzoyl chloride

-
-
1135370-76-1
2-(morpholin-4-yl)ethyl 4-chloro-3-chlorosulfonylbenzoate

| Conditions | Yield |
|---|---|
| In dichloromethane for 12h; | 100% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
1169860-18-7
2-morpholin-4-yl-potassium ethoxide

| Conditions | Yield |
|---|---|
| With potassium In ethanol at 0℃; Product distribution / selectivity; | 100% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol


| Conditions | Yield |
|---|---|
| With sodium at 20℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-(morpholin-4-yl)ethanol With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.166667h; Stage #2: 1,2-dichloro-4-fluoro-5-nitrobenzene In tetrahydrofuran; mineral oil at 0℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; caesium carbonate at 110℃; for 0.5h; Microwave irradiation; | 100% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
221040-07-9
4-benzyloxy-2-fluoro-1-nitrobenzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-(morpholin-4-yl)ethanol With sodium hydride In tetrahydrofuran; mineral oil at 20 - 30℃; for 1h; Stage #2: 4-benzyloxy-2-fluoro-1-nitrobenzene In tetrahydrofuran; mineral oil at 20℃; for 1h; | 99.2% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

| Conditions | Yield |
|---|---|
| 99% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
867187-72-2
6-(2-morpholine-4-ylethoxy)-1-oxo-3-phenyl-1H-indene-2-carboxylic acid isopropyl amide

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran; benzene at 0 - 20℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| In toluene for 120h; Reflux; | 98% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
41288-96-4
6-chloropyridine-3-ol

-
-
879487-97-5
4-(2-((6-chloropyridin-3-yl)oxy)ethyl)morpholine

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 20℃; for 4h; | 97% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
16004-15-2
1-bromomethyl-4-iodobenzene

| Conditions | Yield |
|---|---|
| at 72℃; for 18h; | 97% |
| In N,N-dimethyl-formamide at 72℃; Inert atmosphere; | 97% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
95-16-9
1,3-Benzothiazole

-
-
26278-79-5
2-amino-6-hydroxybenzothiazole

-
-
1972-28-7
diethylazodicarboxylate

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; triphenylphosphine In tetrahydrofuran; ethyl acetate | 96% |
| With sodium hydrogencarbonate; triphenylphosphine In tetrahydrofuran; ethyl acetate | 96% |
| With sodium hydrogencarbonate; triphenylphosphine In tetrahydrofuran; ethyl acetate | 96% |

-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
400-74-8
2-Fluoro-5-nitrobenzotrifluoride

-
-
850797-92-1
4-[2-(4-nitro-2-trifluoromethyl-phenoxy)-ethyl]-morpholine

| Conditions | Yield |
|---|---|
| Stage #1: 2-(morpholin-4-yl)ethanol With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.5h; Stage #2: 2-Fluoro-5-nitrobenzotrifluoride In tetrahydrofuran at 20℃; for 4h; | 96% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
910912-09-3
8,8′,8″-(hydroxymethanetriyl)tris[2,2,6,6-tetramethylbenzo[1,2-d:4,5-d′]bis([1,3]dithiole)-4-carboxylic acid]

-
-
1374260-17-9
tris(2,2,6,6-tetramethyl-8-{[2-(morpholin-4-yl)ethyl]carbonyl}-benzo[1,2-d;4,5-d']bis[1,3]dithiol-4-yl)methanol

| Conditions | Yield |
|---|---|
| Stage #1: tris(8-carboxyl-2,2,6,6-tetramethylbenzo[1,2-d;4,5-d']bis[1,3]dithiol-4-yl)methanol With 1,1'-carbonyldiimidazole In tetrahydrofuran at 55℃; for 4h; Inert atmosphere; Stage #2: 2-(morpholin-4-yl)ethanol In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Stage #3: With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 24h; Inert atmosphere; | 96% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 20℃; for 3h; | 96% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol


| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 20℃; for 24h; Mitsunobu reaction; | 95% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
6702-50-7
3-hydroxy-4-methoxybenzoate

-
A
-
250726-35-3
4-Methoxy-3-(2-morpholin-4-yl-ethoxy)benzoic acid methyl ester

-
B
-
4114-28-7
diethyl hydrazodicarboxylate

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In ethyl acetate | A 95% B n/a |

-
-
622-40-2
2-(morpholin-4-yl)ethanol


| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; | 95% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
80-48-8
methyl p-toluene sulfonate

-
-
1206209-06-4
S4-(2-hydroxyethyl)-4-methylmorpholin-4-ium 4-methylbenzenesulfonate

| Conditions | Yield |
|---|---|
| In acetonitrile for 4h; Reflux; | 94.4% |
| In acetonitrile for 4h; Reflux; | 87% |
| In acetonitrile for 4h; Reflux; | 84% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
68325-15-5
3-chloro-isonicotinonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-(morpholin-4-yl)ethanol With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 1h; Stage #2: 3-chloro-isonicotinonitrile In tetrahydrofuran; mineral oil at 20℃; for 18h; | 94% |
| Stage #1: 2-(morpholin-4-yl)ethanol With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 1h; Stage #2: 3-chloro-isonicotinonitrile In tetrahydrofuran; mineral oil at 20℃; for 18h; | 94% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
867187-58-4
1-oxo-6-hydroxy-3-phenyl-1H-indene-2-carboxylic acid ethyl ester

-
-
867187-62-0
6-(2-morpholine-4-ylethoxy)-1-oxo-3-phenyl-1H-indene-2-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran; benzene at 0 - 20℃; for 2h; | 93% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran; benzene at 0 - 20℃; for 2h; | 93% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
1202634-09-0
4-(4-nitro-3-(prop-2-ynyloxy)phenoxyethyl)morpholine

| Conditions | Yield |
|---|---|
| Stage #1: 2-(morpholin-4-yl)ethanol With sodium hydride In N,N-dimethyl-formamide at 20℃; Stage #2: 4-fluoro-1-nitro-2-(prop-2-yn-1-yloxy)benzene In N,N-dimethyl-formamide at -35℃; for 6h; Inert atmosphere; | 93% |
-
-
622-40-2
2-(morpholin-4-yl)ethanol

-
-
540-38-5
4-Iodophenol

-
-
103808-71-5
4-<2-(p-iodophenoxy)ethyl>morpholine

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran; toluene at 20℃; for 17h; Cooling with ice; | 93% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In ethyl acetate | 92% |
| With diisopropyl (E)-azodicarboxylate; triphenylphosphine In tetrahydrofuran | |
| With diisopropyl (E)-azodicarboxylate; triphenylphosphine In tetrahydrofuran |
2-Morpholinoethanol Chemical Properties
IUPAC Name: 2-Morpholin-4-ylethanol
Molecular Formula: C6H13NO2
Molecular Weight: 131.17 g/mol
Molecular structure of 4-Morpholineethanol (CAS NO.622-40-2):
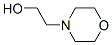
EINECS: 210-734-5
Melting Point: -1 °C
Boiling Point: 227 °C at 757 mm Hg(lit.)
Density: 1.083 g/mL at 25 °C(lit.)
Refractive Index: n20/D 1.476(lit.)
Flash Point: 211 °F
Water Solubility: miscible
Molar Refractivity: 34.48 cm3
Molar Volume: 123.6 cm3
XLogP3-AA: -0.8
H-Bond Donor: 1
H-Bond Acceptor: 3
Rotatable Bond Count: 2
Exact Mass: 131.094629
MonoIsotopic Mass: 131.094629
Topological Polar Surface Area: 32.7
Heavy Atom Count: 9
Canonical SMILES: C1COCCN1CCO
InChI: InChI=1S/C6H13NO2/c8-4-1-7-2-5-9-6-3-7/h8H,1-6H2
InChIKey: KKFDCBRMNNSAAW-UHFFFAOYSA-N
Classification Code: Mutation data; Skin / Eye Irritant
2-Morpholinoethanol Uses
4-Morpholineethanol (CAS NO.622-40-2) is commonly used in organic synthesis. It is also a kind of very important chemical pharmaceutical intermediates.
2-Morpholinoethanol Toxicity Data With Reference
| 1. | skn-rbt 10 mg/24H open | JIHTAB Journal of Industrial Hygiene and Toxicology. 30 (1948),63. | ||
| 2. | eye-rbt 20 mg SEV | AJOPAA American Journal of Ophthalmology. 29 (1946),1363. | ||
| 3. | dns-rat:lvr 5 g/L | MUREAV Mutation Research. 136 (1984),153. | ||
| 4. | orl-rat LD50:5500 mg/kg | JACTDZ Journal of the American College of Toxicology. 1 (1990),16. | ||
| 5. | ipr-mus LD50:3600 mg/kg | CANCAR Cancer. 2 (1949),1055. | ||
| 6. | scu-mus LD50:2650 mg/kg | AIPTAK Archives Internationales de Pharmacodynamie et de Therapie. 109 (1957),108. | ||
| 7. | par-mus LDLo:4800 mg/kg | CBCCT* “Summary Tables of Biological Tests” National Research Council Chemical-Biological Coordination Center. 7 (1955),691. | ||
| 8. | skn-gpg LD50:2500 mg/kg | JIHTAB Journal of Industrial Hygiene and Toxicology. 30 (1948),63. |
2-Morpholinoethanol Consensus Reports
Reported in EPA TSCA Inventory.
2-Morpholinoethanol Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S37/39:Wear suitable gloves and eye/face protection.
WGK Germany: 1
RTECS: QE3510000
F: 10-23
HS Code: 29349990
Moderately toxic by skin contact, subcutaneous, and intraperitoneal routes. Mildly toxic by ingestion and parenteral routes. Mutation data reported. A skin and severe eye irritant. Combustible when exposed to heat or flame; can react with oxidizing materials. To fight fire, use alcohol foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of NOx.
2-Morpholinoethanol Specification
4-Morpholineethanol (CAS NO.622-40-2) is also named as 2-(4-Morpholinyl)ethanol ; 2-Morpholinoethanol ; 4-(2-Hydroxyethyl)morpholine ; 4-27-00-00056 (Beilstein Handbook Reference) ; AI3-03872 ; BRN 0104375 ; Ethanol, 2-(morpholinyl)- ; Ethanol, 2-morpholino- ; Hydroxyethylmorpholine ; Morpholine ethanol ; Morpholineethanol ; N-(2-Hydroxyethyl)morfolin ; N-(2-Hydroxyethyl)morfolin [Czech] ; N-(2-Hydroxyethyl)morpholine ; N-beta-Hydroxyethylmorpholine ; NSC 1946 ; beta-Morpholinoethanol ; beta-Oxyaethyl-morpholin ; beta-Oxyaethyl-morpholin [German] ; beta-Oxyaethyl-morpholin [German] . 4-Morpholineethanol (CAS NO.622-40-2) is clear colourless to yellow liquid.
Related Products
- 2-Morpholinoethanol
- 62240-27-1
- 62240-37-3
- 62243-26-9
- 62244-44-4
- 62244-48-8
- 622-45-7
- 6224-63-1
- 622-46-8
- 62247-21-6
- 62247-32-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View